Advanced Photocatalytic Synthesis of Aryl Beta-Aminoketones for Scalable Pharmaceutical Manufacturing
Advanced Photocatalytic Synthesis of Aryl Beta-Aminoketones for Scalable Pharmaceutical Manufacturing
The landscape of organic synthesis is continuously evolving towards greener, more efficient methodologies that can withstand the rigorous demands of industrial scale-up. A significant breakthrough in this domain is documented in patent CN110003110B, which discloses a novel preparation method for aryl beta-aminoketone compounds. This technology represents a paradigm shift from traditional thermal or stoichiometric activation strategies to a sophisticated photoredox catalytic system. By leveraging the inherent ring strain of electron-rich aryl cyclopropanes, this process enables selective carbon-carbon bond cleavage under remarkably mild conditions. For R&D directors and process chemists, this offers a compelling alternative to legacy routes that often suffer from harsh reaction parameters and complex waste streams. The ability to utilize visible light as the primary energy source not only aligns with modern sustainability goals but also opens new avenues for constructing complex nitrogen-containing scaffolds essential for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the ring-opening of cyclopropane derivatives to access valuable acyclic structures has been fraught with synthetic challenges. Traditional strategies often rely on the direct polarization of the cyclopropane ring using strong Lewis acids, as illustrated in early literature. 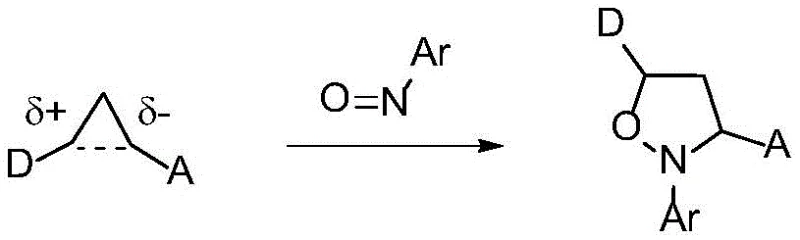 However, this approach severely restricts the substrate scope, typically requiring specific electronic configurations that are not always compatible with complex drug molecules. Furthermore, other established methods involve the use of precious transition metals. For instance, rhodium-catalyzed cycloaddition-fragmentation strategies have been employed, yet they necessitate expensive metal catalysts and often require specialized ligands to achieve selectivity.
However, this approach severely restricts the substrate scope, typically requiring specific electronic configurations that are not always compatible with complex drug molecules. Furthermore, other established methods involve the use of precious transition metals. For instance, rhodium-catalyzed cycloaddition-fragmentation strategies have been employed, yet they necessitate expensive metal catalysts and often require specialized ligands to achieve selectivity. 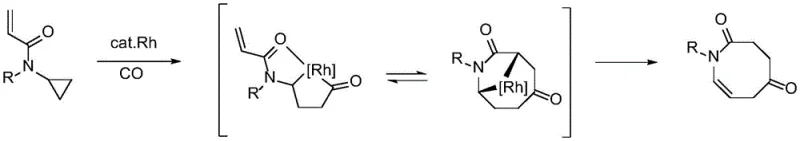 Similarly, palladium-catalyzed enantioselective ring openings via beta-carbon elimination offer stereocontrol but introduce the burden of removing trace heavy metals from the final product, a critical hurdle in pharmaceutical manufacturing.
Similarly, palladium-catalyzed enantioselective ring openings via beta-carbon elimination offer stereocontrol but introduce the burden of removing trace heavy metals from the final product, a critical hurdle in pharmaceutical manufacturing. 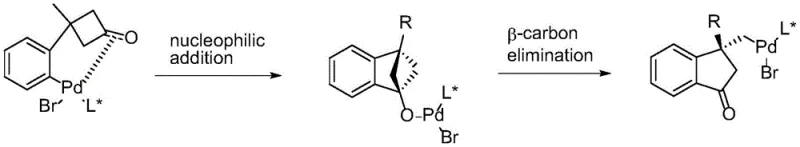 Additionally, hydroboration strategies using Lewis acid electrophilic activation have been explored, but these are often limited by the incompatibility of various affinity reagents, rendering them unsuitable for broad application.
Additionally, hydroboration strategies using Lewis acid electrophilic activation have been explored, but these are often limited by the incompatibility of various affinity reagents, rendering them unsuitable for broad application. 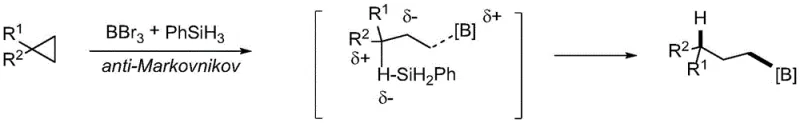 Collectively, these conventional methods share common pain points: the need for tedious pre-functionalization steps, the requirement for specific directing groups to ensure site selectivity, and the reliance on costly or toxic reagents that complicate supply chain management and environmental compliance.
Collectively, these conventional methods share common pain points: the need for tedious pre-functionalization steps, the requirement for specific directing groups to ensure site selectivity, and the reliance on costly or toxic reagents that complicate supply chain management and environmental compliance.
The Novel Approach
In stark contrast to the limitations of prior art, the methodology disclosed in CN110003110B introduces a streamlined, photocatalytic pathway that bypasses the need for pre-functionalization entirely. This novel approach utilizes electron-rich aryl cyclopropanes as the starting materials, exploiting their thermodynamic ring tension to drive the reaction forward. The core innovation lies in the use of single-electron oxidation to generate aryl radical cations, which subsequently facilitates a nucleophilic attack by an amino source. 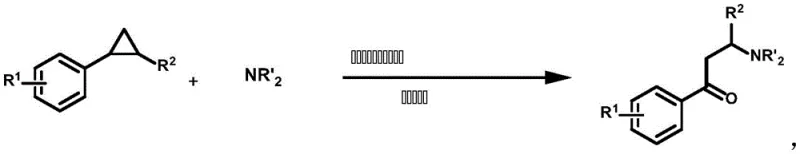 This mechanism allows for the direct conversion of simple, commercially available cyclopropanes into high-value beta-aminoketones without the need for complex protecting group manipulations. The target products, characterized by the general structure shown below, are versatile intermediates for further derivatization.
This mechanism allows for the direct conversion of simple, commercially available cyclopropanes into high-value beta-aminoketones without the need for complex protecting group manipulations. The target products, characterized by the general structure shown below, are versatile intermediates for further derivatization. 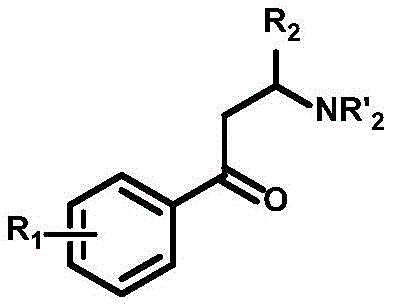 By operating at room temperature under visible light irradiation and an oxygen atmosphere, this process eliminates the energy-intensive heating or cooling cycles associated with thermal catalysis. The simplicity of the reagent system—comprising merely the substrate, an inexpensive nitrogen source like pyrazole, a drying agent, and a trace amount of photocatalyst—drastically simplifies the operational protocol, making it highly attractive for both laboratory discovery and large-scale production environments.
By operating at room temperature under visible light irradiation and an oxygen atmosphere, this process eliminates the energy-intensive heating or cooling cycles associated with thermal catalysis. The simplicity of the reagent system—comprising merely the substrate, an inexpensive nitrogen source like pyrazole, a drying agent, and a trace amount of photocatalyst—drastically simplifies the operational protocol, making it highly attractive for both laboratory discovery and large-scale production environments.
Mechanistic Insights into Photoredox-Catalyzed C-C Bond Cleavage
To fully appreciate the robustness of this synthetic route, one must delve into the intricate mechanistic details that govern the transformation. The reaction initiates with the excitation of the Iridium-based photocatalyst, [Ir(dF(CF3)ppy)2(5,5’-bpy)](PF6), upon absorption of blue light (450 nm). In its excited state, the photocatalyst acts as a potent single-electron oxidant, abstracting an electron from the electron-rich aryl moiety of the cyclopropane substrate. This critical step generates a highly reactive aryl radical cation intermediate. The formation of this radical cation significantly weakens the adjacent carbon-carbon bonds within the strained three-membered ring, effectively lowering the activation energy required for ring opening. Subsequently, the nucleophilic nitrogen source, such as pyrazole or triazole, attacks the activated benzylic position. This nucleophilic assault drives the cleavage of the C-C bond, relieving the ring strain and forming a new C-N bond. The resulting benzylic radical species then interacts with molecular oxygen present in the reaction atmosphere. This interaction leads to the formation of the ketone functionality through a radical oxidation pathway, ultimately yielding the stable aryl beta-aminoketone product after proton transfer and catalyst regeneration. This elegant cascade ensures high atom economy and minimizes the formation of unwanted byproducts, as the reaction pathway is tightly controlled by the redox potentials of the catalyst and substrates.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid or base-mediated pathways. Because the reaction proceeds via a radical manifold rather than ionic intermediates, it avoids many of the rearrangement side reactions that typically plague carbocation chemistry. The use of molecular oxygen as the terminal oxidant is particularly beneficial; it is a green reagent that produces water as the only stoichiometric byproduct, thereby avoiding the accumulation of heavy metal waste or halogenated salts often seen in stoichiometric oxidation methods. Furthermore, the mild reaction conditions (25°C) prevent thermal degradation of sensitive functional groups that might be present on the aryl ring or the amine component. This selectivity is crucial for maintaining high purity profiles, reducing the burden on downstream purification processes such as chromatography or recrystallization. The ability to tolerate a wide array of substituents—including halogens, esters, ethers, and fused polycyclic systems—demonstrates the versatility of this radical mechanism, ensuring that the process remains robust even when scaling up to multi-kilogram batches where heat and mass transfer effects can otherwise exacerbate impurity formation.
How to Synthesize Aryl Beta-Aminoketone Efficiently
Implementing this photocatalytic protocol in a practical setting requires careful attention to the reaction setup and reagent quality to ensure reproducibility and high yields. The process is designed to be operationally simple, avoiding the need for gloveboxes or strictly anhydrous conditions beyond the use of a standard drying agent. The key to success lies in the precise control of the light source and the oxygen atmosphere, which drive the catalytic cycle. Below is a summary of the standardized procedure derived from the patent examples, which serves as a foundation for process optimization.
- Under an atmospheric atmosphere, sequentially add aryl cyclopropane, an ammonia source (such as pyrazole or triazole), a drying agent (like 4A molecular sieves), and a photocatalyst ([Ir(dF(CF3)ppy)2(5,5’-bpy)](PF6)) into a solvent (1,2-dichloroethane) to form a homogeneous mixture.
- Replace the atmosphere with oxygen and irradiate the mixture with blue LED light (wavelength 450nm, intensity 8W) at room temperature for 16 to 48 hours to facilitate the single-electron oxidation and ring-opening reaction.
- Upon completion of the reaction, isolate and collect the target aryl beta-aminoketone compound from the reaction product through standard purification techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for pre-functionalized starting materials or exotic directing groups, the supply chain becomes more resilient and less susceptible to bottlenecks associated with niche chemical suppliers. The reliance on commodity chemicals like aryl cyclopropanes and simple heterocyclic amines ensures a stable and cost-effective supply base. Moreover, the removal of expensive transition metals like Rhodium and Palladium from the catalyst system represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. These precious metals are subject to volatile market pricing and require rigorous recovery protocols to meet regulatory limits; replacing them with a trace amount of an Iridium photocatalyst (used at only 0.2 mol%) drastically lowers the direct material cost and simplifies the metal clearance workflow.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of energy-intensive heating and cooling cycles. Operating at ambient temperature (25°C) using LED light sources significantly reduces the utility consumption associated with reactor heating jackets or cryogenic cooling systems. Additionally, the avoidance of stoichiometric oxidants or harsh reagents reduces the cost of waste disposal and treatment. The simplified workup procedure, which does not require complex extraction sequences to remove heavy metal residues, further contributes to overall process efficiency. By streamlining the synthesis into a single pot with minimal reagents, the labor hours required per batch are reduced, leading to substantial cost savings in terms of man-hours and facility occupancy time.
- Enhanced Supply Chain Reliability: The robustness of this method enhances supply chain continuity by reducing dependency on single-source suppliers for specialized catalysts or reagents. The use of atmospheric oxygen or pure oxygen as an oxidant removes the need for storing and handling hazardous chemical oxidants, thereby improving site safety and reducing regulatory compliance burdens. The broad substrate scope means that a single manufacturing line can potentially produce a wide variety of analogues by simply swapping the aryl cyclopropane input, providing flexibility to respond rapidly to changing market demands or clinical trial requirements without extensive re-validation of the entire process.
- Scalability and Environmental Compliance: From an environmental standpoint, this technology aligns perfectly with green chemistry principles. The use of visible light as a renewable energy source and oxygen from the air minimizes the carbon footprint of the manufacturing process. The absence of toxic heavy metal waste streams simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations. The process is inherently scalable; photochemical reactions can be efficiently scaled using flow chemistry reactors or large-scale LED arrays, ensuring that the transition from gram-scale discovery to ton-scale commercial production is seamless. This scalability ensures that the supply of high-purity intermediates can be maintained consistently, supporting long-term commercial agreements with downstream API manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the synthesis.
Q: Does this photocatalytic method require expensive transition metal catalysts like Rhodium or Palladium?
A: No, unlike conventional methods that often rely on expensive transition metals such as Rhodium or Palladium for oxidative addition, this patented process utilizes a highly efficient Iridium-based photocatalyst at an extremely low loading of only 0.2 mol%, significantly reducing raw material costs.
Q: What are the safety considerations regarding the oxygen atmosphere in this reaction?
A: The reaction operates under a standard oxygen atmosphere at room temperature using visible light as the energy source. While oxygen is required for the benzyl radical generation step, the mild conditions (25°C) and lack of high-pressure hydrogen or hazardous reagents make the process inherently safer and easier to manage compared to high-energy thermal processes.
Q: Can this method accommodate diverse substrate scopes for drug discovery?
A: Yes, the method demonstrates excellent functional group tolerance. It successfully converts a wide range of electron-rich aryl cyclopropanes, including those with halogens, esters, ethers, and fused ring systems, into the corresponding beta-aminoketones without requiring tedious pre-functionalization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Beta-Aminoketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis route described in CN110003110B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors capable of delivering uniform irradiation and precise temperature control, which are critical for maximizing the yield and purity of aryl beta-aminoketones. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards, providing you with a reliable supply of critical building blocks for your drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this green synthesis route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us help you accelerate your timeline to market with a supply chain partner committed to innovation, quality, and sustainability.
