Advanced Synthesis of Star-Shaped BODIPY-Perylene Diimide Derivatives for High-Performance Optoelectronics
Advanced Synthesis of Star-Shaped BODIPY-Perylene Diimide Derivatives for High-Performance Optoelectronics
The rapid evolution of organic photovoltaics and light-emitting diodes demands materials that combine broad spectral absorption with superior morphological stability. Patent CN113444117A introduces a sophisticated molecular architecture: a star-shaped compound where a central BODIPY (boron-dipyrromethene) core is bridged to four perylene diimide (PDI) arms. This specific configuration, represented as formula (I) in the patent documentation, addresses critical challenges in non-fullerene acceptor design by leveraging the distinct photophysical properties of both chromophores. The resulting molecule exhibits strong absorption spanning from the visible to the near-infrared region (400nm to 750nm), making it an exceptional candidate for light-harvesting antennas and active layers in organic solar cells. By integrating these two powerful fluorophores through rigid ethynyl linkers, the technology offers a pathway to tune energy levels and enhance charge transport properties essential for next-generation electronic devices.
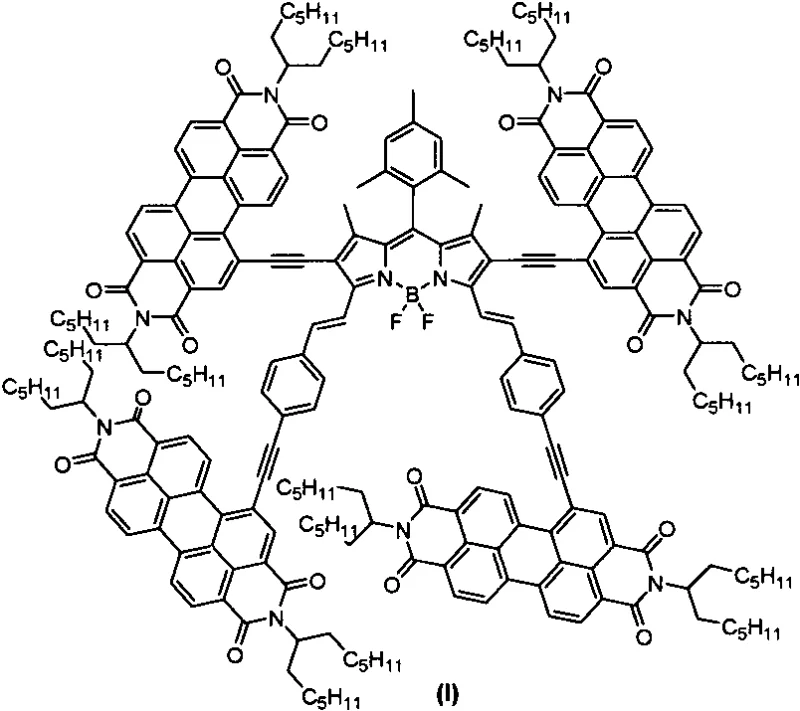
For procurement specialists and R&D teams seeking a reliable OLED material supplier, understanding the structural nuances of this invention is paramount. The patent discloses not just the final molecule, but a robust synthetic methodology that ensures reproducibility. The star-shaped topology is not merely aesthetic; it serves a functional purpose in suppressing the notorious tendency of planar PDI molecules to aggregate excessively, which often leads to phase separation issues in thin films. This innovation represents a significant step forward in cost reduction in electronic chemical manufacturing by providing a modular synthesis route that utilizes well-established coupling reactions, thereby minimizing the risk of batch-to-batch variability often associated with complex organic syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, modifying perylene diimides to extend their conjugation or alter their solubility has presented significant synthetic hurdles. Traditional bay-site functionalization often requires harsh reaction conditions that can degrade the sensitive imide rings or lead to mixtures of regioisomers that are difficult to separate. Furthermore, linear or planar extensions of the PDI core frequently exacerbate π-π stacking interactions. While strong stacking is beneficial for charge mobility in some contexts, uncontrolled aggregation in solution processing leads to large crystalline domains that disrupt the nanoscale morphology required for efficient exciton dissociation in bulk heterojunction solar cells. Conventional methods often struggle to balance the trade-off between extending the absorption spectrum and maintaining the solubility necessary for solution-based fabrication techniques like spin-coating or inkjet printing.
The Novel Approach
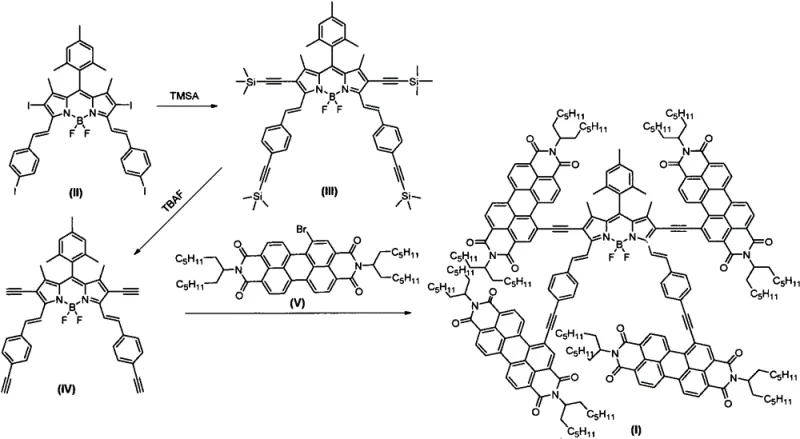
The methodology outlined in CN113444117A circumvents these issues through a strategic "core-branch" design implemented via palladium-catalyzed cross-coupling. Instead of direct fusion which might planarize the system too much, the inventors utilize a BODIPY core functionalized with terminal alkynes to couple with brominated PDI derivatives. This approach introduces a degree of torsional freedom and steric bulk via the four-arm arrangement, effectively acting as a "molecular spacer" that prevents excessive face-to-face stacking. The use of Sonogashira coupling allows for the precise construction of this complex architecture under relatively mild thermal conditions (65-85°C). This novel route ensures that the resulting high-purity organic semiconductor materials retain the excellent fluorescence quantum yield of the BODIPY parent nucleus while inheriting the high electron affinity of the PDI units, creating a push-pull system ideal for charge separation.
Mechanistic Insights into Pd-Catalyzed Sonogashira Coupling
The heart of this synthesis lies in the efficient execution of the Sonogashira coupling reaction, a cornerstone of modern organic electronics synthesis. The mechanism involves a catalytic cycle where a palladium(0) species, generated in situ from tetrakis(triphenylphosphine)palladium, undergoes oxidative addition into the carbon-iodine or carbon-bromine bonds of the BODIPY and PDI precursors respectively. Simultaneously, a copper(I) co-catalyst facilitates the activation of the terminal alkyne protons, forming a copper-acetylide species. The transmetallation step transfers the alkynyl group to the palladium center, followed by reductive elimination to forge the new carbon-carbon triple bond. This dual-catalyst system is critical for achieving the high selectivity observed in the patent, ensuring that the coupling occurs exclusively at the halogenated positions without affecting the sensitive imide functionalities or the boron-fluorine coordination in the BODIPY core.
Controlling impurities in such a multi-step synthesis is vital for device performance, as trace metal residues can act as charge traps. The patent specifies the use of standard purification techniques like silica gel column chromatography after each key transformation. In the first step, the silylation of tetraiodo-BODIPY protects the reactive alkyne termini, preventing premature polymerization or side reactions. The subsequent deprotection using tetrabutylammonium fluoride (TBAF) is a clean, high-yielding process that regenerates the terminal alkynes without disturbing the rest of the molecular framework. This modularity allows for rigorous quality control at the intermediate stage (Formula IV), ensuring that only the correct tetra-functional core proceeds to the final coupling, thereby simplifying the purification of the final star-shaped product and enhancing the overall purity profile required for commercial scale-up of complex photoelectric molecules.
How to Synthesize BODIPY Bridged Tetraperylene Diimide Derivative Efficiently
The synthesis protocol described in the patent provides a clear, step-by-step guide for reproducing this advanced material in a laboratory or pilot plant setting. The process begins with the protection of the BODIPY core, followed by deprotection and final assembly, allowing chemists to isolate and characterize intermediates to ensure quality before committing expensive PDI precursors to the final step. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and conditions required to achieve the reported results, serving as a foundational SOP for process development teams aiming to integrate this material into their device fabrication workflows.
- React tetraiodo-BODIPY with trimethylsilyl acetylene using Pd(PPh3)4 and CuI catalysts in THF/diisopropylamine at 65-85°C to form the silylated intermediate.
- Perform desilylation by treating the intermediate with tetrabutylammonium fluoride (TBAF) in THF at 0°C to generate the tetraalkynyl BODIPY core.
- Execute the final Sonogashira coupling between the tetraalkynyl BODIPY and monobromoperylene diimide derivative using Pd/Cu catalysis to yield the star-shaped target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers distinct logistical and economic benefits over more exotic material classes. The reliance on commodity chemicals such as tetrahydrofuran, triethylamine, and commercially available palladium catalysts means that the raw material supply chain is robust and less susceptible to the volatility seen with specialized reagents. Furthermore, the reaction conditions are moderate, avoiding the need for cryogenic temperatures or ultra-high pressure equipment, which translates to lower capital expenditure for manufacturing facilities. For a reliable OLED material supplier, this translates to a more predictable production schedule and the ability to respond rapidly to fluctuating market demands for organic electronic components without significant retooling costs.
- Cost Reduction in Manufacturing: The synthetic strategy eliminates the need for multiple protection-deprotection sequences on the PDI arms themselves, as the reactivity is controlled via the BODIPY core. By utilizing a convergent synthesis where four PDI arms are attached in a single final step, the process maximizes atom economy relative to linear synthesis methods. Although the patent examples demonstrate specific lab-scale yields, the use of standard catalysts like Pd(PPh3)4 at 10% loading suggests a process that can be further optimized for industrial throughput. The removal of transition metal residues is streamlined due to the solubility differences between the large star-shaped product and the catalyst ligands, reducing the cost associated with downstream purification and metal scavenging processes.
- Enhanced Supply Chain Reliability: The precursors identified in the patent, such as trimethylsilyl acetylene and monobromoperylene diimide derivatives, are established intermediates in the fine chemical industry. This ensures that reducing lead time for high-purity organic semiconductor materials is achievable because suppliers do not need to develop new supply chains for obscure starting materials. The robustness of the Sonogashira coupling also implies a tolerance to minor variations in reagent quality, providing a buffer against supply chain disruptions. This reliability is crucial for long-term contracts in the display and photovoltaic sectors where consistency is valued over marginal price fluctuations.
- Scalability and Environmental Compliance: The reaction solvents specified (THF, dichloromethane, triethylamine) are widely used in the pharmaceutical and fine chemical industries, meaning that waste streams can be managed using existing solvent recovery infrastructure. The absence of highly toxic heavy metals beyond the standard palladium/copper catalytic system simplifies environmental compliance and waste disposal protocols. As the industry moves towards greener manufacturing, the ability to recycle the organic amine bases and recover palladium from the reaction mixture adds a layer of sustainability that aligns with the corporate social responsibility goals of major electronics manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this BODIPY-PDI technology. These answers are derived directly from the mechanistic data and utility claims presented in the patent documentation, providing clarity for stakeholders evaluating this material for integration into organic photovoltaic modules or light-emitting devices. Understanding these specifics helps in assessing the feasibility of adoption and the potential performance gains over incumbent materials.
Q: What are the primary advantages of the star-shaped structure in this BODIPY-PDI derivative?
A: The four-arm star-shaped architecture effectively weakens intermolecular π-π interactions, which inhibits self-aggregation during film formation. This structural feature enhances solubility and processability while maintaining strong absorption in the visible and near-infrared regions, crucial for organic solar cells and OLED applications.
Q: How does the Sonogashira coupling strategy improve upon conventional PDI modification methods?
A: Traditional bay-site modifications can be sterically hindered or require harsh conditions. This patent utilizes a robust Sonogashira coupling strategy that links the electron-deficient PDI arms to the electron-rich BODIPY core via rigid ethynyl bridges. This ensures extended conjugation for red-shifted absorption while utilizing mild reaction conditions (65-85°C) compatible with sensitive functional groups.
Q: Is this synthesis route scalable for industrial production of organic semiconductors?
A: Yes, the synthesis relies on standard palladium-catalyzed cross-coupling chemistry using commercially available reagents like trimethylsilyl acetylene and tetrakis(triphenylphosphine)palladium. The purification involves standard silica gel column chromatography, indicating a pathway that is amenable to scale-up for producing high-purity electronic chemicals without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BODIPY Bridged Tetraperylene Diimide Derivative Supplier
As the demand for high-efficiency organic semiconductors grows, partnering with a manufacturer who understands the intricacies of complex molecular assembly is essential. NINGBO INNO PHARMCHEM stands ready to support your development pipeline with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs capable of verifying the stringent purity specifications required for electronic grade materials, ensuring that every batch of BODIPY-PDI derivative meets the exacting standards needed for high-performance OLED and OPV applications. We understand that in the fast-paced world of electronic chemicals, consistency and purity are not just metrics but the foundation of device reliability.
We invite you to engage with our technical team to discuss how this star-shaped architecture can be tailored to your specific device requirements. Whether you need a Customized Cost-Saving Analysis for large-volume procurement or require specific COA data to validate the material against your internal benchmarks, our experts are prepared to assist. Please contact our technical procurement team today to request route feasibility assessments and discover how we can accelerate your path from laboratory discovery to commercial product launch with our advanced synthesis capabilities.
