Scalable Metal-Free Synthesis of Triazolopyridine Intermediates for Commercial Drug Production
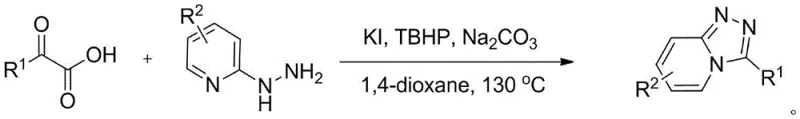
The pharmaceutical industry continuously seeks robust and sustainable synthetic routes for nitrogen-containing heterocycles, particularly those serving as core scaffolds for bioactive molecules. Patent CN108299426B introduces a groundbreaking methodology for the construction of [1,2,4]-triazolo[4,3-a]pyridine derivatives, a class of fused heterocycles known for their potent biological activities, including thrombin inhibition. This innovation addresses critical bottlenecks in traditional synthesis by employing a transition-metal-free protocol that utilizes potassium iodide (KI) as an inexpensive catalyst and tert-butyl hydroperoxide (TBHP) as a green oxidant. The general structural framework of these valuable intermediates is depicted below, showcasing the versatility of the substitution patterns achievable through this novel approach.
![General structural formula of [1,2,4]-triazolo[4,3-a]pyridine compounds showing variable R1 and R2 substituents](/insights/img/triazolopyridine-synthesis-ki-catalyst-pharma-supplier-20260306144249-01.png)
For R&D directors and process chemists, the significance of this patent lies in its ability to generate a diverse library of analogues efficiently. The method tolerates a wide range of functional groups on both the alpha-keto acid and the 2-hydrazinopyridine starting materials, allowing for the rapid exploration of structure-activity relationships (SAR). By avoiding the use of precious metals, the process not only aligns with green chemistry principles but also mitigates the risk of metal contamination in the final active pharmaceutical ingredient (API). This is particularly crucial for cardiovascular drugs where impurity profiles are strictly regulated. The reaction proceeds through a tandem sequence involving cyclization, decarboxylation, and aromatization, delivering the target heterocyclic core in high purity with minimal downstream processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazolopyridine scaffolds has relied heavily on transition metal catalysis, often utilizing copper or palladium complexes to facilitate the necessary bond formations. These conventional pathways present substantial challenges for commercial manufacturing, primarily due to the high cost of the catalysts and the rigorous purification steps required to reduce residual metal levels to parts-per-million specifications. Furthermore, many traditional methods employ harsh reaction conditions, including strong acids or bases and elevated temperatures that can compromise sensitive functional groups, leading to lower overall yields and complex impurity profiles. The reliance on stoichiometric amounts of expensive oxidants or hazardous reagents further exacerbates the environmental footprint and operational costs, making these routes less attractive for large-scale production of pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages a simple yet effective iodide-catalyzed oxidative cyclization strategy that operates under significantly milder conditions. By utilizing molecular oxygen or organic peroxides like TBHP in conjunction with catalytic KI, the reaction achieves efficient C-N and C-C bond formation without the need for toxic heavy metals. This shift represents a paradigm change in how these heterocycles are manufactured, offering a pathway that is not only cost-effective but also environmentally benign. The use of readily available starting materials, such as alpha-keto acids and hydrazinopyridines, ensures a stable supply chain, while the straightforward workup involving standard extraction and chromatography simplifies the isolation of the pure product. This approach effectively resolves the pain points of cost, safety, and scalability associated with legacy synthetic routes.
Mechanistic Insights into KI-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation is a sophisticated example of tandem organocatalysis, where the iodide ion acts as a redox mediator to activate the oxidant. The reaction initiates with the interaction between the alpha-keto acid and the hydrazine moiety, likely forming a hydrazone intermediate that undergoes subsequent oxidation. The presence of TBHP facilitates the generation of reactive iodine species in situ, which promote the cyclization step necessary to close the triazole ring. Following cyclization, the system undergoes a spontaneous decarboxylation event, driven by the thermodynamic stability of the resulting aromatic system. This cascade sequence is highly efficient, minimizing the accumulation of unstable intermediates and ensuring a clean conversion to the final triazolopyridine product. Understanding this mechanism is vital for process optimization, as it highlights the critical balance between oxidant concentration and reaction temperature to maximize yield.
From an impurity control perspective, the metal-free nature of this catalytic cycle offers distinct advantages. Traditional metal-catalyzed reactions often suffer from side reactions such as homocoupling or over-oxidation mediated by the metal center, which can generate difficult-to-remove byproducts. In the KI-catalyzed system, the selectivity is governed by the specific redox potential of the iodine species, which favors the desired cyclization pathway. Additionally, the absence of transition metals eliminates the formation of metal-organic complexes that can persist through purification stages. This results in a cleaner crude reaction mixture, reducing the burden on downstream purification units and enhancing the overall mass balance of the process. For quality control teams, this translates to more consistent batch-to-batch reproducibility and a simpler analytical profile for release testing.
How to Synthesize [1,2,4]-Triazolo[4,3-a]pyridine Efficiently
The practical execution of this synthesis is designed for ease of operation in standard chemical reactors. The protocol typically involves charging the reactor with the alpha-keto acid and 2-hydrazinopyridine substrates in a 1:1 molar ratio, followed by the addition of the catalytic system comprising potassium iodide and sodium carbonate. The reaction is conducted in 1,4-dioxane, a solvent chosen for its ability to dissolve both organic substrates and inorganic salts while maintaining stability at the required reaction temperature of 130°C. Upon heating, the oxidant TBHP is introduced to drive the oxidative cyclization. The detailed standardized operating procedures, including precise addition rates, safety protocols for handling peroxides, and specific quenching methods, are outlined in the comprehensive guide below to ensure safe and reproducible manufacturing.
- Combine alpha-keto acid and 2-hydrazinopyridine substrates in a dry reaction vessel with potassium iodide catalyst and sodium carbonate base.
- Add 1,4-dioxane solvent and TBHP oxidant, then heat the mixture to 130°C for reflux.
- Maintain reaction for 12 hours to complete tandem cyclization and decarboxylation, followed by extraction and silica gel purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this KI-catalyzed synthesis route offers compelling economic and logistical benefits. The primary driver for cost reduction is the substitution of expensive transition metal catalysts with commodity chemicals like potassium iodide and sodium carbonate, which are available in bulk quantities at a fraction of the cost. This fundamental change in the bill of materials directly impacts the gross margin of the intermediate, allowing for more competitive pricing in the global market. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, lowering waste disposal costs and improving the overall process mass intensity (PMI). These efficiencies accumulate to create a leaner, more profitable manufacturing operation that is resilient to fluctuations in the price of precious metals.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as palladium or copper removes a significant variable cost component from the production budget. Additionally, the high atom economy of the decarboxylative cyclization minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable product. This efficiency extends to the utility consumption, as the moderate reaction temperature of 130°C requires less energy input compared to high-temperature pyrolysis methods often used in heterocycle synthesis. Consequently, the total cost of goods sold (COGS) is substantially reduced, enhancing the commercial viability of projects utilizing these intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on widely available, non-specialized reagents. Unlike custom-synthesized ligands or specialized metal complexes that may have long lead times and single-source risks, potassium iodide and TBHP are standard inventory items for most chemical suppliers. This availability ensures continuity of supply even during market disruptions, reducing the risk of production stoppages. Moreover, the robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to diversify their supply base across different geographic regions without compromising on quality or yield.
- Scalability and Environmental Compliance: The green chemistry attributes of this process align perfectly with increasingly stringent environmental regulations. By avoiding toxic heavy metals, the facility reduces its liability regarding hazardous waste treatment and effluent discharge. The simplified workup procedure generates less chemical waste, contributing to a smaller environmental footprint and easier compliance with ISO 14001 standards. From a scalability standpoint, the homogeneous nature of the reaction mixture and the absence of exothermic hazards associated with metal catalysis make the transition from pilot plant to commercial scale straightforward and safe, facilitating rapid time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of the KI-catalyzed method over transition metal catalysis?
A: The KI-catalyzed method eliminates the need for expensive and toxic transition metals like copper or palladium, significantly reducing raw material costs and simplifying the removal of heavy metal residues to meet stringent pharmaceutical purity standards.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes inexpensive reagents such as potassium iodide and sodium carbonate, operates under relatively mild conditions at 130°C, and involves a straightforward workup procedure, making it highly amenable to scale-up from laboratory to industrial tonnage.
Q: What is the biological significance of the synthesized triazolopyridine compounds?
A: These compounds serve as critical intermediates for developing thrombin inhibitors, which are essential in the prevention and treatment of thromboembolic diseases, demonstrating significant in vitro thrombolytic activity compared to blank controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [1,2,4]-Triazolo[4,3-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN108299426B for accelerating drug development pipelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of [1,2,4]-triazolo[4,3-a]pyridine intermediate delivered meets the highest industry standards for potency and impurity control. We are committed to leveraging our technical expertise to optimize this metal-free process for your specific volume requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic benefits specific to your project scale. We encourage you to contact us today to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments tailored to your timeline and budget constraints, ensuring a successful partnership for your antithrombotic drug development programs.
