Advanced Synthesis of Porphyrin Bridged Double BODIPY Derivatives for Optoelectronics
Advanced Synthesis of Porphyrin Bridged Double BODIPY Derivatives for Optoelectronics
The rapid evolution of the optoelectronic industry demands materials that possess not only superior light-harvesting capabilities but also exceptional processability and stability. Patent CN113402536A introduces a groundbreaking chemical architecture: a porphyrin bridged double BODIPY derivative, designated as formula (I). This sophisticated molecule represents a strategic fusion of two distinct fluorophore classes—the macrocyclic porphyrin core and the rigid boron-dipyrromethene (BODIPY) units—linked through covalent bonds to create a push-pull pi-conjugated system. The resulting compound exhibits a widened absorption spectrum and robust light absorption capacity, making it an ideal candidate for next-generation solar cells, liquid crystal materials, and light storage applications. By integrating these functional groups, the invention addresses critical challenges in energy transfer efficiency and molecular stability.
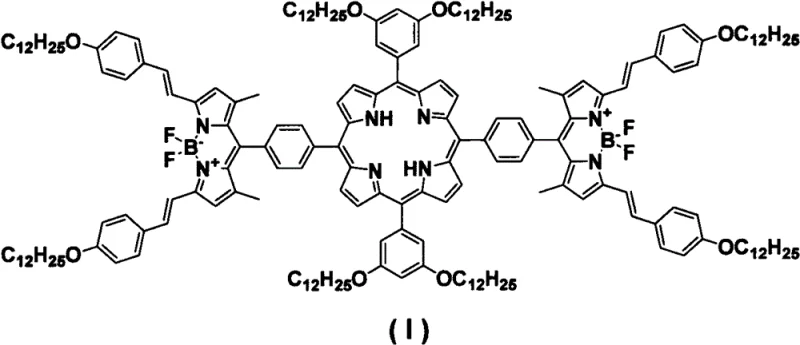
For procurement specialists and R&D teams seeking a reliable display material supplier, understanding the structural nuances of this derivative is paramount. The molecule features a central 5,15-substituted porphyrin ring connected at the 10 and 20 positions to BODIPY moieties via phenyl linkers. Crucially, the periphery is decorated with multiple dodecyloxy chains, which are not merely decorative but serve a vital function in enhancing solubility and preventing aggregation in the solid state. This structural design ensures that the material can be processed into high-quality thin films, a prerequisite for commercial device manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex donor-acceptor dyads involving porphyrins has been plagued by significant hurdles regarding reaction selectivity and purification complexity. Traditional methods often rely on harsh conditions that can degrade the sensitive BODIPY core or lead to the formation of intractable mixtures of regioisomers. Furthermore, achieving the precise stoichiometric coupling required to attach two bulky BODIPY units to a single porphyrin scaffold without over-reaction or incomplete substitution has proven difficult. Many legacy routes suffer from poor atom economy and require extensive chromatographic purification, which drastically increases the cost of goods sold (COGS) and limits the feasibility of commercial scale-up of complex organic fluorophores. Additionally, conventional derivatives often lack sufficient solubility, necessitating the use of hazardous chlorinated solvents during processing, which poses environmental and safety risks in a manufacturing setting.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by employing a modular, two-step synthetic strategy that maximizes efficiency and minimizes waste. As illustrated in the reaction scheme below, the process begins with a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. This step selectively links the brominated porphyrin precursor (II) with the boronate-functionalized BODIPY derivative (III) to form the intermediate (IV). This is followed by a Knoevenagel condensation reaction with p-dodecyloxybenzaldehyde to extend the conjugation and finalize the structure (I). This approach allows for precise control over the molecular architecture, ensuring high purity and consistent batch-to-batch reproducibility.
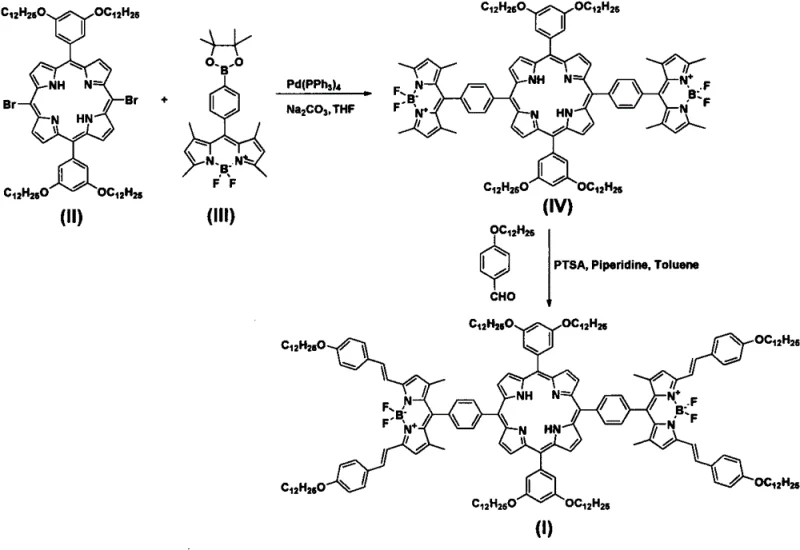
By utilizing this novel pathway, manufacturers can achieve cost reduction in optoelectronic material manufacturing through the use of readily available starting materials and standard catalytic systems. The reaction conditions are notably mild, avoiding the extreme temperatures or pressures that often compromise equipment integrity and operator safety. Furthermore, the high selectivity of the Suzuki coupling ensures that the valuable porphyrin core is preserved, reducing the loss of expensive intermediates. This streamlined process represents a significant leap forward for companies aiming to secure a stable supply chain for high-performance photoactive materials.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Knoevenagel Condensation
The success of this synthesis hinges on the precise orchestration of two distinct catalytic cycles. The first step involves the oxidative addition of the tetrakis(triphenylphosphine)palladium(0) catalyst to the carbon-bromine bonds of the porphyrin derivative (II). This generates a reactive organopalladium species which subsequently undergoes transmetallation with the boronate ester of the BODIPY derivative (III) in the presence of a base such as sodium carbonate. The final reductive elimination step forms the new carbon-carbon bond, linking the porphyrin and BODIPY units while regenerating the active palladium catalyst. This mechanism is highly tolerant of various functional groups, allowing the intricate BODIPY structure to remain intact during the coupling process. The use of a mixed solvent system comprising tetrahydrofuran, methanol, and water facilitates the solubility of both the organic substrates and the inorganic base, ensuring homogeneous reaction kinetics.
Following the coupling, the second stage employs an acid-catalyzed Knoevenagel condensation to introduce the styryl groups. In this mechanism, p-toluenesulfonic acid (PTSA) activates the aldehyde carbonyl group, making it more susceptible to nucleophilic attack by the active methylene group on the BODIPY core. The presence of piperidine acts as a co-catalyst to facilitate proton transfer steps, driving the equilibrium towards the formation of the conjugated double bond. This extension of the pi-system is critical for red-shifting the absorption spectrum, thereby capturing a broader range of solar irradiance. From an impurity control perspective, the rigidity of the final product and the distinct polarity differences between the reactants and the product allow for effective separation via silica gel chromatography, ensuring the delivery of high-purity porphyrin derivatives suitable for sensitive electronic applications.
How to Synthesize Porphyrin Bridged Double BODIPY Derivative Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production. It emphasizes the importance of maintaining an inert atmosphere, typically using argon, to prevent the oxidation of the palladium catalyst and the degradation of sensitive intermediates. The procedure details specific molar ratios, such as a 1:2.0-2.5 ratio of porphyrin to BODIPY boronate, which ensures complete consumption of the limiting porphyrin reagent. Temperature control is also critical, with the Suzuki step maintained at 70-80°C and the condensation step refluxing at 140°C in toluene. Adhering to these parameters is essential for maximizing yield and minimizing side reactions.
- Perform a Suzuki coupling reaction between 5,15-bis(3,5-bis(dodecyloxy)phenyl)-10,20-bisbromoporphyrin and 8-p-boronate phenyl BODIPY derivative using Pd(PPh3)4 catalyst in THF/MeOH/Water at 70-80°C.
- Isolate the intermediate porphyrin bridged bis-BODIPY derivative (IV) via silica gel column chromatography.
- Conduct a Knoevenagel condensation with p-dodecyloxybenzaldehyde in toluene using PTSA and piperidine at 140°C to obtain the final derivative (I).
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition from laboratory curiosity to commercial viability is often the most significant bottleneck. The synthetic route described in CN113402536A offers several tangible benefits that directly address these logistical and financial concerns. By leveraging well-established catalytic methodologies, the process reduces the dependency on exotic reagents or proprietary equipment, thereby simplifying the sourcing strategy. The ability to produce materials with enhanced solubility also translates to lower processing costs downstream, as less solvent is required for formulation and coating.
- Cost Reduction in Manufacturing: The elimination of complex multi-step protection and deprotection sequences significantly lowers the overall operational expenditure. By utilizing a direct coupling strategy, the number of unit operations is minimized, which reduces labor costs, energy consumption, and solvent usage. Furthermore, the high selectivity of the reaction reduces the burden on purification teams, allowing for faster throughput and higher asset utilization rates in the production facility. The use of standard catalysts like Pd(PPh3)4, which are commercially available in bulk, ensures that raw material costs remain predictable and manageable.
- Enhanced Supply Chain Reliability: The starting materials, including brominated porphyrins and boronate esters, are derived from commodity chemicals or established intermediate supply chains. This reduces the risk of supply disruptions associated with niche or single-source reagents. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in utility supplies, such as cooling water temperature or nitrogen pressure, ensuring consistent output. For a reliable electronic chemical supplier, this reliability is key to maintaining long-term contracts with major device manufacturers who cannot afford production delays.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like toluene and THF that are easily recovered and recycled in large-scale distillation units. The absence of heavy metal contaminants in the final product, thanks to efficient purification steps, simplifies regulatory compliance for electronics applications where trace metal content is strictly regulated. Additionally, the improved solubility of the final product reduces the need for aggressive cleaning agents in processing equipment, contributing to a greener manufacturing footprint and aligning with global sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this advanced material. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what potential partners can expect.
Q: What are the primary advantages of this porphyrin-BODIPY hybrid structure?
A: The hybrid structure combines the strong visible light absorption of porphyrins with the high fluorescence quantum yield of BODIPY, enabling efficient energy transfer processes ideal for solar cells and light-harvesting antennas.
Q: How does the synthesis method improve solubility for processing?
A: The introduction of eight dodecyl chains onto the molecular framework significantly enhances solubility in common organic solvents, facilitating solution-processing techniques required for thin-film device fabrication.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route utilizes standard palladium-catalyzed cross-coupling and acid-catalyzed condensation reactions under relatively mild conditions, making it amenable to scale-up with established purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porphyrin Bridged Double BODIPY Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of porphyrin-BODIPY hybrids in the field of renewable energy and advanced displays. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is seamless. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom modifications to the alkyl chains for specific solubility profiles or optimization of the optical bandgap, our technical capabilities are aligned with your innovation roadmap.
We invite you to collaborate with us to unlock the full commercial potential of this technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis can accelerate your product development cycle and enhance your competitive edge in the global market.
