Advanced Synthesis of Chiral Binaphthyl-Pyrene-Pillararene Derivatives for High-Performance CPL Materials
The landscape of optoelectronic materials is undergoing a significant transformation with the advent of advanced chiral macrocycles capable of emitting circularly polarized luminescence (CPL). Patent CN115504885A introduces a groundbreaking binaphthyl-pyrene-pillararene derivative that addresses critical limitations in current CPL technology, specifically the low dissymmetry factor (|glum|) often observed in small molecule emitters. This innovation leverages a sophisticated molecular design that integrates three distinct functional components: a pillararene macrocycle providing a rigid chiral scaffold, binaphthol units imparting stable axial chirality, and pyrene groups delivering intense fluorescence. For R&D directors and procurement specialists seeking a reliable optoelectronic material supplier, this patent represents a pivotal shift towards high-performance materials that can be synthesized through a robust, four-step chemical pathway. The disclosed method not only achieves a remarkable |glum| value of up to 5.6×10^-3 but also utilizes widely available raw materials, suggesting a viable route for cost reduction in chiral macrocycle manufacturing without compromising on optical purity or structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic circularly polarized luminescent materials has been hindered by the inherent trade-off between chirality and fluorescence intensity. Most conventional small molecule compounds exhibit relatively low |glum| values, limiting their efficacy in high-end applications such as 3D displays, optical storage, and biological probes. Furthermore, while pillararenes offer a promising macrocyclic framework due to their unique host-guest properties and potential planar chirality, they suffer from rapid conformational interconversion in solution. This dynamic flipping of aromatic rings leads to racemization, making it exceptionally difficult to isolate stable enantiomers without extensive and costly chiral resolution processes. Additionally, the non-conjugated benzene ring skeleton of unmodified pillararenes results in weak fluorescence emission signals, which fundamentally restricts their utility as standalone emitters in photoelectric devices. These combined factors have created a bottleneck in the commercial scale-up of complex supramolecular architectures for the display industry.
The Novel Approach
The methodology outlined in CN115504885A circumvents these challenges through a strategic covalent modification strategy that locks the chiral conformation while simultaneously enhancing emissive properties. By grafting bulky binaphthyl groups onto the pillararene rim, the synthesis effectively suppresses the conformational flipping, thereby stabilizing the planar chirality of the macrocycle. Simultaneously, the introduction of pyrene fluorophores via esterification linkages dramatically boosts the fluorescence quantum yield. This dual-modification approach ensures that the resulting derivative possesses both the necessary chiral source and the strong emitting group required for superior CPL performance. The synthetic route is designed for operational simplicity, utilizing mild conditions and standard organic transformations that facilitate easier purification and higher overall yields compared to traditional multi-step supramolecular syntheses.
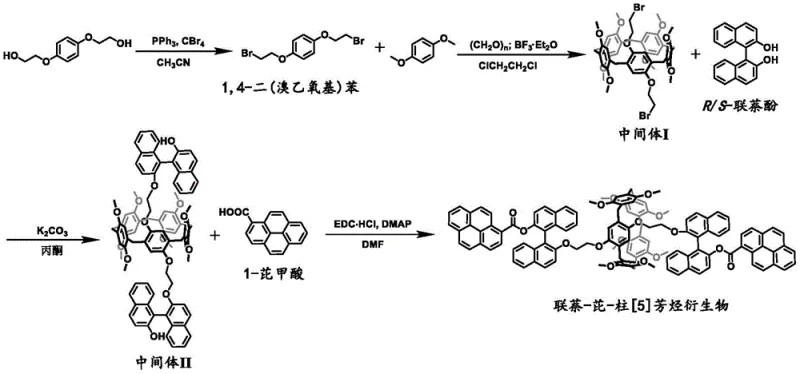
This comprehensive reaction scheme illustrates the logical progression from simple diether precursors to the final complex chiral macrocycle. The process begins with the functionalization of a benzene core, followed by the critical macrocyclization step to form the pillararene backbone. Subsequent steps involve the precise installation of chiral binaphthol units and finally the attachment of the pyrene luminophore. This modular approach allows for fine-tuning of the molecular properties and demonstrates a clear pathway for high-purity optoelectronic material production that minimizes side reactions and impurity formation.
Mechanistic Insights into Supramolecular Chirality Transfer and Fluorophore Conjugation
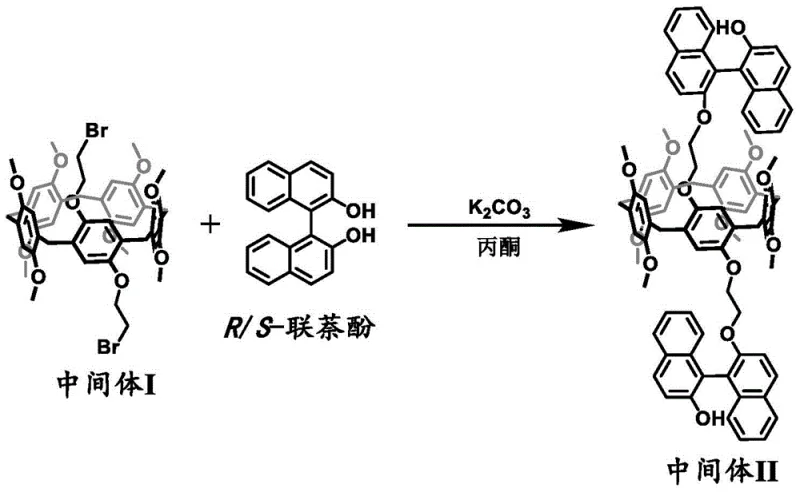
The success of this synthesis relies heavily on the precise control of stereochemistry during the functionalization of the pillararene core. In the third step of the synthesis, the reaction between the brominated pillararene intermediate and binaphthol is critical for establishing the axial chirality that cooperates with the macrocycle's planar chirality. Conducted in acetone with potassium carbonate as a base at temperatures between 60°C and 75°C, this nucleophilic substitution replaces the bromine atoms with bulky binaphthoxy groups. The steric bulk of the binaphthyl moiety acts as a conformational lock, preventing the pillararene ring from inverting. This mechanistic feature is essential because it ensures that once the enantiomers are separated via chiral HPLC, they remain configurationally stable under ambient conditions, a prerequisite for reliable device performance.
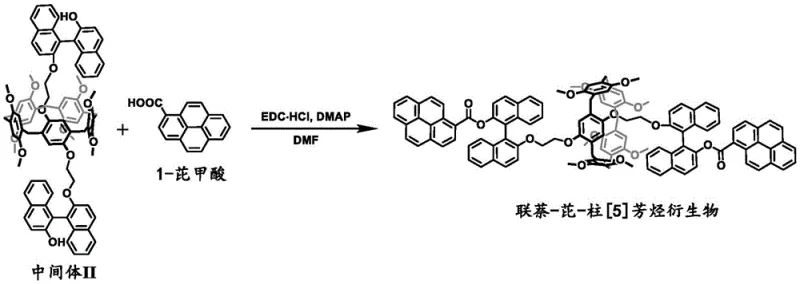
Following the establishment of the chiral framework, the final step involves the conjugation of the fluorescent pyrene unit. This is achieved through an amide-like coupling reaction, specifically an esterification between the hydroxyl groups of the binaphthol-modified intermediate and 1-pyrenecarboxylic acid. Using EDC·HCl and DMAP as coupling agents in DMF solvent at 40°C to 50°C, this step forms stable ester linkages. The choice of pyrene is deliberate; its large planar structure facilitates π-π stacking interactions which, according to the patent data, contribute to the formation of exciplexes that further enhance the circularly polarized emission. The synergy between the fixed chiral environment and the stacked pyrene units results in the observed red-shifted emission and high |glum| values, validating the design hypothesis.
How to Synthesize Binaphthyl-Pyrene-Pillararene Derivative Efficiently
The preparation of this advanced material follows a streamlined four-step protocol that balances yield optimization with operational safety. The initial bromination sets the stage for subsequent coupling reactions, while the macrocyclization requires careful control of stoichiometry to favor the cyclic pentamer over linear oligomers. The final two steps focus on the sequential installation of chirality and fluorescence, where reaction time and temperature are key variables; for instance, extending the reaction time in the binaphthol coupling step from 12 hours to 48 hours significantly improves conversion and reduces mono-substituted byproducts. For detailed laboratory instructions and specific workup procedures, please refer to the standardized guide below.
- Synthesize 1,4-bis(bromoethoxy)benzene via Appel reaction using carbon tetrabromide and triphenylphosphine in acetonitrile.
- Perform macrocyclization with 1,4-dimethoxybenzene and paraformaldehyde using boron trifluoride etherate catalyst to form the pillararene backbone.
- Introduce axial chirality by reacting the brominated intermediate with R/S-binaphthol under basic conditions in acetone.
- Conjugate the fluorescent pyrene unit via esterification with 1-pyrenecarboxylic acid using EDC/DMAP coupling agents.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthetic route described in this patent offers substantial advantages over legacy methods for producing chiral luminescent materials. The reliance on commodity chemicals such as 1,4-dimethoxybenzene, paraformaldehyde, and carbon tetrabromide ensures a stable and resilient raw material supply chain, mitigating risks associated with scarce or geopolitically sensitive reagents. Furthermore, the avoidance of precious metal catalysts like palladium or platinum in the key coupling steps eliminates the need for expensive metal scavenging processes, which are often a significant cost driver in pharmaceutical and electronic grade chemical manufacturing. This simplification of the downstream processing directly translates to cost reduction in optoelectronic material manufacturing, allowing for more competitive pricing structures in the final device market.
- Cost Reduction in Manufacturing: The process utilizes inexpensive Lewis acid catalysts (BF3·Et2O) and standard coupling reagents (EDC/DMAP) rather than rare earth metals or complex organocatalysts. This significantly lowers the direct material cost per kilogram. Additionally, the purification methods described, primarily involving silica gel chromatography and recrystallization, are standard unit operations in fine chemical plants, requiring no specialized equipment investment. The elimination of transition metals also reduces waste treatment costs associated with heavy metal disposal, contributing to a leaner cost structure.
- Enhanced Supply Chain Reliability: All starting materials, including R/S-binaphthol and pyrene derivatives, are commercially available in bulk quantities from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate moderate temperature fluctuations and do not require ultra-low temperatures or high-pressure vessels, ensures consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules for downstream OLED or sensor manufacturers.
- Scalability and Environmental Compliance: The synthesis operates in common organic solvents like acetonitrile, dichloromethane, and DMF, which have well-established recovery and recycling protocols in modern chemical facilities. The reaction temperatures are mild (ranging from 0°C to 75°C), reducing energy consumption compared to high-temperature pyrolysis or cryogenic processes. The solid-state nature of several intermediates facilitates easy isolation via filtration, minimizing solvent usage during workup. These factors collectively support a greener manufacturing profile that aligns with increasingly stringent environmental regulations in the chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral pillararene derivatives. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy for technical decision-makers evaluating this technology for integration into their product pipelines.
Q: What is the primary advantage of this binaphthyl-pyrene-pillararene derivative over traditional small molecule CPL emitters?
A: Unlike traditional small molecules which often suffer from low dissymmetry factors (|glum|), this derivative combines the planar chirality of the pillararene host with the axial chirality of binaphthol and the strong fluorescence of pyrene. This synergistic effect results in a significantly enhanced |glum| value reaching 5.6×10^-3, making it superior for 3D display and optical storage applications.
Q: How does the synthesis method address the stability issues of chiral pillararenes?
A: The patent describes modifying the pillararene side chains with bulky binaphthyl groups. These large steric hindrance groups effectively lock the conformation of the pillararene ring, preventing the rapid aromatic ring flipping that typically leads to racemization. This ensures the stability of the enantiomers required for consistent circularly polarized light emission.
Q: Is this process suitable for large-scale commercial production of optoelectronic intermediates?
A: Yes, the process utilizes mild reaction conditions ranging from ice bath to 75°C and employs commercially available solvents like acetonitrile, dichloromethane, and DMF. The absence of expensive transition metal catalysts and the use of standard purification techniques like silica gel chromatography make the route highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Binaphthyl-Pyrene-Pillararene Derivative Supplier
As the demand for high-performance circularly polarized luminescent materials grows in the 3D display and optical security sectors, having a manufacturing partner with deep expertise in supramolecular chemistry is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex architectures like the binaphthyl-pyrene-pillararene derivative can be delivered with consistent quality. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required for optoelectronic applications, minimizing defects in final devices and maximizing yield for our clients.
We invite R&D and procurement leaders to collaborate with us to optimize this synthesis for your specific volume requirements. By leveraging our process engineering capabilities, we can provide a Customized Cost-Saving Analysis tailored to your production targets. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to accelerate your development timeline for next-generation chiral optical materials.
