Advanced Synthetic Route for Dexpramipexole Dihydrochloride Manufacturing
The pharmaceutical landscape for neurodegenerative disease treatments has seen significant evolution with the emergence of dexpramipexole, the R-enantiomer of pramipexole, showing promise in treating amyotrophic lateral sclerosis (ALS). Patent CN103073519A introduces a pivotal advancement in the manufacturing of this critical active pharmaceutical ingredient (API) by detailing a robust method for preparing dexpramipexole hydrochloride. Unlike earlier methodologies that relied on the cumbersome resolution of racemic mixtures, this innovation utilizes a direct asymmetric reduction strategy. This approach not only streamlines the synthetic pathway but also ensures the integrity of the chiral center, which is paramount for the drug's biological efficacy. By leveraging specific reducing agents under controlled inert atmospheres, the process achieves high conversion rates while maintaining stringent purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure dexpramipexole has been fraught with inefficiencies inherent to chiral resolution techniques. Traditional methods, such as those cited in WO201110959 and EP1884514, typically involve synthesizing the racemic pramipexole first and then attempting to separate the desired R-enantiomer using chiral resolving agents. These processes are notoriously inefficient, often suffering from a theoretical maximum yield of only 50% for the desired isomer unless dynamic kinetic resolution is employed, which adds further complexity. Furthermore, the conditions required for these resolutions can be severe, involving extreme pH levels or temperatures that risk racemization, thereby compromising the optical purity of the final product. The reliance on expensive chiral auxiliaries and the generation of substantial waste from the discarded S-enantiomer make these conventional routes economically and environmentally unsustainable for large-scale commercial operations.
The Novel Approach
In stark contrast, the methodology disclosed in CN103073519A represents a paradigm shift towards efficiency and precision by employing a direct reduction of a chiral precursor. Instead of starting with a racemate, the process begins with 2-amino-6R-propionamido-4,5,6,7-tetrahydrobenzothiazole, a molecule that already possesses the correct stereochemistry at the 6-position. The core innovation lies in the selective reduction of the amide carbonyl group to a methylene group without affecting the adjacent chiral center or the sensitive benzothiazole ring system. This strategy effectively bypasses the need for resolution entirely, theoretically allowing for yields approaching 100% relative to the chiral starting material. The reaction conditions are remarkably mild, often proceeding at room temperature, which significantly reduces energy consumption and simplifies the engineering controls required for reactor safety.
Mechanistic Insights into Borohydride-Mediated Amide Reduction
The heart of this synthetic breakthrough is the sophisticated application of borohydride chemistry activated by a Lewis acid. The patent elucidates the use of sodium borohydride (NaBH4) in combination with boron trifluoride (BF3) as the preferred reducing system. Mechanistically, the Lewis acidic BF3 coordinates with the carbonyl oxygen of the propionamido group, significantly increasing the electrophilicity of the carbonyl carbon. This activation allows the hydride ion from the borohydride species to attack the carbonyl carbon more readily than it would under neutral conditions. This specific interaction is crucial because unactivated amides are generally resistant to reduction by sodium borohydride alone. The transition state formed during this hydride transfer is carefully managed to ensure that the stereochemical information at the adjacent 6R position is preserved, preventing any epimerization that could lead to the formation of the unwanted S-enantiomer impurity.
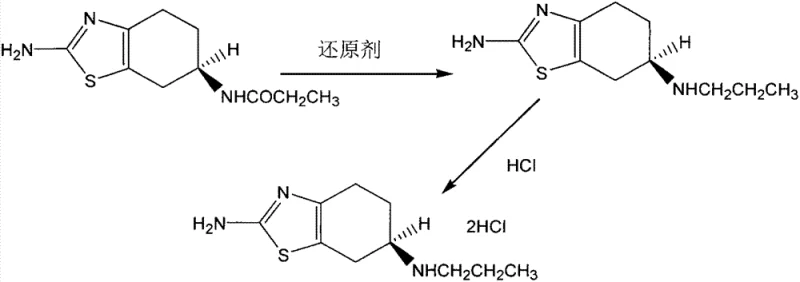
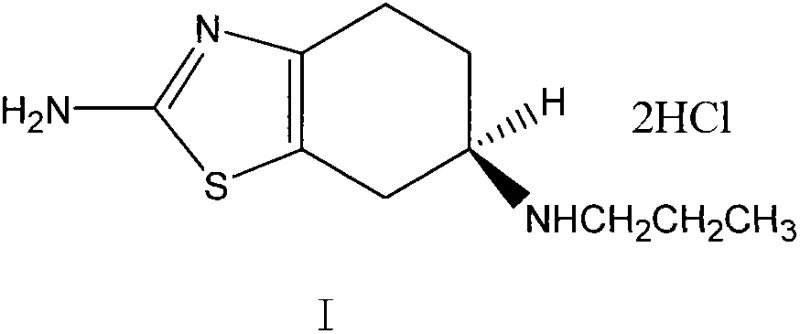
Following the reduction step, the resulting free base, 2-amino-6R-propylamino-4,5,6,7-tetrahydrobenzothiazole, is isolated and subsequently converted into its pharmaceutically acceptable salt form. The patent details the contact of this intermediate with hydrochloric acid (HCl), either as a gas or in an aqueous solution, within solvents such as methanol or acetone. This salt formation step is critical for stabilizing the molecule and improving its solubility profile for downstream formulation. The choice of solvent during this stage influences the crystallization behavior of the final dexpramipexole dihydrochloride, impacting particle size distribution and bulk density, which are key physical attributes for tablet compression. The entire sequence, from reduction to salt formation, is designed to be telescoped or performed with minimal intermediate purification, thereby reducing solvent usage and processing time while maximizing the overall throughput of the manufacturing line.
How to Synthesize Dexpramipexole Dihydrochloride Efficiently
The execution of this synthesis requires precise control over reaction parameters to ensure consistent quality and yield. The process initiates by dissolving the chiral amide starting material in an anhydrous organic solvent, with tetrahydrofuran (THF) being the preferred medium due to its excellent solubilizing properties and compatibility with the reducing agents. Under a protective atmosphere of nitrogen or argon to prevent moisture ingress and oxidation, the reducing system is introduced. The reaction temperature is maintained within a broad but controlled range, typically between -40°C and 100°C, with ambient temperature (25°C) often proving optimal for balancing reaction rate and selectivity. Once the reduction is complete, indicated by the consumption of the starting material, the mixture is worked up to isolate the amine intermediate, which is then immediately subjected to acidification to precipitate the final hydrochloride salt product.
- Dissolve 2-amino-6R-propionamido-4,5,6,7-tetrahydrobenzothiazole in an organic solvent like THF under inert gas protection.
- Add a reducing agent system, preferably Sodium Borohydride (NaBH4) combined with Boron Trifluoride (BF3), and stir at room temperature.
- Isolate the intermediate amine and react with HCl gas or aqueous HCl in a solvent like methanol to form the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits that extend beyond simple chemical transformation. By eliminating the resolution step, the process fundamentally alters the cost structure of dexpramipexole manufacturing. The removal of chiral resolving agents, which are often proprietary and expensive, directly reduces the bill of materials. Furthermore, the avoidance of discarding half the product (the unwanted enantiomer) effectively doubles the material efficiency compared to classical resolution, leading to substantial raw material savings. This efficiency gain translates into a more competitive pricing model for the final API, allowing pharmaceutical companies to better manage their cost of goods sold (COGS) while maintaining healthy margins in a competitive therapeutic market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the utilization of commodity chemicals rather than specialized chiral reagents. Sodium borohydride and boron trifluoride are widely available industrial chemicals with stable supply chains and low price volatility. By replacing complex enzymatic or chromatographic separation steps with a straightforward chemical reduction, the capital expenditure required for specialized equipment is significantly lowered. Additionally, the milder reaction conditions reduce the energy load on the facility, as there is no need for cryogenic cooling or high-temperature heating cycles, further contributing to operational cost reductions and a smaller carbon footprint for the manufacturing site.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the simplicity and robustness of this synthetic route. The starting materials, specifically the chiral propionamido-tetrahydrobenzothiazole, can be sourced from established suppliers of fine chemical intermediates, reducing dependency on single-source technology providers. The process tolerance to slight variations in temperature and stoichiometry ensures high batch-to-batch consistency, minimizing the risk of production delays caused by out-of-specification results. This reliability is crucial for maintaining continuous inventory levels and meeting the rigorous delivery schedules demanded by global pharmaceutical partners, ensuring that patient supply is never compromised by manufacturing bottlenecks.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The solvents employed, such as THF and methanol, are well-understood and can be efficiently recovered and recycled through standard distillation processes, minimizing hazardous waste generation. The absence of heavy metal catalysts, which often require complex and costly removal steps to meet strict residual metal limits in APIs, simplifies the downstream purification process. This ease of purification facilitates seamless scale-up from pilot plant quantities to multi-ton commercial production, allowing manufacturers to rapidly respond to market demand surges without the need for extensive process re-validation or new regulatory filings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in CN103073519A, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for procurement specialists assessing the quality and reliability of potential suppliers utilizing this technology.
Q: What is the primary advantage of this synthesis method over traditional resolution?
A: This method avoids the harsh conditions and low yields associated with resolving racemic mixtures, offering a direct route that preserves optical purity.
Q: Which reducing agents are suitable for this transformation?
A: The patent specifies several effective systems, including NaBH4/BF3, KBH4/BF3, and Lithium Aluminum Hydride, with NaBH4/BF3 being preferred for mildness.
Q: Is this process scalable for commercial production?
A: Yes, the use of common solvents like THF and mild reaction temperatures (around 25°C) makes the process highly adaptable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexpramipexole Dihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical supply chain. Our team of expert chemists has extensively analyzed the methodology described in CN103073519A and possesses the technical capability to implement this advanced reduction strategy effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of dexpramipexole dihydrochloride meets the highest international standards for safety and efficacy required by regulatory bodies worldwide.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality makes us the ideal partner for your dexpramipexole supply chain.
