Advanced Synthesis of Terminal Alkynyl Cyanine Dyes for High-Performance Bio-Labeling and Optoelectronics
Advanced Synthesis of Terminal Alkynyl Cyanine Dyes for High-Performance Bio-Labeling and Optoelectronics
The chemical landscape of functional dyes has evolved significantly with the disclosure of patent CN100349869C, which introduces a groundbreaking class of cyanine dyes featuring a terminal alkynyl group on the aromatic ring. This structural innovation addresses a critical limitation in traditional polymethine dyes, which often lack versatile reactive sites for downstream conjugation. By integrating a terminal alkyne moiety, these novel compounds serve not only as superior spectral sensitizers for silver halide emulsions and organic solar cells but also as pivotal intermediates for biological fluorescent labeling. The ability to tune the absorption wavelength across the visible spectrum while maintaining a reactive handle for bio-conjugation represents a substantial leap forward in material science. This technology enables the creation of highly specific probes for bioluminescence analysis and electrofax materials, bridging the gap between fine chemical synthesis and advanced optoelectronic applications.
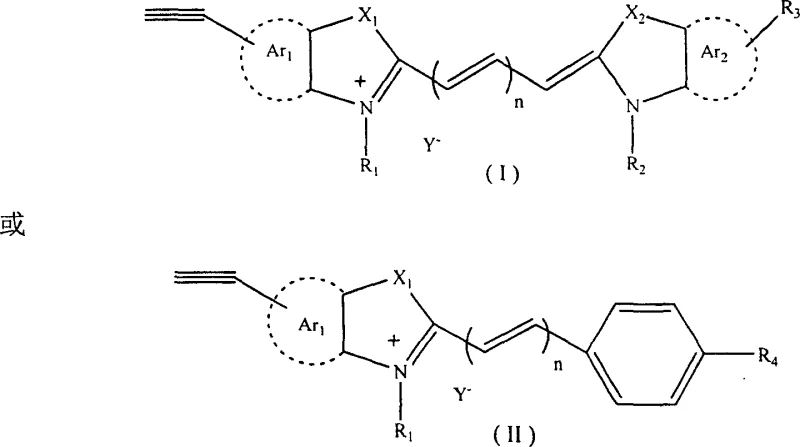
The core innovation lies in the specific molecular architecture where the terminal alkyne is strategically positioned on the aromatic nucleus, distinct from the polymethine chain. This placement ensures that the conjugated system responsible for the dye's color and fluorescence remains intact and stable, while the alkyne group remains accessible for chemical modification. The general structure encompasses a wide range of heterocyclic bases such as indoles, thiazoles, and quinolines, connected by a methine chain where n ranges from 0 to 5. This flexibility allows for precise tuning of the maximum absorption wavelength, with every two added methines shifting the absorption by approximately 100nm. Furthermore, the inclusion of various counterions like iodide, perchlorate, or sulfonates enhances solubility and thermal stability, making these dyes suitable for diverse industrial environments ranging from aqueous biological buffers to organic polymer matrices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of cyanine dyes has been plagued by significant chemical challenges, primarily due to the inherent instability of the polymethine chain under harsh reaction conditions. Traditional methods often require strong bases or high temperatures to introduce active groups, which frequently lead to the degradation of the conjugated system and the formation of complex impurity profiles. Moreover, direct substitution on the aromatic rings of pre-formed cyanine dyes is sterically hindered and electronically deactivated, resulting in poor yields and difficult purification processes. The lack of a robust synthetic route for installing reactive handles like alkynes has restricted the application of these dyes, forcing researchers to rely on less efficient post-synthetic modifications that compromise the overall purity and photophysical properties of the final product. These limitations have created a bottleneck in the supply chain for high-performance labeling reagents and optical materials.
The Novel Approach
The patented synthesis strategy overcomes these hurdles by employing a modular approach that constructs the dye skeleton around the functionalized heterocyclic precursor. Instead of attempting to modify the fragile dye molecule directly, the process first installs the terminal alkyne group onto the heterocyclic ring system via palladium-catalyzed coupling before the final condensation step. This sequence ensures that the sensitive polymethine bridge is formed only in the final stage under relatively mild conditions using acetic anhydride as both solvent and dehydrating agent. The use of acetic anhydride facilitates the condensation of the quaternary salt with the aldehyde component at temperatures between 0°C and 150°C, promoting high conversion rates without decomposing the alkyne functionality. This method significantly simplifies the workflow, allowing for the production of symmetrical and asymmetrical dyes with high structural fidelity and minimal byproduct formation.
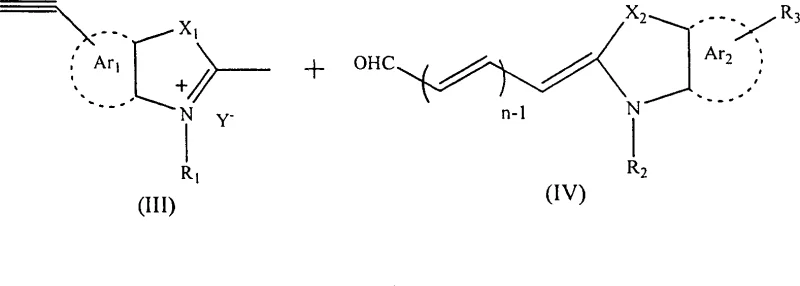
Mechanistic Insights into Heterocyclic Condensation and Alkyne Installation
The mechanistic pathway for generating these advanced dyes involves a sophisticated interplay of organometallic coupling and classical heterocyclic chemistry. The initial installation of the alkyne precursor typically utilizes a Sonogashira coupling reaction, where a bromo-substituted heterocycle reacts with a protected alkyne, such as trimethylsilylacetylene or 2-methyl-3-butyn-2-ol, in the presence of a palladium catalyst and copper iodide co-catalyst. This step is critical for establishing the carbon-carbon triple bond on the aromatic ring with high regioselectivity. Following this, a deprotection step using alkaline reagents like potassium hydroxide or potassium carbonate removes the protecting group to reveal the terminal alkyne. The resulting ethynyl-heterocycle is then quaternized with an alkyl halide to form the reactive methyl-heterocyclic salt, which serves as the nucleophile in the final condensation. This multi-step sequence ensures that the reactive alkyne is fully integrated and protected during the rigorous conditions of quaternization.
Impurity control is meticulously managed through the choice of solvents and reaction parameters during the final condensation phase. The reaction between the quaternary salt and the polymethine aldehyde proceeds via a nucleophilic attack of the methyl group on the carbonyl carbon, followed by dehydration to extend the conjugated system. By optimizing the molar ratio of reactants to 1:1 and controlling the reaction time from 5 minutes to 72 hours, the process minimizes the formation of higher-order oligomers or unreacted starting materials. The workup procedure, which involves pouring the reaction mixture into water to precipitate the solid product, leverages the low solubility of the large ionic dye molecules in aqueous media to achieve a crude separation. Subsequent recrystallization from ethanol or methanol further refines the product, removing trace organic impurities and ensuring the high purity required for sensitive applications like biological imaging and optical data storage.
How to Synthesize Terminal Alkynyl Cyanine Dyes Efficiently
The synthesis of these high-value intermediates requires precise control over reaction stoichiometry and temperature to maximize yield and purity. The process begins with the preparation of the key ethynyl-substituted heterocyclic intermediate, which is then converted into a quaternary salt. This salt is subsequently condensed with a suitable aldehyde component in a polar aprotic solvent or acetic anhydride. The detailed standardized synthesis steps involve specific molar ratios, inert gas protection, and careful temperature ramping to ensure the stability of the terminal alkyne group throughout the transformation. For a comprehensive guide on the exact operational parameters, reagent grades, and purification techniques required for GMP-compliant production, please refer to the technical protocol below.
- Perform Sonogashira coupling between a bromo-heterocycle and a protected alkyne using Pd/Cu catalysis to install the alkynyl precursor.
- Execute deprotection of the alkyne group using alkaline conditions (e.g., KOH or K2CO3) to reveal the terminal triple bond.
- Conduct quaternization with an alkyl halide followed by condensation with a polymethine aldehyde in acetic anhydride to form the final dye.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented synthesis route offers transformative advantages by streamlining the production of complex functional dyes. The reliance on widely available commodity chemicals such as acetic anhydride, simple alkyl halides, and commercially sourced heterocycles significantly reduces the raw material cost burden. Furthermore, the elimination of expensive transition metal catalysts in the final condensation step, reserving them only for the precursor synthesis where they can be recovered, drastically lowers the cost of goods sold. The robustness of the reaction conditions means that specialized high-pressure equipment is not required, allowing for production in standard glass-lined reactors which are common in fine chemical manufacturing facilities. This accessibility translates directly into a more resilient supply chain with reduced dependency on exotic reagents or specialized contract manufacturing organizations.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing acetic anhydride as a dual-purpose solvent and dehydrating agent, which eliminates the need for additional drying agents or complex azeotropic distillation setups. The high atom economy of the condensation reaction ensures that the majority of the starting mass is incorporated into the final product, minimizing waste disposal costs associated with organic solvents. Additionally, the straightforward workup involving precipitation in water avoids the need for energy-intensive chromatographic separations on a large scale, relying instead on simple filtration and recrystallization. These factors collectively contribute to a leaner manufacturing process that is highly competitive in the global market for specialty dyes and intermediates.
- Enhanced Supply Chain Reliability: The synthetic route is designed around stable, shelf-stable intermediates that can be stockpiled without significant degradation, ensuring continuity of supply even during fluctuations in raw material availability. The modularity of the synthesis allows manufacturers to produce a library of different dyes by simply varying the aldehyde component or the N-alkyl group, providing flexibility to meet diverse customer demands without retooling the entire production line. This adaptability reduces lead times for custom orders and enables rapid response to emerging trends in the biotechnology and optical storage sectors. The use of common solvents like ethanol and methanol further simplifies logistics, as these are readily available globally and do not face the same regulatory restrictions as chlorinated or aromatic solvents.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous reagents and the generation of manageable waste streams. The aqueous workup generates a filtrate that is primarily acetic acid and water, which can be easily treated or recycled, aligning with modern green chemistry principles. The solid product isolation is efficient and scalable, avoiding the bottlenecks often associated with oiling out or emulsion formation in liquid-liquid extractions. This environmental profile not only reduces the regulatory burden on the manufacturer but also appeals to downstream customers who are increasingly scrutinizing the sustainability of their supply chains. The process is inherently safe, operating at atmospheric pressure and moderate temperatures, which lowers insurance premiums and operational risks associated with high-energy chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these terminal alkynyl cyanine dyes. The answers are derived directly from the experimental data and structural claims presented in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating these materials into your existing product lines or research workflows.
Q: What is the primary advantage of introducing a terminal alkyne group to cyanine dyes?
A: The terminal alkyne group serves as a highly reactive handle for bio-conjugation, allowing the dye to be easily linked to biomolecules like proteins or DNA via click chemistry, significantly expanding its utility in biological fluorescent labeling.
Q: How does the new synthesis method improve upon traditional cyanine dye production?
A: The patented method utilizes mild condensation conditions with acetic anhydride and robust palladium-catalyzed coupling, avoiding the harsh conditions that typically degrade the sensitive polymethine chain, thereby ensuring higher purity and better stability.
Q: Can these dyes be scaled for industrial optoelectronic applications?
A: Yes, the synthesis relies on commercially available raw materials and straightforward workup procedures like filtration and recrystallization, making the process highly amenable to large-scale manufacturing for solar cells and optical recording media.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terminal Alkynyl Cyanine Dye Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced functional materials, leveraging deep expertise in heterocyclic chemistry to deliver high-purity terminal alkynyl cyanine dyes. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of both pilot-scale research and full-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity of the polymethine chain and the functionality of the terminal alkyne group. Our commitment to quality assurance guarantees that every batch meets the exacting standards required for sensitive applications in biological labeling and optoelectronic device fabrication.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target compounds. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable source of innovation that combines cutting-edge synthetic methodology with the operational excellence necessary for global supply.
