Advanced Biocatalytic Synthesis of (R)-HPBE: Scaling High-Purity Intermediates for Global ACE Inhibitor Production
Advanced Biocatalytic Synthesis of (R)-HPBE: Scaling High-Purity Intermediates for Global ACE Inhibitor Production
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and cost-effective synthesis routes for critical active pharmaceutical ingredient (API) intermediates has led to significant advancements in biocatalytic technologies. A pivotal development in this domain is documented in patent CN102994403A, which discloses a novel microbial strain, Rhodotorula mucilaginosa CCZU-G5, capable of highly stereoselective asymmetric reduction. This technology specifically targets the production of (R)-2-hydroxy-4-phenylbutyric acid ethyl ester ((R)-HPBE), a chiral building block essential for the synthesis of widely prescribed angiotensin-converting enzyme (ACE) inhibitors such as Lisinopril, Enalapril, and Benazepril. By leveraging whole-cell biocatalysis in a water/organic solvent biphasic system, this method addresses longstanding challenges regarding optical purity, molar conversion rates, and environmental impact, positioning itself as a superior alternative to traditional chemical synthesis and resolution techniques for reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of (R)-HPBE has relied heavily on chemical resolution or asymmetric chemical synthesis, both of which present substantial economic and technical bottlenecks for cost reduction in API manufacturing. Chemical resolution methods, which involve separating racemic mixtures into single enantiomers, suffer from an inherent theoretical maximum yield of only 50%, resulting in significant waste of the unwanted (S)-enantiomer and inefficient atom economy. Alternatively, chemical asymmetric synthesis often necessitates the use of expensive chiral ligands or transition metal catalysts, which not only drive up raw material costs but also introduce risks of heavy metal contamination that require rigorous and costly purification steps to meet stringent regulatory standards. Furthermore, these chemical processes frequently operate under harsh conditions involving extreme temperatures or pressures, leading to higher energy consumption and the generation of hazardous by-products that complicate waste management and environmental compliance.
The Novel Approach
In stark contrast, the biocatalytic route described in the patent utilizes the whole cells of Rhodotorula mucilaginosa CCZU-G5 to directly reduce ethyl 2-oxo-4-phenylbutyrate to the desired (R)-enantiomer with exceptional precision. This biological approach operates under mild physiological conditions, typically around 30°C and neutral pH, drastically reducing energy requirements and equipment stress. The use of a biphasic system comprising an organic solvent like isooctane and an aqueous buffer allows for high substrate loading concentrations, overcoming the solubility limitations often faced in purely aqueous enzymatic reactions. Most critically, this method achieves yields exceeding 98% with enantiomeric excess (e.e.) values greater than 99%, effectively eliminating the waste associated with resolution and ensuring the production of high-purity OLED material grade or pharma grade intermediates without the need for complex chiral chromatography.
Mechanistic Insights into Whole-Cell Asymmetric Biocatalysis
The core of this technological breakthrough lies in the sophisticated metabolic machinery of the Rhodotorula mucilaginosa CCZU-G5 strain, which functions as a self-contained micro-reactor. Unlike isolated enzyme systems that require the continuous addition of expensive cofactors like NAD(P)H, whole-cell biocatalysts possess intrinsic cofactor regeneration pathways. In this specific transformation, the microbial cells utilize endogenous dehydrogenases to catalyze the stereoselective reduction of the ketone group on the substrate. To sustain this redox cycle, a co-substrate such as glucose is added to the reaction medium; the cells metabolize the glucose to regenerate the reduced cofactor NAD(P)H from its oxidized form NAD(P)+, which is then immediately consumed in the reduction of the target ketone. This internal recycling mechanism ensures a continuous supply of reducing power without the need for external enzyme coupling systems, significantly simplifying the reaction setup and lowering operational complexity.
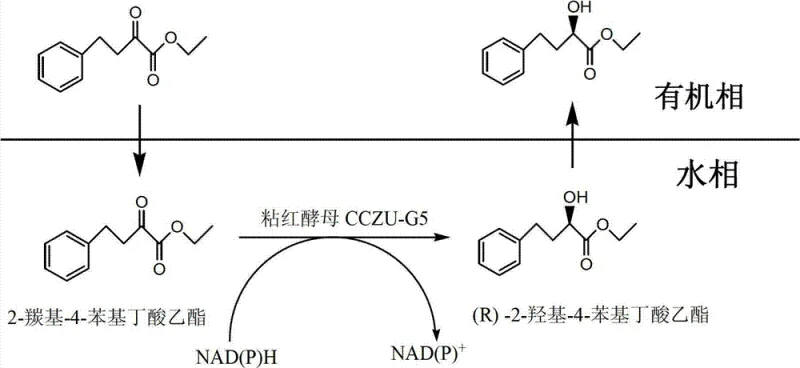
Furthermore, the implementation of a water/organic solvent biphasic system plays a crucial role in enhancing reaction kinetics and product recovery. The organic phase, optimized with solvents like isooctane, serves as a reservoir for the hydrophobic substrate and the resulting product, effectively increasing their local concentration near the biocatalyst interface while minimizing substrate inhibition effects that might occur in a single aqueous phase. Simultaneously, the aqueous phase maintains the viability and metabolic activity of the yeast cells. This dual-phase architecture not only drives the reaction equilibrium towards product formation by extracting the product into the organic layer but also facilitates a straightforward downstream processing workflow. Once the reaction reaches completion, the organic phase containing the high-purity product can be physically separated from the aqueous cell mass, dried, and concentrated, yielding the final (R)-HPBE with minimal impurity profiles and no detectable by-products.
How to Synthesize (R)-2-Hydroxy-4-phenylbutyric Acid Ethyl Ester Efficiently
Implementing this biocatalytic process requires precise control over fermentation parameters and reaction conditions to maximize the catalytic efficiency of the CCZU-G5 strain. The protocol begins with the activation and fermentation of the yeast to generate sufficient wet biomass, followed by the formulation of the biphasic reaction mixture with optimized ratios of organic solvent to buffer. The detailed standardized synthesis steps, including specific media compositions, incubation times, and work-up procedures necessary to achieve the reported >99% e.e. and high yields, are outlined below for technical reference.
- Cultivate Rhodotorula mucilaginosa CCZU-G5 in a fermentation medium containing glucose and peptone at 30°C to obtain wet cells.
- Prepare a biphasic reaction system consisting of isooctane and phosphate buffer (pH 7.5), adding the substrate ethyl 2-oxo-4-phenylbutyrate and glucose as a co-substrate.
- Introduce the wet cells to the system and react at 30°C with shaking, followed by separation of the organic phase and purification to isolate the high-purity (R)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this whole-cell biocatalytic technology translates into tangible strategic benefits that extend beyond mere technical performance. The shift from chemical resolution to biosynthesis fundamentally alters the cost structure of producing chiral intermediates by eliminating the loss of half the starting material, effectively doubling the theoretical output from the same amount of raw substrate. This improvement in atom economy directly correlates to substantial cost savings in raw material procurement, allowing manufacturers to offer more competitive pricing for downstream API producers. Additionally, the mild reaction conditions reduce the dependency on specialized high-pressure reactors and extensive safety infrastructure, lowering capital expenditure (CAPEX) requirements for facility upgrades and enabling more flexible manufacturing scheduling.
- Cost Reduction in Manufacturing: The elimination of expensive chiral chemical catalysts and the removal of external cofactor supplementation significantly lower the variable costs associated with each production batch. Since the microbial cells regenerate their own cofactors using inexpensive carbon sources like glucose, the process avoids the recurring expense of purchasing noble metal catalysts or synthetic chiral ligands. Furthermore, the simplified downstream processing, which relies on phase separation rather than complex crystallization or chromatographic purification, reduces solvent consumption and labor hours, contributing to a leaner and more cost-efficient production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Utilizing a robust microbial strain that can be stored and propagated ensures a consistent and renewable source of catalyst, mitigating the supply risks often associated with sourcing specialized chemical reagents from limited vendors. The fermentation process for generating the biocatalyst is scalable and can be performed in-house or sourced from reliable contract manufacturers, providing greater control over the supply chain continuity. This self-sufficiency reduces lead times for high-purity pharmaceutical intermediates and insulates the production schedule from fluctuations in the global market availability of rare earth metals or complex organic ligands.
- Scalability and Environmental Compliance: The process is inherently green, operating at ambient temperatures and generating minimal hazardous waste, which aligns with increasingly strict global environmental regulations and corporate sustainability goals. The absence of heavy metals and toxic solvents simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing site. From a scalability perspective, the biphasic system is well-suited for large-scale stirred tank reactors, allowing for seamless transition from pilot scale to commercial production volumes of 100 MT or more without significant re-engineering of the process parameters, ensuring stable supply for long-term commercial agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic route for (R)-HPBE production. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on process robustness and quality assurance.
Q: What are the primary advantages of using whole-cell biocatalysis over chemical resolution for (R)-HPBE?
A: Whole-cell biocatalysis eliminates the theoretical 50% yield limit inherent in racemic resolution methods. Furthermore, it utilizes internal cofactor regeneration systems within the microbial cells, removing the need for expensive external coenzymes often required in isolated enzyme processes, thereby significantly reducing raw material costs.
Q: How does the biphasic solvent system improve the reaction efficiency?
A: The water/organic solvent biphasic system, specifically using isooctane, enhances the solubility of the hydrophobic substrate and product while protecting the microbial cells from organic solvent toxicity. This setup shifts the reaction equilibrium towards product formation and simplifies downstream processing by allowing direct extraction of the product into the organic phase.
Q: Is this process scalable for industrial manufacturing of ACE inhibitor intermediates?
A: Yes, the process operates under mild conditions (30°C, atmospheric pressure) and uses robust, self-regenerating biocatalysts that can be produced via standard fermentation. The simplicity of the work-up procedure, involving phase separation and evaporation, makes it highly amenable to large-scale commercial production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-Hydroxy-4-phenylbutyric Acid Ethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality chiral intermediates for the global cardiovascular drug market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of (R)-HPBE meets the exacting standards required for GMP-compliant API synthesis. Our expertise in biocatalysis allows us to optimize fermentation and conversion parameters to deliver consistent quality and yield, supporting our partners in accelerating their drug development timelines.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this advanced biocatalytic technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the most efficient and sustainable manufacturing solutions available in the industry.
