Advanced Acid-Catalyzed Synthesis of 3-Fluoro-5H-indeno[1,2-b]pyridine Derivatives for Pharmaceutical Applications
The landscape of modern pharmaceutical chemistry is increasingly defined by the strategic incorporation of fluorine atoms into heterocyclic scaffolds to enhance metabolic stability and binding affinity. Patent CN113979943A introduces a groundbreaking methodology for the construction of 3-fluoro-5H-indeno[1,2-b]pyridine compounds, a privileged structural motif known for its potent biological activities ranging from bronchodilation to antitumor effects. This innovation addresses a critical gap in the synthesis of complex fluorinated heterocycles by replacing tedious multi-step sequences with a streamlined, acid-catalyzed cascade reaction. By leveraging the unique reactivity of difluoroenol silyl ethers, this technology enables the direct assembly of the pyridine ring fused with an indene system in a single operational step. For research and development teams seeking reliable pharmaceutical intermediate suppliers, this patent represents a pivotal shift towards more efficient and atom-economical manufacturing processes that can accelerate drug discovery timelines.
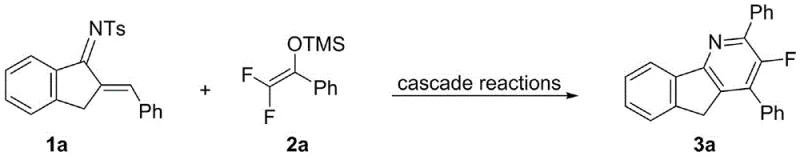
![General structural formula of the novel 3-fluoro-5H-indeno[1,2-b]pyridine compounds showing variable substituents R1, Ar1, and Ar2](/insights/img/3-fluoro-indenopyridine-synthesis-pharma-supplier-20260304234829-02.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indeno[1,2-b]pyridine core has relied on classical organic transformations that are often plagued by inefficiency and operational complexity. Traditional synthetic routes typically involve sequential Michael additions followed by intramolecular condensations, or alternatively, utilize aza-Wittig reagents reacting with alpha,beta-unsaturated ketones. These legacy methods suffer from significant drawbacks, including long synthetic linearities that accumulate yield losses at each step, poor atom utilization rates due to the generation of stoichiometric byproducts, and the frequent requirement for harsh reaction conditions or expensive transition metal catalysts. Furthermore, the introduction of fluorine atoms in specific positions using these older methodologies often requires specialized fluorinating agents that are hazardous and difficult to handle on a large scale, creating bottlenecks for cost reduction in API manufacturing and limiting the scalability required for commercial production.
The Novel Approach
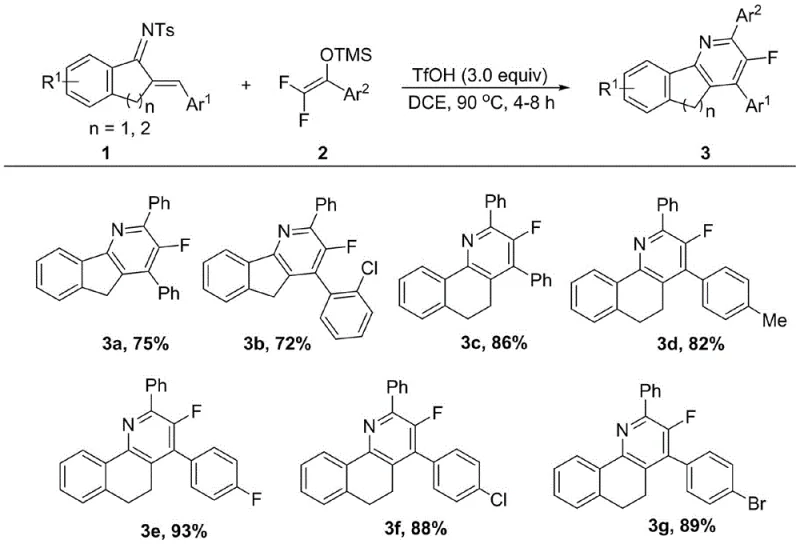
In stark contrast, the methodology disclosed in the patent utilizes a highly efficient tandem reaction strategy that merges bond formation and ring closure into a single pot. By reacting readily available aza-diene compounds with difluoroenol silyl ethers under the promotion of a strong Brønsted acid like trifluoromethanesulfonic acid (TfOH), the process achieves rapid cyclization with exceptional selectivity. This approach eliminates the need for isolating unstable intermediates and drastically simplifies the purification workflow. The reaction proceeds smoothly in common organic solvents such as 1,2-dichloroethane at moderate temperatures, offering a robust platform for generating diverse libraries of fluorinated analogues. This technological leap not only enhances the speed of synthesis but also aligns perfectly with green chemistry principles by minimizing waste and energy consumption, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into TfOH-Catalyzed Cascade Cyclization
The success of this synthetic route hinges on the precise activation of the difluoroenol silyl ether by the superacid catalyst. Mechanistically, the trifluoromethanesulfonic acid likely coordinates with the silyl ether oxygen, facilitating the departure of the silyl group and generating a highly reactive fluorinated carbocation or oxocarbenium species in situ. This electrophilic intermediate is then poised for nucleophilic attack by the electron-rich aza-diene system. The subsequent cascade involves a series of concerted electronic rearrangements, including cyclization and aromatization steps, which ultimately forge the rigid tricyclic framework of the 3-fluoro-5H-indeno[1,2-b]pyridine core. The presence of the fluorine atom at the 3-position is retained throughout this process, demonstrating the compatibility of the C-F bond with the acidic reaction conditions, which is crucial for maintaining the desired physicochemical properties of the final drug candidate.
From an impurity control perspective, the high selectivity of this acid-catalyzed pathway is paramount for ensuring product quality. The reaction conditions are tuned to favor the desired intramolecular cyclization over potential intermolecular polymerization or side reactions that could arise from the highly reactive intermediates. The use of a specific molar ratio of acid to substrates ensures complete conversion while minimizing the formation of over-reacted byproducts or decomposition products. Furthermore, the workup procedure involving a basic quench effectively neutralizes the strong acid and removes any remaining silyl-containing residues, resulting in a crude product profile that is amenable to standard purification techniques. This level of control is essential for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical ingredients, thereby reducing the burden on downstream processing.
How to Synthesize 3-Fluoro-5H-indeno[1,2-b]pyridine Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging the aza-diene and difluoroenol silyl ether into a reactor, followed by the addition of the acid catalyst and heating to promote the reaction. The simplicity of the operation allows for minimal training requirements for technical staff while ensuring reproducible results. For detailed procedural specifics regarding reagent grades, exact stirring rates, and safety protocols, please refer to the standardized synthesis guide below.
- Combine aza-diene compound and difluoroenol silyl ether in 1,2-dichloroethane (DCE) solvent within a reaction vessel.
- Add trifluoromethanesulfonic acid (TfOH) as the catalyst and stir the mixture at 90°C for 4 to 8 hours to facilitate the cascade cyclization.
- Quench the reaction with ethylenediamine and aqueous sodium hydroxide, followed by extraction, drying, and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The shift from multi-step linear syntheses to a convergent one-pot process fundamentally alters the cost structure of manufacturing these valuable intermediates. By consolidating multiple reaction steps into a single unit operation, the process significantly reduces the consumption of solvents, reagents, and labor hours, leading to substantial cost savings in production. Additionally, the elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, further streamlining the manufacturing workflow and reducing the environmental footprint associated with waste disposal.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy and the use of commercially accessible starting materials. Since the reaction achieves high conversion rates with minimal byproduct formation, the overall yield per batch is maximized, directly lowering the cost of goods sold (COGS). The avoidance of specialized fluorinating reagents, which are often costly and require special handling infrastructure, further contributes to a leaner budget. Moreover, the simplified purification process reduces the load on chromatography columns or crystallization units, allowing for faster throughput and better utilization of existing manufacturing assets without the need for significant capital expenditure on new equipment.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the reliance on robust and stable raw materials such as aza-dienes and silyl ethers, which are less prone to degradation compared to sensitive organometallic reagents used in alternative routes. The operational simplicity of the reaction, which tolerates a range of temperatures and does not require inert atmosphere conditions as strictly as some metal-catalyzed processes, reduces the risk of batch failures due to operational deviations. This reliability ensures consistent delivery schedules for downstream customers, mitigating the risks associated with supply disruptions that can plague complex pharmaceutical supply chains. The ability to source precursors from multiple vendors adds an additional layer of security to the supply network.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is facilitated by the exothermic nature of the acid-catalyzed reaction, which can be managed through standard cooling protocols in larger reactors. The absence of toxic heavy metals simplifies the regulatory compliance landscape, particularly regarding residual solvent and metal limits in the final active pharmaceutical ingredient (API). The waste stream generated is primarily organic and acidic, which can be treated using standard neutralization and incineration protocols, aligning with increasingly strict environmental regulations. This ease of scale-up ensures that the technology can seamlessly transition from R&D to commercial production, supporting the growing demand for fluorinated heterocycles in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the capabilities and limitations of the described technology. Understanding these details is crucial for partners evaluating the feasibility of integrating this synthetic route into their existing portfolios.
Q: What is the primary advantage of this new synthesis method over conventional routes?
A: The primary advantage is the significant reduction in synthetic complexity. Unlike traditional multi-step routes involving Michael additions or Wittig reactions, this method utilizes a one-pot cascade reaction that directly constructs the core skeleton from simple starting materials, resulting in higher atom utilization and operational simplicity.
Q: What yields can be expected from this acid-catalyzed process?
A: The patent data indicates excellent reaction efficiency, with isolated yields ranging significantly high across various substrates. For instance, specific derivatives like compound 3e achieved yields as high as 93%, demonstrating the robustness of the TfOH-catalyzed system for producing high-purity intermediates.
Q: Do these compounds exhibit biological activity?
A: Yes, the synthesized 3-fluoro-5H-indeno[1,2-b]pyridine derivatives have demonstrated notable bioactivity. Specifically, compounds 3b and 3e showed significant inhibitory effects on human prostate cancer cells (DU145) with IC50 values of 20.1 μM and 19.3 μM respectively, highlighting their potential as antitumor agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Fluoro-5H-indeno[1,2-b]pyridine Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthetic technology to meet your evolving needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your projects from early-stage development through to full-scale manufacturing. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in handling fluorinated chemistries allows us to navigate the complexities of this synthesis with precision, guaranteeing a consistent and reliable supply of critical building blocks for your drug discovery programs.
We invite you to collaborate with us to explore the full potential of these bioactive scaffolds in your pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and economically viable chemical solution available in the market.
