Revolutionizing Benzothiazole Ether Synthesis: A Metal-Free Photocatalytic Route for Scalable Pharma Intermediates
Revolutionizing Benzothiazole Ether Synthesis: A Metal-Free Photocatalytic Route for Scalable Pharma Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more cost-effective, and scalable synthetic routes. A recent breakthrough detailed in patent CN115353497A introduces a novel synthesis method for 2-(2-benzothiazole) substituted ethanol ether compounds that fundamentally shifts the paradigm from traditional transition metal catalysis to a metal-free photocatalytic strategy. Benzothiazole scaffolds are ubiquitous in medicinal chemistry, serving as critical structural units in a wide array of bioactive molecules, including muscarinic receptor antagonists, antifungal agents, CaS receptor allosteric modulators, and aldose reductase inhibitors.
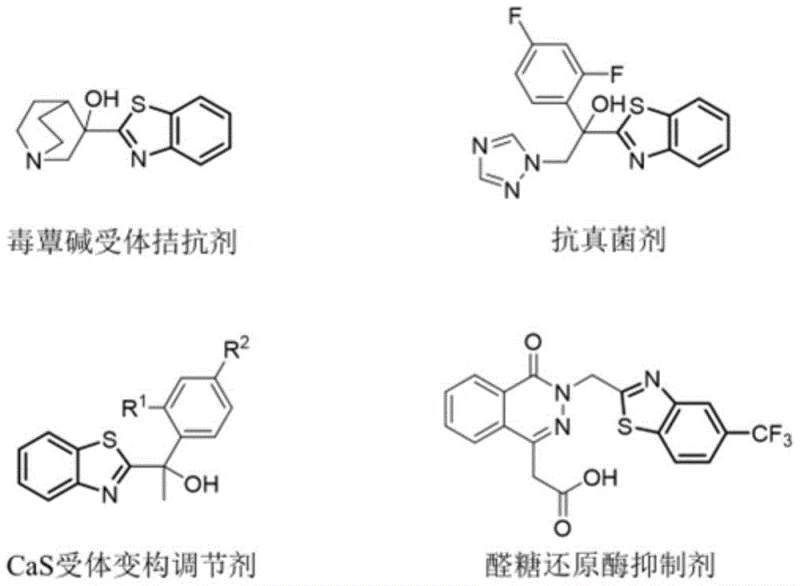
The structural modification of these compounds, particularly at the 2-position carbon, has long been a focal point for organic synthesis researchers aiming to optimize drug potency and pharmacokinetic profiles. However, achieving selective functionalization at the beta-carbon of ether compounds adjacent to these heterocycles has historically presented significant challenges due to the chemical inertness of beta-C-H bonds. This patent addresses these challenges by disclosing a robust, one-step double functionalization strategy that utilizes 6-chlorobenzothiazole, 2-methyl-2-butene, and various alcohols under light irradiation, offering a compelling solution for reliable pharmaceutical intermediate suppliers seeking to enhance their portfolio with high-value heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-C heteroaryl substituted ether compounds has relied heavily on transition metal-catalyzed C-H bond activation strategies, which often involve complex multi-step sequences and expensive reagents. As illustrated in prior art, conventional methods typically start from free alcohols and require coupling with (hetero)aryl iodides using palladium catalysts coordinated with specific directing groups (DG) and ligands. This approach necessitates the use of costly palladium sources such as Pd(OAc)2, specialized ligands, and silver salts like AgTFA, which not only drives up the raw material costs but also introduces significant downstream processing burdens.
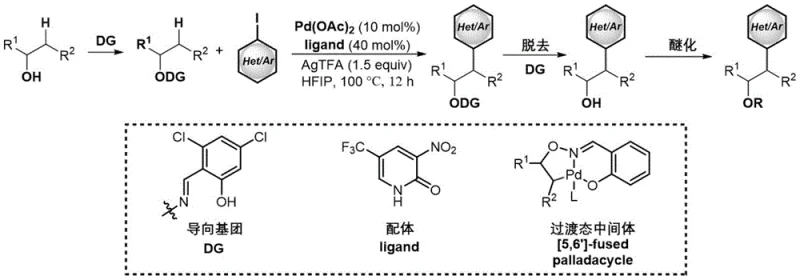
Furthermore, the requirement for pre-functionalization of substrates and the subsequent need to remove trace heavy metals to meet stringent pharmaceutical purity specifications add layers of complexity and time to the manufacturing process. The reliance on directing groups limits the substrate scope and requires additional synthetic steps to install and remove these temporary auxiliaries. Consequently, these conventional pathways often suffer from lower atom economy, higher waste generation, and reduced overall feasibility for large-scale commercial production, creating bottlenecks for supply chain heads looking to secure cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN115353497A offers a streamlined, metal-free alternative that bypasses the need for precious metal catalysts and complex ligand systems entirely. The novel approach employs a photocatalytic radical strategy where 6-chlorobenzothiazole reacts directly with 2-methyl-2-butene and a variety of alcohols in the presence of a cheap persulfate oxidant under light irradiation. This direct coupling occurs under remarkably mild conditions, typically between 25-35°C, eliminating the need for high-temperature heating or cryogenic cooling that characterizes many traditional organometallic reactions.
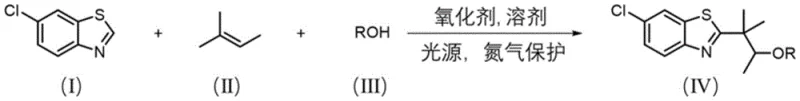
By leveraging the reactivity of olefins and the oxidative power of persulfates activated by light, this method achieves the difficult beta-C functionalization of ethers in a single operational step. The absence of transition metals means that the final products are inherently free from heavy metal contamination, a critical quality attribute for pharmaceutical intermediates. This simplicity translates directly into operational efficiency, as the workup involves standard aqueous quenching and extraction followed by chromatographic purification, drastically simplifying the process flow and enhancing the overall economic viability for industrial applications.
Mechanistic Insights into Photocatalytic Radical Alkylation
The core of this technological advancement lies in its unique mechanistic pathway, which utilizes light energy to drive the formation of reactive radical species without the need for external photocatalysts or metal mediators. Under the irradiation of a light source (blue, white, or violet), the persulfate oxidant undergoes homolytic cleavage to generate sulfate radical anions. These highly reactive species abstract hydrogen atoms from the alpha-position of the alcohol or interact with the olefin to initiate a radical cascade. The resulting carbon-centered radicals then add to the electron-deficient benzothiazole ring or couple with the alkene intermediate, facilitating the construction of the C-C and C-O bonds simultaneously.
This radical mechanism effectively overcomes the kinetic barrier associated with activating the inert beta-C-H bonds of ether compounds, a transformation that is notoriously difficult to achieve via ionic pathways. The selectivity of the reaction is governed by the stability of the intermediate radicals and the electronic properties of the benzothiazole scaffold, ensuring that the substitution occurs preferentially at the desired position. By avoiding the coordination chemistry inherent in palladium catalysis, this method sidesteps issues related to catalyst deactivation or ligand dissociation, leading to more consistent reaction performance across different batches and substrate variations.
From an impurity control perspective, the metal-free nature of the reaction significantly reduces the complexity of the impurity profile. Traditional metal-catalyzed routes often generate metal-complexed byproducts or ligand-derived impurities that are difficult to separate. In this photocatalytic system, the primary byproducts are inorganic salts derived from the persulfate oxidant, which are easily removed during the aqueous workup. This clean reaction profile contributes to the high HPLC purities observed in the examples, often exceeding 99%, thereby reducing the burden on downstream purification teams and ensuring a high-quality output suitable for sensitive pharmaceutical applications.
How to Synthesize 2-(2-Benzothiazole) Substituted Ethanol Ether Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and aligns well with standard organic synthesis protocols. The process begins by charging the key starting materials—6-chlorobenzothiazole, 2-methyl-2-butene, the chosen alcohol, and the persulfate oxidant—into a reaction vessel equipped with magnetic stirring. A suitable solvent such as acetonitrile, dimethyl sulfoxide, or ethyl acetate is added to dissolve the reagents, and the system is purged with nitrogen to create an inert atmosphere essential for radical reactions. The mixture is then subjected to light irradiation while maintaining a temperature between 25-35°C for approximately 24 hours.
- Charge 6-chlorobenzothiazole, 2-methyl-2-butene, a selected alcohol, and a persulfate oxidant into a reaction vessel with an appropriate solvent such as acetonitrile or DMSO.
- Replace the atmosphere with nitrogen three times to ensure an inert environment, then irradiate the mixture with a blue, white, or violet light source while stirring at 25-35°C.
- After 24 hours, quench the reaction with saturated sodium bicarbonate, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from metal-catalyzed to metal-free photocatalysis represents a significant opportunity for strategic cost optimization and risk mitigation. The elimination of palladium catalysts and specialized ligands removes a major cost driver from the bill of materials, as these reagents are not only expensive but also subject to volatile market pricing and supply constraints. Furthermore, the use of commodity chemicals like persulfates and simple alcohols ensures a stable and reliable supply chain, reducing the risk of production delays caused by the shortage of exotic reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the removal of expensive transition metals and the simplification of the purification workflow. Without the need for scavengers to remove residual palladium or complex chromatography to separate metal-ligand complexes, the overall processing time and consumable costs are significantly lowered. The high atom economy of the reaction means that a larger proportion of the starting materials ends up in the final product, minimizing waste disposal costs and maximizing yield efficiency per kilogram of input.
- Enhanced Supply Chain Reliability: Sourcing high-purity pharmaceutical intermediates often hinges on the availability of key reagents. This method relies on widely available bulk chemicals such as 6-chlorobenzothiazole and common alcohols, which are produced at scale by numerous global suppliers. This diversification of the supply base reduces dependency on single-source vendors for critical catalysts, thereby enhancing the resilience of the supply chain against geopolitical disruptions or logistical bottlenecks that frequently impact the availability of precious metals.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the metal-free nature of this synthesis aligns perfectly with green chemistry principles and increasingly stringent environmental regulations. The absence of heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing process. Additionally, the mild reaction conditions (ambient temperature and pressure) lower energy consumption compared to high-temperature thermal processes, making the scale-up from gram to ton quantities safer and more energy-efficient for industrial facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this novel route for commercial production.
Q: What are the primary advantages of this photocatalytic method over traditional transition metal catalysis?
A: This method eliminates the need for expensive palladium catalysts and specific ligands, significantly reducing raw material costs and simplifying the removal of toxic heavy metal residues from the final product.
Q: What types of alcohols are compatible with this synthesis protocol?
A: The protocol demonstrates broad substrate scope, successfully accommodating methyl, ethyl, isopropyl, cyclohexyl, and various hydroxy-functionalized alcohols including ethylene glycol and neopentyl glycol derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (25-35°C) without pre-functionalization of substrates, utilizing cheap persulfate oxidants and simple workup procedures, making it highly feasible for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Benzothiazole) Substituted Ethanol Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free photocatalytic technology in the production of high-value benzothiazole derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-(2-benzothiazole) substituted ethanol ether compounds delivered meets the highest industry standards for pharmaceutical intermediates.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this innovative synthesis route for their drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can accelerate your timeline to market while optimizing your overall production costs.
