Scalable Green Synthesis of 3-Chloro-2,4,5-Trifluorobenzoic Acid for Pharma
Scalable Green Synthesis of 3-Chloro-2,4,5-Trifluorobenzoic Acid for Pharma
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing critical fluoroquinolone intermediates. A recent breakthrough detailed in patent CN116178128A introduces a robust preparation method for 3-chloro-2,4,5-trifluorobenzoic acid, a key building block for next-generation antibacterial agents like Sitafloxacin. This innovation addresses long-standing challenges in the sector by shifting away from costly and complex phthalic acid derivatives toward a streamlined, four-step sequence originating from the economically viable 1,2,4-trifluorobenzene. By optimizing reaction conditions and reagent selection, this methodology not only enhances product purity to exceed 99.9% but also significantly mitigates environmental impact through reduced waste generation. For global supply chain leaders, this represents a pivotal opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials with improved cost structures and operational stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-chloro-2,4,5-trifluorobenzoic acid has relied heavily on starting materials such as 3,4,5,6-tetrafluorophthalic acid or its anhydride. As illustrated in prior art routes, these conventional pathways often necessitate harsh reaction conditions, including high-temperature decarboxylation and multiple protection-deprotection sequences that inherently lower overall atom economy. The reliance on such specialized fluorinated phthalic derivatives creates a bottleneck in the supply chain, as these precursors are not only expensive but also subject to volatile market pricing and limited availability from a small number of manufacturers. Furthermore, the traditional processes frequently generate substantial quantities of hazardous wastewater and solid residues due to the aggressive reagents required for chlorination and hydrolysis steps. This environmental burden complicates regulatory compliance and increases the total cost of ownership for downstream API manufacturers who must account for extensive waste treatment protocols.
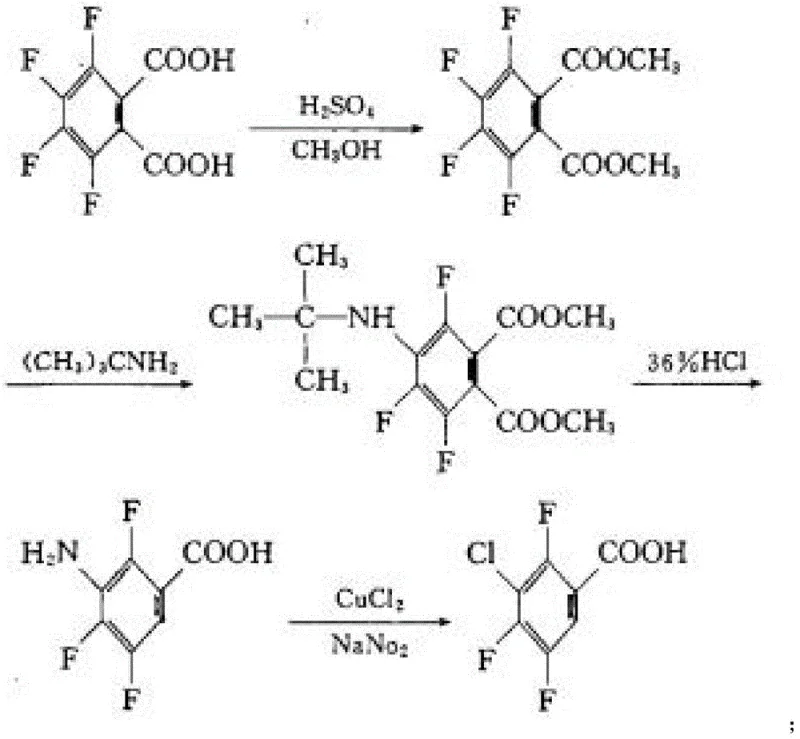
The Novel Approach
In stark contrast, the novel approach disclosed in the patent leverages 1,2,4-trifluorobenzene as a foundational feedstock, which is widely available and significantly more cost-effective than its phthalic counterparts. This strategic shift simplifies the synthetic architecture into a logical progression of bromination, selective chlorination, Grignard carboxylation, and final purification. The elimination of complex decarboxylation steps reduces the thermal load on the reactor system, thereby enhancing process safety and operational controllability. By utilizing N-chlorosuccinimide (NCS) for the chlorination step, the method achieves high regioselectivity under mild acidic conditions, avoiding the formation of poly-chlorinated by-products that typically plague free-radical chlorination methods. This streamlined workflow not only accelerates production cycles but also ensures a consistent impurity profile, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing where batch-to-batch consistency is paramount.
Mechanistic Insights into the Four-Step Synthetic Sequence
The core of this technological advancement lies in the precise control of electrophilic aromatic substitution and organometallic transformations. The process initiates with the bromination of 1,2,4-trifluorobenzene at controlled temperatures between 10°C and 30°C. Under these conditions, the electron-withdrawing fluorine atoms direct the incoming bromine electrophile to the most activated position, yielding 2,4,5-trifluorobromobenzene with high fidelity. Following isolation, the intermediate undergoes a crucial chlorination step where N-chlorosuccinimide acts as a source of electrophilic chlorine in the presence of concentrated sulfuric acid. This specific reagent combination allows for the introduction of the chlorine atom at the 3-position without disrupting the existing fluorine substituents or the bromine handle, a selectivity challenge that often requires expensive catalysts in other methodologies. The resulting 3-chloro-2,4,5-trifluorobromobenzene serves as the perfect precursor for the subsequent carbon-carbon bond formation.
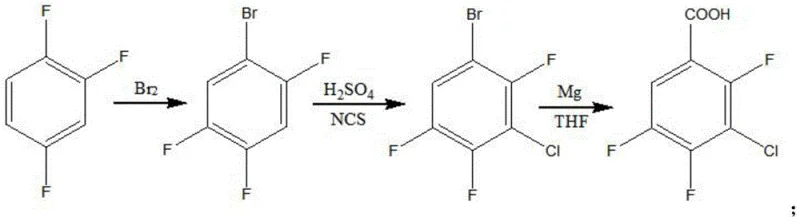
The final transformation involves the generation of a Grignard reagent using magnesium turnings in anhydrous tetrahydrofuran (THF), followed by quenching with dry ice (solid carbon dioxide). This carboxylation step effectively converts the carbon-bromine bond into a carboxylic acid group, completing the molecular skeleton of the target molecule. The reaction is carefully managed by initiating the Grignard formation at 35-40°C and then cooling to 5-10°C before the addition of dry ice to control the exotherm and prevent side reactions such as Wurtz coupling. The crude acid is then subjected to a rigorous recrystallization process using methanol and activated carbon, which removes trace organic impurities and residual metals. This multi-stage purification strategy ensures that the final product meets the stringent purity specifications required for fluoroquinolone antibiotic synthesis, demonstrating a deep understanding of impurity control mechanisms essential for regulatory approval.
How to Synthesize 3-Chloro-2,4,5-Trifluorobenzoic Acid Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and minimize by-product formation. The patent outlines a standardized protocol where the molar ratios of 1,2,4-trifluorobenzene, bromine, and N-chlorosuccinimide are balanced to ensure complete conversion at each stage. Operators must maintain anhydrous conditions during the Grignard step to prevent premature quenching of the organomagnesium species, which would drastically reduce yield. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive walkthrough of the specific operational parameters, including stirring rates, addition speeds, and workup procedures necessary for successful replication. Adhering to these guidelines allows manufacturing teams to achieve the reported yields of over 90% while maintaining the high purity standards demanded by the pharmaceutical sector.
- Bromination of 1,2,4-trifluorobenzene at 10-30°C to form 2,4,5-trifluorobromobenzene.
- Chlorination using N-chlorosuccinimide (NCS) and concentrated sulfuric acid at 5-10°C to yield 3-chloro-2,4,5-trifluorobromobenzene.
- Grignard reaction with magnesium in THF followed by carboxylation with dry ice and acidification to obtain the crude acid.
- Recrystallization from methanol with activated carbon decolorization to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this new manufacturing process offers profound advantages that directly address the pain points of procurement managers and supply chain directors. The primary driver of value is the substitution of expensive, niche starting materials with commodity-grade 1,2,4-trifluorobenzene. This fundamental change in the bill of materials results in substantial cost savings by lowering the baseline raw material expenditure, which is often the largest component of production costs in fine chemical synthesis. Furthermore, the simplified four-step sequence reduces the number of unit operations required, leading to shorter cycle times and higher throughput capacity within existing infrastructure. By eliminating the need for complex decarboxylation reactors and extensive waste treatment associated with older phthalic acid routes, facilities can operate with greater efficiency and lower overhead, translating into a more competitive pricing structure for the final intermediate without compromising on quality.
- Cost Reduction in Manufacturing: The transition to 1,2,4-trifluorobenzene as the starting material eliminates the dependency on high-cost tetrafluorophthalic acid derivatives, which are subject to supply constraints and price volatility. Additionally, the use of N-chlorosuccinimide instead of gaseous chlorine or sulfuryl chloride simplifies handling requirements and reduces the need for specialized corrosion-resistant equipment, further driving down capital and operational expenditures. The high yield reported in the patent examples indicates that less raw material is wasted per kilogram of product, enhancing the overall material efficiency of the plant.
- Enhanced Supply Chain Reliability: Sourcing 1,2,4-trifluorobenzene is significantly more straightforward than procuring specialized fluorinated phthalic anhydrides, as it is a bulk chemical produced by multiple vendors globally. This diversification of the supply base mitigates the risk of single-source bottlenecks and ensures continuity of supply even during market disruptions. The robustness of the reaction conditions, which do not require extreme pressures or cryogenic temperatures beyond standard industrial capabilities, means that the process can be easily transferred between different manufacturing sites, providing flexibility in production planning and inventory management.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, generating minimal amounts of hazardous waste compared to traditional methods. The absence of heavy metal catalysts and the use of recyclable solvents like THF and methanol simplify the effluent treatment process, helping manufacturers meet increasingly strict environmental regulations. The mild reaction temperatures and atmospheric pressure operations reduce energy consumption, contributing to a lower carbon footprint for the manufacturing site. This environmental compatibility not only reduces disposal costs but also aligns with the sustainability goals of major pharmaceutical customers who prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this critical intermediate. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this new supply source into your existing manufacturing workflows. The answers highlight the specific advantages of the new route over legacy technologies, focusing on purity, scalability, and regulatory compliance.
Q: Why is the new synthesis route superior to traditional phthalic acid methods?
A: Traditional methods often rely on expensive 3,4,5,6-tetrafluorophthalic acid derivatives which involve complex decarboxylation steps and generate significant waste. The new route utilizes cost-effective 1,2,4-trifluorobenzene, simplifying the process to four mild steps with yields over 90% and purity exceeding 99.9%.
Q: What are the critical quality control parameters for this intermediate?
A: The process emphasizes strict GC tracking throughout the reaction to ensure maximum conversion. The final product undergoes recrystallization to remove trace impurities, achieving pharmaceutical-grade purity suitable for fluoroquinolone antibiotic synthesis like Sitafloxacin.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are mild (temperatures between 5°C and 50°C) and utilize standard reagents like NCS and Magnesium. The absence of volatile or highly unstable intermediates makes it safe and operable for pilot and commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-2,4,5-Trifluorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. We are committed to delivering products with stringent purity specifications and supporting our clients with rigorous QC labs that verify every batch against the highest industry standards. Our facility is equipped to handle the specific reagents and conditions required for this green synthesis route, guaranteeing a stable and continuous supply of 3-chloro-2,4,5-trifluorobenzoic acid for your fluoroquinolone projects.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project needs. Let us partner with you to drive efficiency and reliability in your pharmaceutical manufacturing operations.
