Scalable Palladium-Catalyzed Synthesis of Alpha-Aryl-Difluoromethyl Compounds for Global Supply Chains
The introduction of fluorine atoms into organic molecules, particularly the difluoromethyl group, has become a cornerstone strategy in modern medicinal chemistry and agrochemical development due to its profound impact on metabolic stability and lipophilicity. Patent CN108341739B discloses a groundbreaking preparation method for alpha-aryl, heteroaryl, or alkenyl-alpha,alpha-difluoromethyl compounds that addresses critical limitations in existing synthetic routes. This technology leverages a palladium-catalyzed coupling reaction between readily available boronic acid derivatives and chlorodifluoromethane, operating under relatively mild thermal conditions ranging from 40°C to 140°C. For R&D directors and procurement specialists, this represents a significant shift away from hazardous fluorinating agents towards a more sustainable and economically viable manufacturing paradigm. The ability to perform late-stage fluorination on complex bioactive molecules without compromising sensitive functional groups opens new avenues for drug discovery and process optimization, positioning this methodology as a vital tool for the reliable pharmaceutical intermediate supplier seeking to enhance their portfolio with high-value fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-aryl or heteroaryl-alpha,alpha-difluoroalkyl blocks has relied heavily on deoxofluorination reagents such as DAST (Diethylaminosulfur Trifluoride) or Deoxofluor. While effective in certain contexts, these conventional methods suffer from severe drawbacks that hinder their application in large-scale commercial production. The primary concern is the inherent toxicity and instability of these fluorinating agents, which pose significant safety risks to personnel and require specialized containment infrastructure, thereby drastically increasing operational costs. Furthermore, these reactions often exhibit poor functional group compatibility, necessitating extensive protection and deprotection steps that lengthen the synthetic timeline and reduce overall atom economy. The harsh reaction conditions associated with traditional fluorination can also lead to decomposition of complex drug-like molecules, limiting their utility in late-stage functionalization strategies. Consequently, the industry has long sought a safer, more robust alternative that can deliver high-purity difluoromethyl compounds without the logistical and safety burdens of legacy technologies.
The Novel Approach
The novel approach detailed in the patent data utilizes a transition metal-catalyzed cross-coupling strategy that fundamentally reshapes the economic and safety profile of difluoromethylation. By employing chlorodifluoromethane (ClCF2H), an inexpensive and industrially abundant gas, as the fluorine source, the method eliminates the need for costly and dangerous deoxofluorination reagents. The reaction proceeds in the presence of a palladium salt, a base, and an aryl phenol compound, facilitating the coupling of formula A or D compounds with formula B under controlled temperatures. This catalytic system demonstrates exceptional substrate scope, accommodating a wide array of aryl, heteroaryl, and alkenyl boronic acids while maintaining excellent tolerance for diverse functional groups. The operational simplicity, combined with the use of cheap and easily obtainable raw materials, translates directly into substantial cost savings and a reduced environmental footprint, making it an ideal candidate for cost reduction in API manufacturing and the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
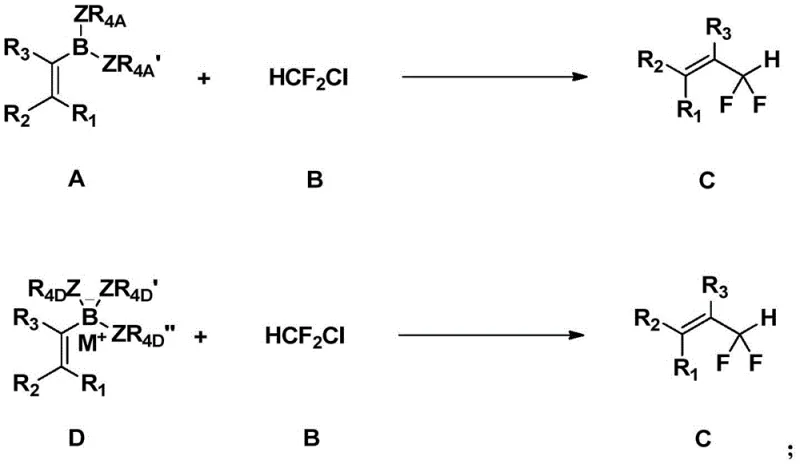
Mechanistic Insights into Pd-Catalyzed Difluoromethylation
The mechanistic underpinning of this transformation involves a sophisticated palladium catalytic cycle that ensures high efficiency and selectivity. The process initiates with the oxidative addition of the palladium catalyst to the carbon-chlorine bond of chlorodifluoromethane, generating a key difluoromethyl-palladium intermediate. This species then undergoes transmetallation with the aryl boronic acid or ester, facilitated by the base and the aryl phenol cocatalyst, which likely assists in activating the boron species and stabilizing the transition state. The presence of bulky phosphine ligands, such as Xantphos, plays a crucial role in promoting the reductive elimination step, which releases the desired alpha-aryl-difluoromethyl product and regenerates the active palladium(0) catalyst. This carefully balanced cycle minimizes side reactions such as beta-fluorine elimination, which is a common pitfall in difluoromethyl metal chemistry. For technical teams, understanding this mechanism highlights the importance of ligand selection and base optimization in achieving maximum yield and purity, ensuring that the process remains robust even when scaling from milligram to multi-kilogram quantities.
Impurity control is another critical aspect where this methodology excels, driven by the mild reaction conditions and the specific choice of additives. The inclusion of hydroquinone as an additive serves to suppress the formation of oxidative byproducts and stabilizes the catalytic system against degradation over prolonged reaction times. The use of mild bases like potassium carbonate, rather than strong alkoxides, prevents the hydrolysis of sensitive ester or amide functionalities often present in advanced intermediates. Furthermore, the reaction's tolerance to moisture and air, relative to other organometallic processes, simplifies the workup procedure and reduces the risk of generating difficult-to-remove inorganic salts. This high level of chemoselectivity ensures that the final product meets stringent purity specifications required for regulatory submission, reducing the burden on downstream purification processes and enhancing the overall reliability of the supply chain for high-purity pharmaceutical intermediates.
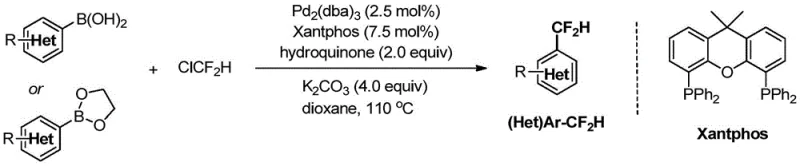
How to Synthesize Alpha-Aryl-Difluoromethyl Compounds Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and safety. The general procedure involves charging a reactor with the aryl boronic acid substrate, palladium catalyst, ligand, base, and hydroquinone under an inert atmosphere. Solvent selection is critical, with 1,4-dioxane or toluene preferred for their ability to dissolve both organic substrates and inorganic bases while maintaining stability at elevated temperatures. The chlorodifluoromethane reagent is introduced either as a gas or a solution, and the mixture is heated to between 80°C and 110°C for a period of 24 to 48 hours. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining aryl boronic acid or ester, palladium catalyst (e.g., Pd2(dba)3), ligand (e.g., Xantphos), base (e.g., K2CO3), and hydroquinone in a solvent like dioxane.
- Introduce chlorodifluoromethane (ClCF2H) gas or solution into the reactor under anhydrous and oxygen-free conditions, ensuring proper pressure control.
- Heat the mixture to 80-110°C for 24-48 hours, then purify the resulting alpha-aryl-difluoromethyl product via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The substitution of expensive, specialized fluorinating reagents with commodity-grade chlorodifluoromethane results in a drastic reduction in raw material costs, which is a primary driver for margin improvement in competitive markets. Additionally, the mild reaction conditions reduce energy consumption and eliminate the need for cryogenic cooling or high-pressure reactors, further lowering the capital expenditure required for implementation. The robustness of the catalytic system ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or extensive rework. This reliability is essential for maintaining continuous supply lines to downstream customers who depend on just-in-time delivery models for their own manufacturing schedules.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the use of low-cost starting materials and the elimination of hazardous waste disposal fees associated with traditional fluorination. By avoiding toxic reagents like DAST, manufacturers save significantly on safety compliance, personal protective equipment, and waste treatment protocols. The high atom economy of the coupling reaction means less raw material is wasted, and the simplified purification process reduces solvent usage and processing time. These factors combine to create a leaner, more cost-effective production model that enhances competitiveness without sacrificing product quality or performance standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the key reagents, specifically aryl boronic acids and chlorodifluoromethane, which are produced globally by multiple suppliers. This diversification of the supply base mitigates the risk of shortages that can occur with proprietary or niche reagents. Furthermore, the stability of the boronic acid starting materials allows for long-term storage and inventory management, enabling manufacturers to buffer against market fluctuations. The scalability of the process ensures that production volumes can be ramped up quickly to meet surges in demand, providing a secure and dependable source of critical intermediates for global pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: The environmental profile of this method aligns with increasingly strict global regulations regarding chemical manufacturing and emissions. The absence of heavy metal waste streams and toxic fluorinating byproducts simplifies the permitting process and reduces the environmental liability of the production facility. The reaction's compatibility with standard stainless steel reactors facilitates easy scale-up from pilot plant to commercial production without the need for exotic materials of construction. This ease of scale-up, combined with the reduced environmental footprint, positions manufacturers to meet corporate sustainability goals while maintaining operational efficiency and regulatory compliance across international jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoromethylation technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on substrate scope, reaction conditions, and scalability. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing workflows and product pipelines.
Q: What are the advantages of this Pd-catalyzed method over traditional fluorination?
A: Unlike traditional methods using toxic reagents like DAST, this protocol utilizes cheap chlorodifluoromethane and mild conditions, offering better functional group compatibility and safety.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent demonstrates gram-scale synthesis with high yields, using commercially available raw materials and standard equipment, indicating strong scalability potential.
Q: What types of substrates are compatible with this reaction?
A: The method supports a wide range of substrates including aryl, heteroaryl, and alkenyl boronic acids, tolerating various functional groups like esters, ketones, and halides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Aryl-Difluoromethyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced fluorination technologies in driving the next generation of therapeutic and agricultural products. Our team of expert chemists has extensively evaluated the methodology described in CN108341739B and possesses the technical capability to adapt and optimize this route for diverse client needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of difluoromethyl intermediate meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this innovative chemistry for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and target specifications. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in palladium-catalyzed coupling can accelerate your development timelines and reduce your overall cost of goods sold. Together, we can build a more resilient and efficient supply chain for the future of fine chemicals.
