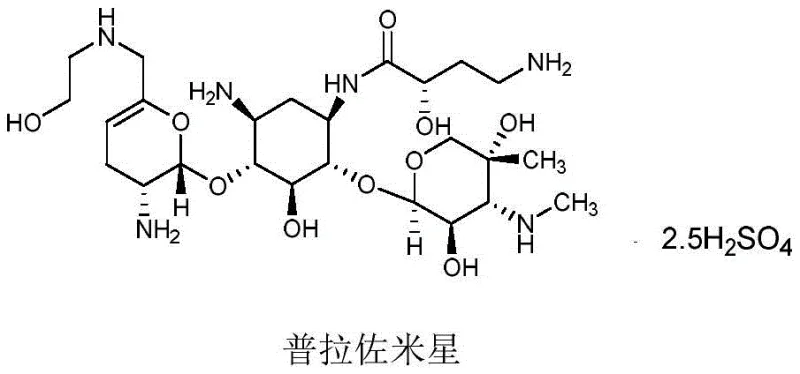
Scalable Prazomicin Synthesis: A Novel Phosphoric Acid Deprotection Strategy for Commercial Production
The landscape of aminoglycoside antibiotic manufacturing is undergoing a significant transformation driven by the need for more efficient and environmentally sustainable synthetic routes. Patent CN111116688B introduces a groundbreaking preparation method for Prazomicin, a next-generation antibiotic critical for treating complicated urinary tract infections and resistant bacterial strains. This innovation specifically addresses the bottlenecks associated with the final deprotection steps, utilizing a phosphoric acid solution to remove tert-butoxycarbonyl (-Boc) protecting groups under remarkably mild conditions. Unlike traditional methods that rely on harsh fluorinated acids or heavy metal catalysts, this approach facilitates the direct isolation of Prazomicin free base solids with purity levels exceeding 95% through simple post-treatment procedures. For R&D Directors and Procurement Managers seeking a reliable antibiotic supplier, this technology represents a pivotal shift towards cost-effective and scalable production capabilities. The structural integrity of the complex aminoglycoside scaffold is meticulously preserved, ensuring that the biological activity required for inhibiting bacterial protein synthesis remains uncompromised throughout the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prazomicin has been plagued by inefficient deprotection strategies that impose severe burdens on both operational costs and environmental compliance. Prior art, such as the methods disclosed in patent CN200880117193.1, relies heavily on trifluoroacetic acid (TFA) to remove protecting groups, necessitating multiple cycles of organic solvent dilution and concentration. This not only consumes vast quantities of expensive solvents but also requires purification via reverse-phase high-performance liquid chromatography, a technique that is notoriously difficult to scale for industrial mass production. Furthermore, alternative routes utilizing palladium on carbon for hydrogenation introduce the risk of heavy metal contamination, requiring rigorous and costly removal steps to meet safety standards. The generation of impurities, such as reduced double bonds during catalytic hydrogenation, further complicates the purification landscape, leading to significantly lower yields and increased waste disposal pressures. These conventional pathways are fundamentally misaligned with the demands of modern green chemistry and lean manufacturing principles.
The Novel Approach
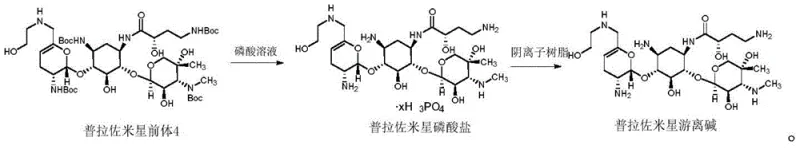
In stark contrast to these cumbersome legacy processes, the novel method disclosed in CN111116688B leverages the unique chemical properties of phosphoric acid to achieve a streamlined deprotection workflow. By reacting Prazomicin precursor 4 with a phosphoric acid solution, preferably at concentrations between 80% and 85%, the process efficiently cleaves the -Boc protecting groups while simultaneously forming a Prazomicin phosphate intermediate that can be easily precipitated. This precipitation step is a critical innovation, as it allows for the physical separation of the product from the reaction matrix without the need for complex chromatographic columns or excessive solvent washing. The subsequent conversion to the free base is achieved through a simple anion exchange resin treatment, which adsorbs the phosphoric acid and releases the pure antibiotic. This approach drastically simplifies the operational workflow, reduces the consumption of hazardous reagents, and enhances the overall safety profile of the manufacturing facility.
Mechanistic Insights into Phosphoric Acid-Catalyzed Deprotection
The efficacy of this novel synthesis route lies in the specific interaction between the phosphoric acid and the acid-labile tert-butoxycarbonyl moieties present on the Prazomicin precursor. Phosphoric acid acts as a proton donor that facilitates the cleavage of the carbamate bond, releasing isobutylene and carbon dioxide while liberating the free amine groups essential for the antibiotic's activity. Unlike stronger mineral acids that might degrade the sensitive glycosidic bonds within the aminoglycoside structure, phosphoric acid provides a controlled acidic environment that maintains the stereochemical integrity of the molecule. The formation of the phosphate salt intermediate is thermodynamically favorable in the presence of anti-solvents like ethanol, driving the equilibrium towards product formation and minimizing the presence of side reactions. This mechanistic pathway ensures that the reaction proceeds to completion with minimal formation of byproducts, thereby reducing the burden on downstream purification units and maximizing the recovery of the valuable active pharmaceutical ingredient.
Impurity control is another cornerstone of this mechanistic advantage, particularly when compared to methods involving heavy metal catalysts or fluorinated acids. The absence of palladium or other transition metals eliminates the risk of metal-catalyzed side reactions, such as the reduction of olefinic bonds, which are common pitfalls in hydrogenation-based deprotection strategies. Furthermore, the use of phosphoric acid avoids the introduction of fluorinated impurities that are difficult to remove and pose significant environmental hazards. The anion exchange resin step serves as a final polishing mechanism, selectively removing phosphate ions while allowing the Prazomicin free base to pass through into the filtrate. This dual mechanism of selective precipitation followed by ion exchange ensures that the final product meets stringent purity specifications, with HPLC data confirming levels consistently above 95% and often reaching nearly 99% in optimized examples.
How to Synthesize Prazomicin Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity while maintaining operational safety. The process begins with the dissolution of the precursor in a suitable organic solvent, followed by the controlled addition of the acid reagent under inert atmosphere conditions. Temperature management is critical, with the reaction preferably conducted at room temperature to prevent thermal degradation of the sensitive antibiotic scaffold. Following the deprotection, the addition of an anti-solvent induces precipitation, allowing for the isolation of the intermediate salt. The final conversion to the free base involves a straightforward resin treatment and concentration step. For a detailed breakdown of the standardized operating procedures and specific parameter ranges, please refer to the technical guide below.
- Dissolve Prazomicin precursor 4 in dichloromethane and add 85% phosphoric acid solution dropwise at room temperature to remove Boc groups.
- Precipitate the resulting Prazomicin phosphate solid by adding ethanol, followed by filtration and dissolution in methanol.
- Treat the solution with anion exchange resin to adsorb phosphoric acid, then filter and concentrate to obtain high-purity Prazomicin free base.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this phosphoric acid-based deprotection method offers substantial advantages for procurement managers and supply chain heads focused on cost reduction in antibiotic manufacturing. The elimination of expensive fluorinated reagents and heavy metal catalysts directly translates to a significant decrease in raw material expenditures, while the simplified workup procedure reduces the consumption of organic solvents and energy. The ability to isolate the product via precipitation rather than chromatography significantly shortens the production cycle time, enhancing the overall throughput of the manufacturing facility. These efficiencies contribute to a more robust and resilient supply chain, capable of meeting high-volume demands without the bottlenecks associated with complex purification technologies. Additionally, the reduced environmental footprint aligns with increasingly stringent global regulatory standards, mitigating the risk of compliance-related disruptions.
- Cost Reduction in Manufacturing: The substitution of trifluoroacetic acid with phosphoric acid eliminates the need for costly fluorinated waste disposal protocols and reduces the reliance on expensive chromatographic resins. By enabling direct precipitation of the intermediate, the process minimizes solvent usage and the associated costs of solvent recovery and disposal. The removal of heavy metal catalysts further reduces expenses related to metal scavenging and validation testing, resulting in a leaner and more cost-effective production model that enhances profit margins for high-purity antibiotic intermediates.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents like phosphoric acid and ethanol ensures a consistent supply of raw materials, reducing the risk of procurement delays associated with specialized or hazardous chemicals. The robustness of the reaction conditions, which tolerate room temperature operations, simplifies equipment requirements and reduces the likelihood of process failures due to thermal control issues. This reliability ensures continuous production capabilities, allowing suppliers to maintain steady inventory levels and meet delivery commitments for complex polymer additives and pharmaceutical intermediates without interruption.
- Scalability and Environmental Compliance: The simplicity of the precipitation and filtration steps makes this process highly scalable from laboratory benchtop to multi-ton commercial production without the need for specialized high-pressure or cryogenic equipment. The reduction in hazardous waste generation, particularly the absence of fluorinated byproducts and heavy metal residues, simplifies environmental compliance and reduces the burden on waste treatment facilities. This eco-friendly profile not only lowers operational costs but also enhances the corporate sustainability image, appealing to partners who prioritize green chemistry principles in their supply chain partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Prazomicin synthesis route. These insights are derived directly from the patent data and are intended to clarify the operational benefits and technical feasibility for potential manufacturing partners. Understanding these details is crucial for evaluating the route's suitability for your specific production requirements and quality standards.
Q: Why is phosphoric acid preferred over trifluoroacetic acid for Prazomicin deprotection?
A: Phosphoric acid allows for the direct precipitation of the phosphate salt, eliminating the need for complex chromatographic purification and extensive solvent washing required by trifluoroacetic acid methods, thereby significantly reducing operational costs and environmental waste.
Q: What purity levels can be achieved with this novel preparation method?
A: The disclosed method consistently yields Prazomicin free base with HPLC purity exceeding 95%, with specific examples demonstrating purity levels up to 99.08%, meeting stringent requirements for antibiotic active pharmaceutical ingredients.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes mild reaction conditions at room temperature, avoids hazardous heavy metal catalysts, and employs simple precipitation and filtration steps, making it highly scalable and safe for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prazomicin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the phosphoric acid deprotection route disclosed in CN111116688B and confirmed its potential for high-efficiency commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of Prazomicin meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this superior synthesis method into your production portfolio. Together, we can drive down costs and enhance the reliability of your antibiotic supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
