Scalable Synthesis of 1,3,4,6-Tetraacetyl-L-Gulose for High-Value Antibiotic Intermediates
The pharmaceutical industry's relentless pursuit of potent antitumor agents has placed significant focus on the efficient production of Bleomycin A2, a glycopeptide antibiotic with remarkable DNA cleavage capabilities. Central to the molecular architecture of Bleomycin A2 is the disaccharide unit containing L-gulose, a rare sugar that has historically posed significant synthetic challenges due to its scarcity and complex stereochemistry. Patent CN103848874A introduces a transformative methodology for synthesizing 1,3,4,6-tetraacetyl-L-gulose, a critical intermediate, by leveraging glucuronic acid lactone as a readily available starting material. This innovation represents a paradigm shift from relying on expensive, naturally occurring L-sugars to utilizing abundant D-series precursors through a clever inversion and functionalization strategy. By circumventing the need for rare chiral pool materials, this process not only democratizes access to this key building block but also establishes a robust foundation for the cost reduction in API manufacturing associated with complex oncology drugs. The technical elegance of this route lies in its balance of rigorous stereocontrol and operational simplicity, addressing the dual needs of R&D precision and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-gulose derivatives has been plagued by the inherent limitations of starting material availability and the complexity of stereochemical manipulation. Traditional routes often relied on D-mannose or D-glucose, requiring extensive oxidation state exchanges at the C1 and C6 positions, which introduced multiple low-yielding steps and difficult purification challenges. Other methods utilized L-xylose or enzymatic processes, which, while effective on a small scale, suffered from prohibitive costs when scaled to industrial levels due to the expense of the starting sugars or the fragility of biocatalysts. Furthermore, earlier synthetic pathways frequently employed over-protection strategies that necessitated harsh deprotection conditions, leading to product degradation and inconsistent impurity profiles. These conventional approaches created a bottleneck in the supply chain for Bleomycin intermediates, resulting in long lead times and volatile pricing structures that hindered the broader development of glycopeptide-based therapeutics. The reliance on scarce natural resources meant that supply continuity was always at risk, making long-term project planning difficult for procurement teams managing complex antibiotic portfolios.
The Novel Approach
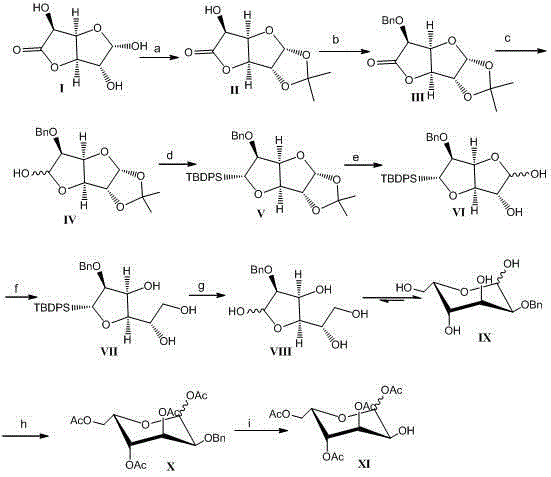
The methodology disclosed in the patent data offers a streamlined, nine-step sequence that fundamentally alters the economic landscape of L-gulose production. By initiating the synthesis with glucuronic acid lactone, a commodity chemical, the process bypasses the scarcity issues associated with L-sugars entirely. The route employs a strategic sequence of acetonylidene protection, benzyl protection, and selective lactone reduction to establish the necessary carbon framework with high fidelity. A key innovation is the use of DIBAL-H for the controlled reduction of the lactone to a hemiacetal at low temperatures, followed by robust silyl protection with TBDPS to lock in the stereochemistry. This approach minimizes the number of isolation steps and utilizes reagents that are standard in fine chemical manufacturing, such as sodium borohydride and acetic anhydride. The final stages involve a clean deprotection and acetylation sequence, culminating in a catalytic hydrogenation that delivers the target 1,3,4,6-tetraacetyl-L-gulose with a favorable beta-to-alpha anomeric ratio. This logical progression from cheap feedstock to high-value intermediate exemplifies how modern organic synthesis can drive cost reduction in pharmaceutical intermediate manufacturing through intelligent route design rather than brute-force optimization.
Mechanistic Insights into Selective Lactone Reduction and Protection
The core mechanistic brilliance of this synthesis resides in the precise management of oxidation states and protecting group orthogonality. The initial reduction of the benzyl-protected lactone using diisobutylaluminum hydride (DIBAL-H) at temperatures between -40°C and -10°C is critical; this step selectively reduces the carbonyl to a hemiacetal without over-reducing to the diol, preserving the functionality needed for subsequent ring closure. The introduction of the tert-butyldiphenylsilyl (TBDPS) group serves a dual purpose: it protects the newly formed hemiacetal hydroxyl from unwanted side reactions and provides steric bulk that influences the conformational preference of the sugar ring during later transformations. Following the removal of the acetonylidene group under acidic conditions, a second reduction with sodium borohydride converts the remaining hemiacetal mixture into a stable polyol structure. This step is performed at low temperatures (-40°C to 0°C) to maintain stereochemical integrity and prevent epimerization. The final cyclization to the pyranose form occurs spontaneously upon acid-mediated removal of the silyl group, driven by the thermodynamic stability of the six-membered ring. This cascade of mechanistic events ensures that the final acetylation step proceeds on a well-defined scaffold, minimizing the formation of regioisomers and simplifying the purification of the final tetraacetyl product.
Impurity control is inherently built into the choice of reagents and the sequence of operations. The use of silver oxide for benzylation, while traditional, is optimized here to proceed in solvents like ethyl acetate or DMF, allowing for easy filtration of silver salts and minimizing heavy metal contamination in the final product. The patent specifies that the total yield can reach 25% to 35%, which is substantial for a nine-step synthesis of a complex carbohydrate derivative. By avoiding transition metal catalysts in the early stages and reserving palladium-catalyzed hydrogenation solely for the final debenzylation, the process limits the potential for metal-induced degradation of the sensitive sugar backbone. The crystallization and chromatography protocols described, such as using ethyl acetate and petroleum ether mixtures, are designed to remove specific byproducts like TBDPS-OH, which shares similar polarity with the intermediate. This attention to detail in impurity profiling ensures that the resulting 1,3,4,6-tetraacetyl-L-gulose meets the stringent purity specifications required for coupling with the peptide moiety of Bleomycin, thereby reducing the risk of downstream failures in drug substance manufacturing.
How to Synthesize 1,3,4,6-Tetraacetyl-L-Gulose Efficiently
The execution of this synthetic route requires careful attention to temperature control and stoichiometry, particularly during the reduction and protection phases. The process begins with the dissolution of glucuronic acid lactone in acetone, followed by the dropwise addition of sulfuric acid to effect acetonylidene protection, a reaction that must be quenched carefully with sodium bicarbonate to prevent acid-catalyzed hydrolysis. Subsequent benzylation utilizes freshly prepared silver oxide and benzyl bromide, where the molar ratios are tightly controlled between 0.6:1 and 1.5:1 to maximize conversion while minimizing waste. The critical DIBAL-H reduction step demands strict adherence to the -40°C to -10°C temperature window to ensure the formation of the hemiacetal intermediate without over-reduction. For operators seeking a standardized protocol to implement this technology in a pilot or production setting, the detailed operational parameters regarding solvent choices, reaction times, and workup procedures are essential for reproducibility. The following guide outlines the critical operational milestones derived from the patent examples to assist technical teams in replicating this high-yield pathway.
- Protect glucuronic acid lactone with acetonylidene and benzyl groups, followed by selective lactone reduction using DIBAL-H at low temperatures.
- Protect the resulting hemiacetal with TBDPS, remove the acetonylidene group, and perform a second reduction with NaBH4 to establish the alcohol framework.
- Deprotect the silyl group to allow cyclization into the pyranose form, followed by per-acetylation and final catalytic hydrogenolysis to yield the target tetraacetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages beyond mere technical feasibility. The primary driver of value is the substitution of scarce, expensive L-sugar starting materials with glucuronic acid lactone, a commodity chemical available in bulk quantities from multiple global suppliers. This shift fundamentally decouples the production of Bleomycin intermediates from the volatility of the rare sugar market, ensuring a more stable and predictable supply chain. By eliminating the need for specialized enzymatic processes or multi-step oxidations of common sugars, the manufacturing process becomes significantly more linear and easier to scale. The operational simplicity translates directly into reduced manufacturing overhead, as the process utilizes standard reactor equipment and does not require specialized containment for hazardous or exotic reagents. Furthermore, the high total yield reported in the patent data implies a more efficient use of raw materials, reducing the overall material cost per kilogram of the final intermediate. This efficiency is crucial for maintaining margin integrity in the competitive oncology drug market, where cost pressures are intensifying.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials and the use of cost-effective reagents like sodium borohydride and acetic anhydride drastically lower the bill of materials. By avoiding complex enzymatic steps or rare natural products, the process removes significant cost drivers associated with sourcing and quality control of exotic inputs. The streamlined nature of the synthesis reduces the number of unit operations, which in turn lowers labor costs and energy consumption per batch. Additionally, the ability to recycle solvents such as ethyl acetate and methanol further contributes to the overall economic efficiency of the process. These factors combine to create a manufacturing profile that is highly competitive, allowing for substantial cost savings that can be passed down the supply chain or reinvested in R&D.
- Enhanced Supply Chain Reliability: Relying on glucuronic acid lactone as the feedstock ensures a robust supply base, as this chemical is produced on a large scale for various industrial applications. This abundance mitigates the risk of supply disruptions that often plague niche carbohydrate markets. The synthetic route's tolerance for standard industrial conditions means that production can be easily transferred between different manufacturing sites without significant re-validation, enhancing supply continuity. Moreover, the stability of the intermediates, particularly the protected species, allows for flexible scheduling and inventory management, reducing the pressure on just-in-time delivery models. This resilience is vital for pharmaceutical companies aiming to secure long-term supplies of critical antibiotic intermediates against a backdrop of global logistical uncertainties.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reactions that are exothermic but manageable within standard jacketed reactors. The avoidance of heavy metal catalysts in the early stages simplifies waste treatment and reduces the environmental footprint of the manufacturing process. The final hydrogenation step uses supported palladium catalysts which can be filtered and recovered, aligning with green chemistry principles and regulatory expectations for metal residues in pharmaceuticals. The use of common organic solvents facilitates compliance with environmental regulations regarding VOC emissions and waste disposal. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the supply chain, a factor increasingly important to stakeholders and end-users in the healthcare sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3,4,6-tetraacetyl-L-gulose. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your existing drug development pipelines. The responses cover aspects ranging from raw material sourcing to the specific stereochemical outcomes of the synthesis, providing a comprehensive overview for decision-makers.
Q: What is the primary advantage of using glucuronic acid lactone over traditional L-sugar starting materials?
A: Glucuronic acid lactone is significantly more abundant and cost-effective compared to rare L-type monosaccharides. This route avoids the high costs associated with sourcing scarce natural L-sugars or performing complex enzymatic conversions, thereby streamlining the supply chain for large-scale antibiotic production.
Q: How does this synthesis method ensure high stereochemical purity for Bleomycin intermediates?
A: The process utilizes precise protective group strategies, specifically the use of TBDPS and acetonylidene groups, to control stereochemistry during reduction steps. The final catalytic hydrogenation step yields a beta-to-alpha configuration ratio greater than 92:8, ensuring the high purity required for downstream glycosylation in Bleomycin A2 synthesis.
Q: Is this synthetic route suitable for industrial scale-up beyond gram quantities?
A: Yes, the patent explicitly highlights the method's suitability for industrial production. The reactions utilize common solvents like ethyl acetate and methanol, avoid exotic catalysts, and operate under controllable temperature conditions (-40°C to room temperature), making the transition from laboratory to multi-kilogram manufacturing feasible and robust.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4,6-Tetraacetyl-L-Gulose Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antibiotics like Bleomycin depends on the consistent availability of high-quality sugar intermediates. Our technical team has thoroughly analyzed the synthetic pathway described in CN103848874A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to life. We understand that the transition from laboratory scale to industrial manufacturing involves nuanced challenges in heat transfer, mixing, and purification that only experienced CDMO partners can navigate effectively. Our facilities are equipped with the rigorous QC labs and analytical capabilities necessary to verify the stringent purity specifications demanded by global regulatory bodies. We are committed to delivering 1,3,4,6-tetraacetyl-L-gulose that not only meets but exceeds the structural and impurity profiles required for downstream glycosylation, ensuring the integrity of your final drug product.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can enhance your supply chain resilience and cost structure. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to request specific COA data and route feasibility assessments to validate our capabilities against your internal benchmarks. Let us collaborate to secure a sustainable and economical supply of this critical pharmaceutical intermediate, driving your oncology projects forward with confidence and reliability.
