Advanced Synthetic Route for 3-Bromo-2-aminothiophene: Scalable Manufacturing for Pharma Intermediates
Advanced Synthetic Route for 3-Bromo-2-aminothiophene: Scalable Manufacturing for Pharma Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing heterocyclic scaffolds, particularly thiophene derivatives which serve as critical building blocks for bioactive molecules. Patent CN107880015B, published in May 2020, introduces a groundbreaking synthetic strategy for the preparation of 3-bromo-2-aminothiophene, a pivotal intermediate in the synthesis of various anti-inflammatory, analgesic, and anti-tumor agents. This innovation departs from traditional, cumbersome pathways by utilizing a direct lithiation-transmetallation sequence followed by a mild copper-catalyzed amination. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in process chemistry, offering a route that not only simplifies the operational workflow but also drastically enhances the economic viability of producing high-purity pharmaceutical intermediates. The method leverages readily available starting materials like 3-bromothiophene and employs standard reagents such as lithium diisopropylamide or lithium hexamethyldisilazide, ensuring that the supply chain remains resilient and cost-efficient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of amino-thiophene derivatives often relied on complex multi-step sequences that introduced significant safety hazards and cost burdens. Specifically, literature references such as U.S. Pat. No. 20110152243 and reports in Bioorganic & Medicinal Chemistry Letters describe routes involving the Curtius rearrangement of 3-bromothiophene carboxylic acid. This conventional approach necessitates the use of diphenyl azidophosphate, a potentially explosive and hazardous reagent that requires stringent safety protocols and specialized handling equipment, thereby inflating operational costs. Furthermore, these traditional methods typically involve the installation and subsequent removal of protecting groups, such as the tert-butoxycarbonyl (Boc) group, to manage the reactivity of the amine functionality. These additional synthetic steps not only reduce the overall atom economy but also extend the production timeline, leading to lower throughput and higher waste generation. The reliance on such indirect pathways creates bottlenecks in commercial scale-up of complex pharmaceutical intermediates, making it difficult for manufacturers to meet the growing demand for thiophene-based active pharmaceutical ingredients without compromising on margin or safety standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN107880015B offers a streamlined, direct avenue to 3-bromo-2-aminothiophene that circumvents the pitfalls of the Curtius rearrangement. By employing a strategic lithiation of 3-bromothiophene followed by trapping with a boron or silicon electrophile, the process generates a reactive intermediate that can be directly converted to the amine using aqueous ammonia under cuprous oxide catalysis. This approach eliminates the need for hazardous azides and avoids the economic drain of protection-deprotection cycles. The reaction conditions are remarkably mild, often proceeding at room temperature in the final amination step, which significantly reduces energy consumption compared to high-temperature rearrangements. Moreover, the use of commodity chemicals like ammonia water and simple ether solvents ensures that the cost reduction in fine chemical manufacturing is substantial, as the raw material inventory is both inexpensive and globally accessible. This novel route transforms a previously challenging synthesis into a robust, two-step operation that is ideally suited for industrial amplification.
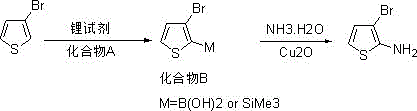
Mechanistic Insights into Lithiation-Transmetallation-Amination Cascade
The core of this synthetic breakthrough lies in the precise control of regioselectivity during the initial deprotonation phase. When 3-bromothiophene is treated with a strong non-nucleophilic base such as lithium diisopropylamide (LDA) or lithium hexamethyldisilazide (LiHMDS) in an ethereal solvent like tetrahydrofuran or 2-methyltetrahydrofuran, the proton at the C2 position is selectively abstracted. This selectivity is driven by the coordination of the lithium cation with the sulfur atom and the kinetic acidity of the C2-H bond, which is enhanced by the adjacent bromine substituent. The reaction temperature is critically maintained between -80°C and 0°C during this deprotonation stage to prevent side reactions such as halogen-metal exchange or polymerization, ensuring the formation of a clean 2-lithio-3-bromothiophene species. Subsequent addition of electrophiles like trimethyl borate or trimethylchlorosilane allows for the efficient trapping of this organolithium intermediate, generating stable organoboron or organosilicon compounds that serve as precursors for the final transformation.
The final conversion to the amine is mediated by a copper-catalyzed coupling mechanism that is both elegant and efficient. In the presence of cuprous oxide (Cu2O) and aqueous ammonia, the organoboron or organosilicon intermediate undergoes a transmetallation to form a copper-amido species, which then reductively eliminates to yield the desired 3-bromo-2-aminothiophene. A key mechanistic insight provided by the patent is the sensitivity of this step to temperature; maintaining the reaction at room temperature is crucial because elevating the temperature decreases the solubility of ammonia in the organic solvent, thereby suppressing the reaction rate and yield. This understanding allows for tight impurity control, as the mild conditions prevent the degradation of the sensitive thiophene ring or the formation of poly-aminated byproducts. The result is a product with high purity, often exceeding 98% as demonstrated in the experimental examples, which minimizes the need for extensive downstream purification and aligns perfectly with the rigorous quality standards required for reliable pharmaceutical intermediate supplier operations.
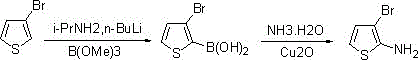
How to Synthesize 3-Bromo-2-aminothiophene Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to strict temperature controls and stoichiometric ratios as outlined in the patent embodiments. The process begins with the generation of the lithium amide base in situ or the use of a commercial solution, followed by the careful addition of 3-bromothiophene at cryogenic temperatures to ensure exclusive C2-lithiation. Once the lithiated species is formed, it is quenched with the appropriate boron or silicon source, and the resulting intermediate is isolated or telescoped directly into the amination reactor. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are provided in the structured guide below to ensure reproducibility and safety during scale-up.
- Deprotonate 3-bromothiophene using a lithium reagent such as LDA or LiHMDS in an ether solvent at low temperatures ranging from -80°C to 0°C.
- React the lithiated intermediate with a boron ester (e.g., trimethyl borate) or chlorosilane to generate the corresponding organoboron or organosilicon compound.
- Perform a copper-catalyzed amination reaction using aqueous ammonia and cuprous oxide in an organic solvent like methanol or acetonitrile at room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lithiation-based methodology offers transformative benefits that extend far beyond simple yield improvements. By shifting away from the Curtius rearrangement, manufacturers can eliminate the procurement of high-risk azide reagents and expensive protecting group chemicals, leading to a simplified supply chain that is less vulnerable to regulatory scrutiny and vendor shortages. The use of commodity feedstocks like 3-bromothiophene and ammonia water ensures a stable and continuous supply of raw materials, reducing lead time for high-purity pharmaceutical intermediates and allowing for more agile response to market demands. Furthermore, the operational simplicity of the process, which avoids extreme temperatures and high-pressure conditions in the final step, translates to lower capital expenditure on specialized reactor equipment and reduced energy costs, driving significant overall cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The elimination of the Boc protection and deprotection steps inherently reduces the number of unit operations, which directly lowers labor, solvent, and utility costs associated with each batch. Additionally, the replacement of expensive palladium catalysts or hazardous azides with inexpensive cuprous oxide and ammonia water results in a drastic reduction in raw material expenses. The high atom economy of the direct amination means less waste is generated per kilogram of product, further decreasing waste disposal fees and enhancing the overall profit margin for the final active ingredient.
- Enhanced Supply Chain Reliability: Since the key reagents such as LDA, LiHMDS, and trimethyl borate are widely produced bulk chemicals, the risk of supply disruption is minimized compared to relying on niche, custom-synthesized intermediates required by older methods. The robustness of the reaction conditions, particularly the tolerance for room temperature amination, ensures that production schedules are not delayed by complex heating or cooling requirements, facilitating just-in-time manufacturing strategies. This reliability is critical for maintaining the continuity of supply for downstream drug manufacturers who depend on consistent availability of key thiophene scaffolds.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial amplification, utilizing solvents like methanol and acetonitrile which are easily recovered and recycled in standard distillation units. The avoidance of heavy metal catalysts like palladium simplifies the purification process and ensures that the final product meets stringent residual metal specifications without the need for costly scavenging resins. From an environmental perspective, the reduction in hazardous waste and the use of safer reagents align with green chemistry principles, helping companies meet increasingly strict environmental regulations and sustainability goals without compromising on production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is essential for process chemists and technical buyers evaluating the feasibility of adopting this technology for their own production lines. The answers reflect the specific advantages and operational parameters defined in CN107880015B, providing a clear roadmap for successful technology transfer.
Q: What are the key advantages of this lithiation method over the traditional Curtius rearrangement?
A: The lithiation method avoids the use of hazardous azide reagents and expensive protecting groups like Boc, offering a safer, more direct, and cost-effective pathway with high atom economy.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states the process uses easily available raw materials and simple operations, making it highly suitable for amplification and industrial-scale manufacturing.
Q: What represents the critical quality attribute in the final amination step?
A: Maintaining room temperature during the cuprous oxide catalyzed reaction is critical, as increasing the temperature reduces ammonia solubility and negatively impacts the reaction yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-2-aminothiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic routes play in the competitiveness of the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN107880015B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative method to life. We are committed to delivering high-purity 3-bromo-2-aminothiophene that meets stringent purity specifications, utilizing our rigorous QC labs to ensure every batch conforms to the highest international standards. Our state-of-the-art facilities are equipped to handle the low-temperature lithiation steps safely and efficiently, guaranteeing a consistent supply of this valuable intermediate for your drug development programs.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive value and efficiency in your production of thiophene-based therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
