Advanced Manufacturing of Quaternary Phosphonium Salts for Vitamin A and Carotenoid Synthesis
Introduction to Advanced Phosphonium Salt Manufacturing
The production of high-purity quaternary phosphonium salts represents a critical bottleneck in the synthesis of complex polyunsaturated organic materials, particularly within the pharmaceutical and nutraceutical sectors. Patent CN1894265A introduces a transformative methodology that addresses the longstanding challenges of solubility and yield loss associated with traditional synthesis routes. By utilizing a specifically engineered ternary solvent mixture comprising water, an alcohol, and a hydrocarbon, this innovation enables the reaction of tertiary phosphines with unsaturated electrophiles under conditions that prevent undesirable precipitation. This technical advancement is not merely a laboratory curiosity but a robust industrial solution designed to enhance the efficiency of producing key intermediates for Vitamin A and carotenoid manufacturing. For R&D directors and procurement specialists, understanding the mechanistic advantages of this solvent system is essential for optimizing supply chains and reducing the cost of goods sold for high-value active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of phosphonium salts has been plagued by significant processing inefficiencies stemming from the physicochemical properties of the products themselves. In conventional single-solvent systems, such as pure acetone or methanol, the resulting quaternary salts often exhibit low solubility, leading to premature crystallization or precipitation during the reaction phase. This phenomenon creates severe operational hurdles, including reactor fouling, difficult filtration processes, and substantial mechanical losses of the product trapped in filter cakes. Comparative data from the patent highlights these deficiencies starkly; for instance, performing the reaction in acetone resulted in a yield of only 69.2%, primarily due to these precipitation issues. Furthermore, traditional methods frequently require excessive volumes of solvent to keep the product in solution, which escalates both raw material costs and the environmental burden of waste disposal, making them economically unsustainable for large-scale commercial operations.
The Novel Approach
The novel approach detailed in the patent circumvents these limitations through the strategic implementation of a ternary solvent system, typically composed of methanol, heptane, and water. This specific combination creates a unique solvation environment that accommodates both the organic reactants and the ionic product effectively. By balancing the polarity of the mixture, the process ensures that the phosphonium salt remains dissolved throughout the reaction, thereby eliminating the formation of solid precipitates that hinder processing. The impact on efficiency is profound, with experimental results demonstrating yields reaching up to 99.9% when using the optimized ternary mixture, compared to significantly lower figures in standard solvents. This method not only simplifies the work-up procedure by avoiding complex filtration steps but also facilitates the direct recycling of the solvent mixture, offering a sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates without compromising on purity or throughput.
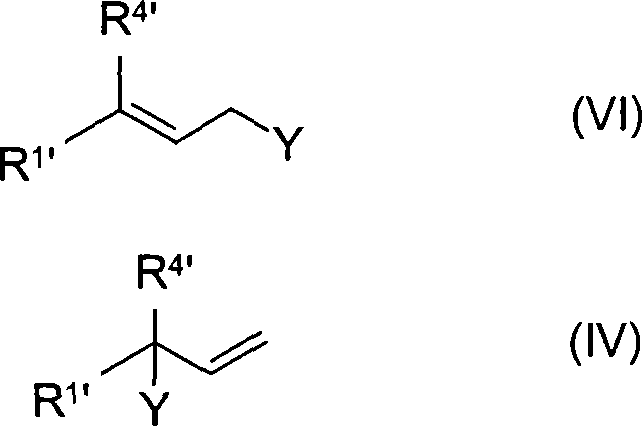
Mechanistic Insights into Ternary Solvent Stabilization
The core mechanism driving the success of this invention lies in the delicate thermodynamic balance achieved by the three-component solvent system. In a standard polar solvent, the ionic nature of the phosphonium salt drives it out of solution as the concentration increases, but the addition of a non-polar hydrocarbon component like heptane modifies the solvation shell surrounding the ions. The water and alcohol components provide the necessary dielectric constant to stabilize the charged phosphonium species, while the hydrocarbon phase ensures compatibility with the lipophilic tertiary phosphine and the unsaturated electrophilic reagents. This biphasic or multiphasic capability allows the reaction to proceed homogeneously or in a controlled heterogeneous manner, preventing the localized supersaturation that triggers nucleation and crystal growth. Consequently, the reaction kinetics are optimized, and the integrity of sensitive functional groups, such as conjugated double bonds found in vitamin precursors, is preserved against degradation that might occur in harsher, single-phase environments.
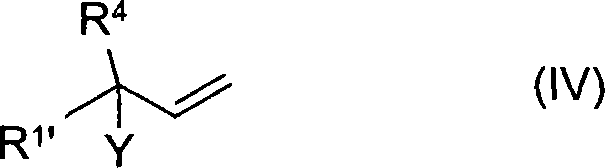
Furthermore, the impurity profile of the final product is significantly improved due to the enhanced solubility characteristics of the medium. In traditional processes, co-precipitation of side products or unreacted starting materials often occurs alongside the desired salt, necessitating rigorous and yield-diminishing purification steps. The ternary system keeps these potential impurities in solution or allows for their selective partitioning during the extractive work-up phase. This inherent purification effect means that the resulting phosphonium salts possess higher chemical purity directly from the reactor, reducing the need for downstream recrystallization. For quality control teams, this translates to a more consistent impurity spectrum and a reduced risk of batch-to-batch variability, which is critical when these intermediates are destined for the synthesis of regulated substances like high-purity OLED material precursors or pharmaceutical actives where trace contaminants can have significant biological implications.
How to Synthesize Quaternary Phosphonium Salts Efficiently
The execution of this synthesis requires precise control over solvent ratios and addition rates to maintain the stability of the ternary mixture. The process begins with the preparation of the solvent blend, ensuring that the water content is sufficient to stabilize the ionic species without causing phase separation that would inhibit reaction kinetics. Detailed standardized operating procedures for mixing the methanol, heptane, and water components, followed by the sequential addition of the acid catalyst and electrophile, are critical for reproducibility. Operators must adhere to strict temperature protocols, typically maintaining the reaction between room temperature and 70°C, to prevent thermal degradation of the unsaturated chains. For a comprehensive guide on the exact molar ratios, stirring speeds, and quenching procedures required to achieve the reported 99.9% yields, please refer to the technical documentation below.
- Prepare the ternary solvent mixture consisting of methanol, heptane, and water in specific weight ratios to ensure phase stability.
- Charge tertiary phosphine into the solvent mixture and add acid catalyst slowly while maintaining temperature control between room temperature and 70°C.
- Introduce the unsaturated electrophile (alcohol or halide) and stir until conversion is complete, followed by extractive work-up and solvent recycling.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ternary solvent technology offers substantial strategic benefits for procurement managers and supply chain directors focused on cost optimization and reliability. The primary driver of value is the dramatic improvement in process yield, which directly correlates to a reduction in the cost per kilogram of the final intermediate. By eliminating the massive yield losses associated with precipitation and filtration in older methods, manufacturers can produce significantly more product from the same amount of raw materials. This efficiency gain is compounded by the ability to recover and reuse the solvent mixture, which drastically lowers the consumption of volatile organic compounds and reduces waste treatment expenses. These factors combine to create a leaner manufacturing process that is less susceptible to fluctuations in raw material pricing, providing a more stable cost structure for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of product loss due to precipitation translates directly into improved material efficiency. Unlike conventional methods where significant portions of the product are lost in filter cakes or mother liquors, this process retains the product in the solution phase until work-up. Additionally, the removal of transition metal catalysts or the need for extensive recrystallization steps further streamlines the operation. This qualitative improvement in process efficiency leads to substantial cost savings by maximizing the output from every batch and minimizing the expenditure on solvents and energy required for drying and purification steps.
- Enhanced Supply Chain Reliability: The robustness of the ternary solvent system enhances the predictability of production schedules. Traditional methods often suffer from batch failures or extended cycle times due to handling solid precipitates and clogged filters. By maintaining a fluid reaction mixture, the process flow becomes smoother and more amenable to automation and continuous processing techniques. This reliability ensures that delivery timelines are met consistently, reducing the risk of stockouts for downstream customers who rely on these intermediates for just-in-time manufacturing of finished pharmaceutical products or agrochemical formulations.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer and mixing issues, particularly when solids are involved. This liquid-phase methodology scales linearly with fewer engineering challenges, facilitating the transition from pilot plant to full commercial production. Moreover, the reduced solvent consumption and the capability for solvent recycling align with increasingly stringent environmental regulations. This compliance reduces the regulatory burden on the supply chain and minimizes the carbon footprint of the manufacturing process, appealing to partners committed to sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphonium salt synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, focusing on the practical implications for industrial application. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into existing production lines or new facility designs. The responses cover aspects ranging from solvent composition flexibility to the specific types of electrophiles compatible with the system.
Q: Why is a ternary solvent system preferred over single solvents for phosphonium salt synthesis?
A: Traditional single solvents like acetone often cause the product to precipitate prematurely, leading to filtration losses and processing difficulties. The ternary system (water/alcohol/hydrocarbon) maintains the salt in solution, allowing for near-quantitative yields and easier handling.
Q: What are the primary applications of the phosphonium salts produced by this method?
A: These salts serve as critical precursors for Wittig reactions used in the total synthesis of high-value polyunsaturated compounds, specifically carotenoids like beta-carotene and retinoids such as Vitamin A acetate.
Q: Can the solvent mixture be recycled in this process?
A: Yes, a key advantage of this invention is the ability to separate the ternary solvent mixture from the reaction components after completion and recycle it back into the process, significantly reducing waste and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphonium Salts Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply chain for vitamins and fine chemicals. Our technical team has extensively analyzed the potential of the ternary solvent methodology described in CN1894265A and possesses the expertise to implement these advanced protocols effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are realized in practical, large-volume manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of phosphonium salt meets the exacting standards required for subsequent Wittig olefination reactions in the synthesis of carotenoids and retinoids.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your specific product portfolio. We encourage you to request specific COA data and route feasibility assessments to validate the superior quality and consistency of our intermediates. Let us collaborate to optimize your supply chain and drive down the cost of production for your high-value end products through superior chemical engineering.
