Advanced Perylene Diimide Derivatives for High-Efficiency Optoelectronic Material Production
The rapid evolution of the optoelectronic sector demands materials that can transcend the Shockley-Queisser limit, and recent advancements documented in patent CN113354639A highlight a significant breakthrough in this domain. This intellectual property details a novel class of perylene diimide derivatives engineered specifically to induce singlet fission in the solid state, a phenomenon critical for enhancing the external quantum efficiency of organic semiconductor solar cells. By strategically modifying the bay positions of the perylene core with specific aromatic conjugated systems, these materials achieve triplet energy levels ranging from 1.10 eV to 1.28 eV, addressing the long-standing challenge of low photoelectric conversion efficiency in traditional small molecule systems. For R&D directors and procurement strategists, understanding the synthesis and application of these derivatives is paramount, as they represent a viable pathway toward industrial mass production of high-performance photoelectric materials. The technical depth of this patent provides a robust foundation for developing reliable optoelectronic material supplier networks capable of delivering next-generation components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic photovoltaic materials has been hindered by the inherent limitations of conventional perylene diimide structures, which often fail to exhibit efficient singlet fission in the film state. Traditional synthesis routes frequently result in molecules where singlet excitons decay primarily through fluorescence or non-radiative pathways, leading to fluorescence quantum yields in thin films that are significantly lower than those observed in solution states. This discrepancy indicates a lack of effective intermolecular coupling required to split singlet excitons into triplet pairs, thereby capping the potential efficiency of solar cells built upon these architectures. Furthermore, older methodologies often struggle with controlling the torsion angle of the molecule and the energy level of the frontier molecular orbitals, which are critical factors influencing molecular accumulation and electronic properties. Without precise structural modification, the resulting materials lack the necessary photo-thermal stability and electron mobility required for rigorous commercial applications, forcing manufacturers to rely on less efficient alternatives that cannot meet the escalating demands of the renewable energy market.
The Novel Approach
The innovative strategy outlined in the patent data overcomes these barriers by introducing ortho-biphenyl, meta-biphenyl, para-biphenyl, alpha-naphthalene, or beta-naphthalene aromatic conjugated systems directly into the bay positions of the perylene diimide backbone. This structural engineering expands the pi-conjugated skeleton of the entire molecule, profoundly influencing the coupling effect among perylene diimide molecules and meeting the basic energy requirements for singlet fission. As illustrated in the specific structure of PDI-C5-2 below, the precise placement of these substituents allows the material to maintain high stability while achieving fluorescence quantum yields in thin films ranging from 6% to 22%, a marked improvement driven by the generation of triplet excitons.
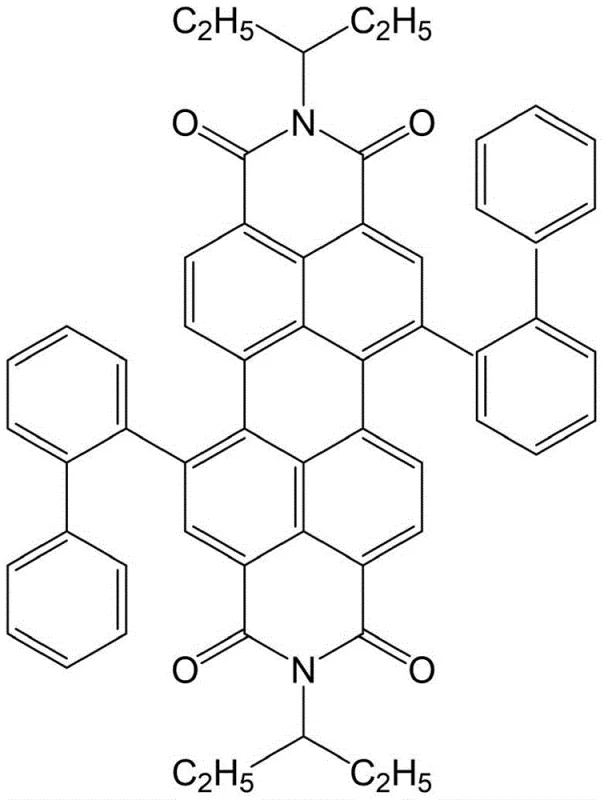
This novel approach not only enhances the photophysical properties but also simplifies the manufacturing process, utilizing a straightforward Suzuki-Miyaura coupling reaction that operates under relatively mild conditions of 125-135°C. The ability to tune the substituents allows for the customization of electronic properties, making this platform technology highly versatile for various optoelectronic applications including airport effect transistors and industrial dyes. By shifting the focus from simple core synthesis to strategic bay-functionalization, this method provides a clear route for cost reduction in electronic chemical manufacturing, offering a competitive edge for supply chain leaders seeking high-performance materials with scalable production potential.
Mechanistic Insights into Suzuki-Miyaura Bay Functionalization
The core of this technological advancement lies in the palladium-catalyzed cross-coupling mechanism that facilitates the attachment of bulky aromatic groups to the sterically hindered bay region of the perylene core. The reaction proceeds through a catalytic cycle involving oxidative addition of the 6,12-dibromo-N,N-pentane-perylene diimide to the tetrakistriphenylphosphine palladium catalyst, followed by transmetallation with the aryl boronic acid species in the presence of anhydrous potassium carbonate. This base plays a critical role in activating the boronic acid and facilitating the transfer of the organic group to the palladium center, ultimately leading to reductive elimination which forms the new carbon-carbon bond and regenerates the active catalyst. The general structural framework allowing for this diverse functionalization is depicted below, highlighting the versatility of the R group which dictates the final electronic characteristics of the derivative.
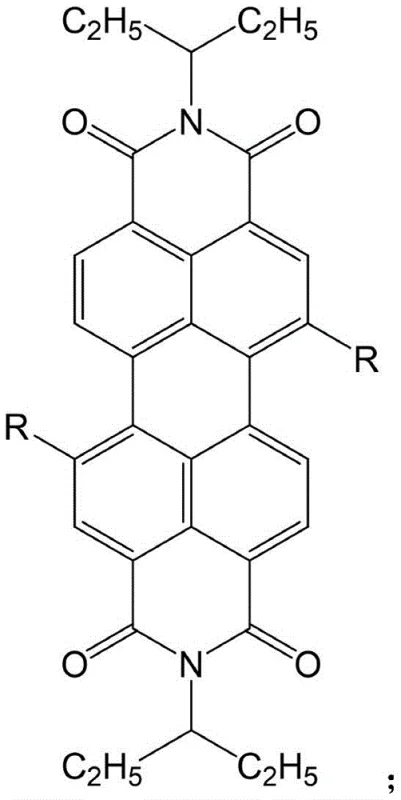
From an impurity control perspective, the mechanism is designed to minimize side reactions such as homocoupling of the boronic acid or debromination of the perylene core, which are common pitfalls in complex cross-coupling scenarios. The use of anhydrous toluene or similar aprotic solvents ensures that water-sensitive intermediates remain stable, while the specific molar concentrations of reactants—such as maintaining the dibromo-perylene diimide at 0.033 to 0.044 mol/L—optimize the collision frequency for the desired cross-coupling event. Furthermore, the purification protocol involving sequential extraction with dichloromethane, water, and saturated saline solution effectively removes residual palladium species and inorganic salts, ensuring that the final high-purity optoelectronic materials meet the stringent specifications required for device fabrication. This rigorous control over the reaction environment and workup procedure is essential for reducing lead time for high-purity display & optoelectronic materials, as it minimizes the need for extensive reprocessing.
How to Synthesize Perylene Diimide Derivatives Efficiently
The synthesis of these advanced materials follows a three-stage protocol that begins with the formation of the N,N-pentane-perylene diimide core, followed by regioselective bromination, and concludes with the pivotal coupling reaction. Each step is optimized for yield and purity, utilizing standard laboratory equipment that translates easily to pilot plant operations, ensuring that the transition from benchtop discovery to commercial scale-up of complex polymer additives and small molecules is seamless. The detailed standardized synthesis steps, including specific reagent grades, stirring rates, and temperature ramping profiles, are provided in the technical guide below to assist process engineers in replicating these results with high fidelity.
- Preparation of N,N-pentane-perylene diimide precursor via imidazole-mediated condensation at 135-150°C.
- Bromination of the perylene core at the 6,12-positions using liquid bromine and iodine catalyst in dichloromethane.
- Final Suzuki-Miyaura coupling with aryl boronic acids at 125-135°C to introduce biphenyl or naphthalene groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical performance, directly impacting the bottom line through operational efficiencies. The elimination of exotic or hard-to-source reagents in favor of commercially available boronic acids and standard palladium catalysts significantly reduces raw material volatility, ensuring a more predictable cost structure for long-term contracts. Moreover, the robustness of the reaction conditions, which tolerate a range of solvents including chloroform and tetrahydrofuran, provides flexibility in sourcing and inventory management, allowing manufacturing teams to adapt quickly to market fluctuations without compromising product quality. This resilience in the supply chain is critical for maintaining continuous production schedules in the fast-paced electronics sector.
- Cost Reduction in Manufacturing: The streamlined three-step synthesis avoids the need for expensive transition metal removal resins often required in other catalytic processes, as the palladium residues can be effectively extracted using standard dichloromethane washes. This simplification of the downstream processing workflow drastically lowers the consumption of specialized purification media and reduces the overall solvent load, leading to significant cost savings in waste treatment and material usage. Additionally, the high stability of the intermediate 6,12-dibromo-N,N-pentane-perylene diimide allows for batch storage, enabling manufacturers to decouple the initial synthesis steps from the final customization, further optimizing inventory turnover and capital allocation.
- Enhanced Supply Chain Reliability: By relying on fundamental organic building blocks like perylene tetracarboxylic anhydride and aminopentane, the production of these derivatives is not dependent on single-source proprietary intermediates that often create bottlenecks. The modular nature of the Suzuki coupling means that different aromatic substituents can be swapped in without altering the core process infrastructure, providing agility to produce various grades of high-purity OLED material or solar cell components from a single production line. This flexibility ensures that supply chain leaders can respond rapidly to changing customer specifications without incurring the delays associated with retooling or qualifying new vendors.
- Scalability and Environmental Compliance: The reaction operates at temperatures between 125-135°C, which is well within the safe operating limits of standard glass-lined or stainless steel reactors, facilitating easy scale-up from kilogram to multi-ton quantities without requiring high-pressure equipment. The use of inert gas protection and controlled tail gas treatment for bromine handling demonstrates a commitment to environmental safety, aligning with increasingly strict global regulations on volatile organic compounds and hazardous emissions. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of production shutdowns due to environmental violations, securing the long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this perylene diimide technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the feasibility of integration into existing manufacturing workflows and to highlight the specific performance metrics that differentiate this material class from conventional alternatives. Understanding these details is crucial for technical teams evaluating the potential for licensing or procurement.
Q: What is the primary advantage of bay-substituted perylene diimides in thin films?
A: The introduction of aromatic conjugated systems at the bay position facilitates singlet fission, generating higher triplet energy levels (1.10-1.28 eV) crucial for next-generation solar cells.
Q: How does the Suzuki coupling method improve yield compared to traditional methods?
A: The patented process utilizes optimized molar concentrations and inert gas protection, achieving yields of 35-40% for the final coupling step with high stability.
Q: Are these derivatives suitable for large-scale optoelectronic manufacturing?
A: Yes, the synthesis uses commercially available reagents and standard purification techniques like column chromatography, making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perylene Diimide Derivative Supplier
As the demand for high-efficiency photoelectric materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to the expertise required to translate complex patent chemistry into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee stringent purity specifications for every batch of perylene diimide derivatives we produce. We understand that consistency is key in the electronics industry, and our robust quality management systems are designed to detect and eliminate trace impurities that could compromise device performance, providing you with a reliable agrochemical intermediate supplier level of dependability for your electronic materials.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your product roadmap. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this synthetic route, along with specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a competitive advantage in the optoelectronic market with materials that combine cutting-edge performance with supply chain resilience.
