Scalable Dual-Enzyme Synthesis of Benzoquinone Intermediates for Oncology Drug Manufacturing
Scalable Dual-Enzyme Synthesis of Benzoquinone Intermediates for Oncology Drug Manufacturing
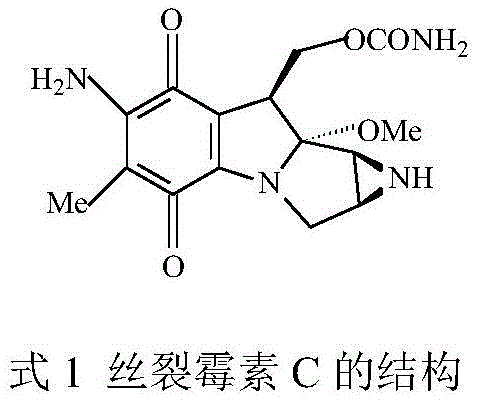
The pharmaceutical industry's relentless pursuit of safer, more effective oncology treatments has placed significant emphasis on the efficient synthesis of Mitomycin C analogs, particularly those featuring stable benzoquinone mother nuclei. A pivotal advancement in this domain is detailed in patent CN107653273B, which discloses a novel dual-enzyme one-pot method for synthesizing 2,3-dimethyl-5-alkylamino-1,4-benzoquinone. This technology leverages a synergistic cross-linked laccase and lipase aggregate system to catalyze a tandem phenol oxidation and C-N cross-coupling reaction. By addressing the critical bottleneck of product inhibition inherent in single-enzyme systems, this innovation achieves conversion rates exceeding 95% under mild, environmentally benign conditions. For R&D directors and supply chain leaders, this represents a transformative shift from hazardous chemical oxidation to precise biocatalysis, offering a reliable pathway for producing high-purity pharmaceutical intermediates essential for next-generation antitumor agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of amino-p-benzoquinones, which serve as the pharmacophore for Mitomycin C antibiotics, has been plagued by significant technical and economic hurdles. Traditional chemical routes often rely on the direct reaction of primary amines with expensive and inherently unstable 1,4-benzoquinone raw materials, leading to complicated reaction steps and difficult handling procedures. Furthermore, conventional chemical catalysis frequently suffers from poor regioselectivity, resulting in complex impurity profiles that necessitate costly and yield-reducing purification processes. Even when attempting enzymatic solutions, previous iterations utilizing single laccase systems faced a fundamental biochemical barrier: the accumulation of the p-benzoquinone product exerts a strong inhibitory effect on the laccase enzyme itself. This product inhibition severely curtails catalytic efficiency, often capping yields at unacceptably low levels, such as the roughly 32.9% reported in prior art, while generating substantial amounts of unwanted by-products that compromise the purity required for clinical applications.
The Novel Approach
The breakthrough described in the patent data introduces a sophisticated dual-enzyme cascade that elegantly circumvents these historical limitations through a concerted one-pot strategy. By employing a cross-linked aggregate of Myceliophthora thermophila laccase and Aspergillus niger lipase, the process creates a micro-environment where the oxidation of 2,3-dimethylhydroquinone and the subsequent C-N coupling occur simultaneously. The lipase component plays a crucial role by rapidly consuming the p-benzoquinone intermediate as soon as it is generated by the laccase, thereby preventing the buildup of inhibitory concentrations. This synergistic interaction not only stabilizes the reaction kinetics but also drives the equilibrium forward, resulting in a dramatic improvement in yield to over 95%. This approach eliminates the need for unstable quinone starting materials and harsh chemical oxidants, establishing a robust, green manufacturing protocol that aligns perfectly with modern sustainability mandates in fine chemical production.
Mechanistic Insights into Dual-Enzyme Cascade Catalysis
The core of this technological advancement lies in the precise orchestration of two distinct enzymatic activities within a single reaction vessel, effectively creating a self-regulating catalytic cycle. Initially, the laccase, a copper-containing polyphenol oxidase, catalyzes the four-electron oxidation of the inexpensive and stable 2,3-dimethylhydroquinone substrate to generate the reactive 2,3-dimethyl-p-benzoquinone intermediate. In a traditional single-enzyme setup, this intermediate would accumulate and deactivate the catalyst; however, in this dual-system, the co-immobilized lipase immediately engages the quinone. The lipase facilitates a Michael-type addition or C-N cross-coupling between the electrophilic quinone and the nucleophilic aliphatic primary amine donor. This rapid consumption of the quinone maintains its concentration at a level that is optimal for the coupling reaction but too low to inhibit the laccase, effectively decoupling the oxidation step from the inhibition constraint.
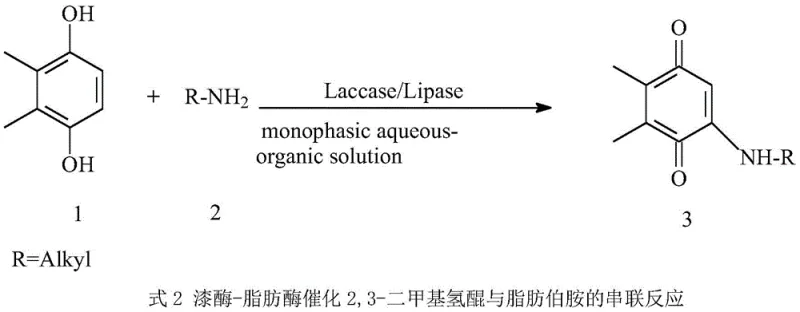
Beyond the kinetic advantages, this mechanistic design offers profound benefits for impurity control and product quality, which are paramount for regulatory compliance in API manufacturing. The high specificity of the enzymes ensures that the amination occurs selectively at the desired position on the benzoquinone ring, minimizing the formation of regioisomers or over-oxidized by-products that are common in metal-catalyzed chemical processes. The use of cross-linked enzyme aggregates (CLEAs) further enhances this selectivity by providing a rigid protein matrix that stabilizes the active conformation of the enzymes against denaturation in the monophasic aqueous-organic solvent system. This structural integrity allows the reaction to proceed at room temperature with high fidelity, ensuring that the final 2,3-dimethyl-5-alkylamino-1,4-benzoquinone product is obtained with exceptional purity after simple recrystallization, thereby reducing the burden on downstream purification units and ensuring a consistent quality profile for the final drug substance.
How to Synthesize 2,3-Dimethyl-5-Alkylamino-1,4-Benzoquinone Efficiently
Implementing this dual-enzyme synthesis requires careful attention to the preparation of the biocatalyst and the maintenance of specific reaction parameters to maximize the synergistic effect. The process begins with the co-aggregation of the two enzymes using ammonium sulfate precipitation followed by cross-linking with glutaraldehyde, creating a robust heterogeneous catalyst capable of withstanding mechanical stirring and repeated reuse. The reaction is conducted in a monophasic system comprising a pH 7.0 phosphate buffer and a minor fraction of water-miscible organic solvent, such as methanol or DMSO, to ensure solubility of the organic substrates while maintaining enzyme hydration. Detailed standard operating procedures regarding enzyme loading ratios, aeration rates, and workup protocols are critical for reproducibility.
- Prepare the biocatalyst by co-aggregating Myceliophthora thermophila laccase and Aspergillus niger lipase using ammonium sulfate precipitation and glutaraldehyde cross-linking to form stable CLEAs.
- Conduct the reaction in a monophasic aqueous-organic system (pH 7.0 buffer with 5% organic solvent) at room temperature, maintaining an amine donor to hydroquinone molar ratio of 1: 1.
- Upon completion, filter the reusable enzyme aggregate, extract the product with ethyl acetate, wash with bicarbonate solution, and recrystallize from ethanol to obtain high-purity red crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this dual-enzyme biocatalytic process offers compelling strategic advantages that extend far beyond simple yield improvements. The elimination of expensive, unstable quinone raw materials and hazardous chemical oxidants fundamentally alters the cost structure of the synthesis, replacing volatile commodity prices with the stability of commercially available enzymes. Furthermore, the ability to operate at room temperature and atmospheric pressure significantly reduces energy consumption and capital expenditure on specialized high-pressure or cryogenic reactor infrastructure. The robustness of the cross-linked enzyme aggregates means that the biocatalyst acts as a reusable asset rather than a single-use consumable, drastically lowering the cost of goods sold (COGS) over the lifecycle of the production campaign and insulating the supply chain from fluctuations in catalyst pricing.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the high atom economy and the reusability of the biocatalyst, which can be recycled for multiple batches without significant loss of activity. By avoiding the use of precious metal catalysts and the associated expensive removal steps required to meet residual metal specifications in pharmaceuticals, manufacturers can achieve substantial cost savings in downstream processing. The high selectivity of the enzymatic route minimizes waste generation and reduces the volume of solvents and reagents needed for purification, leading to a leaner, more cost-effective manufacturing footprint that directly improves margin potential for high-volume API intermediates.
- Enhanced Supply Chain Reliability: Reliance on stable, commercially sourced enzymes and simple hydroquinone starting materials mitigates the supply risks associated with specialized, custom-synthesized chemical reagents that often have long lead times and limited supplier bases. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or sensitivity to moisture and oxygen, ensuring consistent output and reliable delivery schedules. Additionally, the simplified workflow, which combines oxidation and coupling into a single pot, shortens the overall production cycle time, allowing for faster response to market demand fluctuations and improved inventory turnover rates for critical oncology intermediates.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this green chemistry approach aligns seamlessly with increasingly stringent environmental regulations regarding solvent use and heavy metal discharge. The aqueous-based nature of the reaction medium and the absence of toxic heavy metals simplify wastewater treatment and reduce the environmental liability of the manufacturing site. The process is inherently scalable, as demonstrated by the successful operation in jacketed reactors with efficient aeration, facilitating a smooth transition from pilot scale to multi-ton commercial production without the need for complex process re-engineering, thus ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this dual-enzyme technology, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into existing manufacturing lines. The answers provided reflect the specific performance metrics and operational boundaries defined in the intellectual property, ensuring accurate expectations for yield, purity, and catalyst longevity.
Q: How does the dual-enzyme system overcome the limitations of single-enzyme catalysis in benzoquinone synthesis?
A: In single-enzyme systems, the accumulation of p-benzoquinone products inhibits laccase activity, drastically reducing yield. The dual-enzyme system introduces lipase to immediately catalyze the C-N cross-coupling of the generated quinone with the amine donor. This tandem reaction consumes the inhibitory quinone intermediate in situ, relieving product inhibition and boosting overall conversion efficiency significantly.
Q: What are the stability and reusability characteristics of the cross-linked enzyme aggregates (CLEA) used in this process?
A: The cross-linked enzyme aggregates demonstrate exceptional operational stability under mild reaction conditions. Post-reaction, the solid biocatalyst can be simply filtered, washed with ethyl acetate and water, and reused for 8 to 12 consecutive cycles without significant loss of catalytic activity, ensuring consistent process economics.
Q: Why is this enzymatic route preferred over traditional chemical synthesis for Mitomycin C analogs?
A: Traditional chemical methods often suffer from poor selectivity, require harsh conditions, and involve unstable quinone raw materials that complicate handling. This enzymatic route operates at room temperature in a green solvent system, avoids expensive transition metal catalysts, and achieves yields exceeding 95%, making it safer and more environmentally compliant for large-scale API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dimethyl-5-Alkylamino-1,4-Benzoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology intermediates requires more than just a laboratory recipe; it demands a partner with deep expertise in process optimization and industrial scale-up. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to plant floor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced biocatalytic technologies like the dual-enzyme system described in CN107653273B, we help our partners secure a competitive edge through superior quality and sustainable manufacturing practices.
We invite global pharmaceutical companies and contract manufacturers to collaborate with us to unlock the full potential of this green synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this enzymatic approach can optimize your supply chain economics. Please contact us today to request specific COA data for our benzoquinone intermediates and to discuss comprehensive route feasibility assessments that can accelerate your drug development timelines and secure your raw material supply for the future.
