Scalable Blue-Light Mediated Synthesis of 3-Thioether Quinoxalinone Intermediates for Pharma
Introduction to Advanced Photocatalytic Synthesis
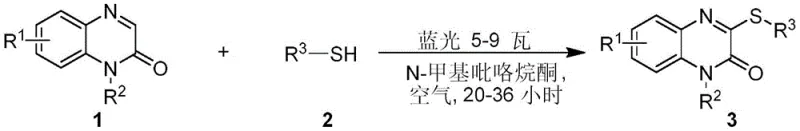
The pharmaceutical industry continuously seeks efficient, sustainable, and cost-effective methodologies for constructing complex heterocyclic scaffolds essential for drug development. A significant breakthrough in this domain is documented in patent CN110540525B, which discloses a novel synthetic method for 3-thioether-based quinoxalinone compounds. These heterocyclic structures are pivotal in medicinal chemistry, exhibiting potent biological activities including inhibition of Plasmodium falciparum, aldose reductase, and HIV replication. The disclosed technology represents a paradigm shift from classical multi-step syntheses to a direct, visible-light-mediated functionalization strategy. By leveraging blue light irradiation at room temperature, this process eliminates the need for harsh reaction conditions, toxic heavy metal catalysts, and excessive stoichiometric reagents. For R&D directors and procurement specialists, this innovation offers a compelling value proposition: a streamlined pathway to high-purity intermediates that aligns with modern green chemistry principles while potentially reducing overall manufacturing lead times and operational costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-substituted quinoxalinone derivatives has been a laborious and chemically intensive endeavor. The classical synthetic route typically initiates with quinoxaline-2,3(1H,4H)-dione as the starting material. This precursor must undergo a rigorous four-step sequence involving chlorination, selective hydrolysis, alkylation, and finally, a nucleophilic substitution tandem reaction to install the desired thioether moiety. Each of these steps introduces significant inefficiencies; the chlorination step often requires hazardous reagents, while the subsequent alkylation and substitution reactions frequently demand strong bases in stoichiometric or excess amounts to drive the equilibrium forward. These harsh conditions not only pose safety risks and environmental burdens due to waste generation but also limit the functional group tolerance, often leading to decomposition of sensitive substrates. Furthermore, the cumulative yield loss across four distinct isolation and purification stages drastically impacts the overall economic viability of producing these valuable pharmaceutical intermediates on a commercial scale.
The Novel Approach
In stark contrast, the methodology outlined in CN110540525B offers a remarkably concise and elegant solution. This innovative approach utilizes a direct coupling between quinoxalinone and thiophenol derivatives under visible light irradiation. The reaction proceeds smoothly in N-methylpyrrolidone (NMP) solvent at room temperature under an air atmosphere, requiring no inert gas protection or cryogenic cooling. The simplicity of the setup—merely mixing the substrates and exposing them to a low-power blue light source (5-9 Watts)—dramatically lowers the barrier to entry for implementation. This single-step transformation bypasses the need for pre-functionalization of the quinoxalinone ring, thereby conserving raw materials and reducing solvent consumption associated with intermediate isolations. The robustness of this photocatalytic system allows for a wide range of substrates to be converted efficiently, providing a versatile platform for generating diverse libraries of bioactive molecules.
Mechanistic Insights into Visible-Light Mediated Thiolation
The core of this technological advancement lies in the activation of the sulfur-hydrogen bond in thiophenols via visible-light photoredox catalysis or direct photo-excitation. Under blue light irradiation, the thiophenol species can generate thiyl radicals, which are highly reactive intermediates capable of attacking the electron-deficient positions on the quinoxalinone ring. This radical mechanism circumvents the high energy barriers associated with traditional ionic nucleophilic substitutions that require strong bases. The reaction environment is exceptionally mild, maintaining neutrality or near-neutrality, which is crucial for preserving acid- or base-sensitive functional groups often present in complex drug candidates. The use of air as the oxidant further simplifies the process, suggesting that molecular oxygen may play a role in regenerating the active catalytic species or facilitating the oxidative coupling, thus eliminating the need for external chemical oxidants that could generate hazardous byproducts.
From an impurity control perspective, this mechanism offers superior selectivity. Traditional methods involving chlorination often produce poly-chlorinated byproducts or regioisomers that are difficult to separate, complicating the purification process and lowering the final purity profile. The photocatalytic route described herein demonstrates high regioselectivity for the 3-position of the quinoxalinone scaffold. The mild conditions minimize side reactions such as over-oxidation of the sulfur atom to sulfoxides or sulfones, which are common pitfalls in thiol chemistry. Consequently, the crude reaction mixtures are cleaner, facilitating easier downstream processing via standard extraction and flash column chromatography. This enhanced purity profile is critical for pharmaceutical applications where strict regulatory limits on impurities must be met, ensuring that the final API intermediates comply with stringent quality standards without requiring extensive recrystallization or preparative HPLC.
How to Synthesize 3-Thioether Quinoxalinone Efficiently
Implementing this synthesis protocol in a laboratory or pilot plant setting is straightforward and requires minimal specialized equipment beyond a standard blue LED light source. The process begins with the precise weighing of the quinoxalinone substrate and the corresponding thiophenol derivative, typically in a molar ratio favoring the thiol to ensure complete conversion of the heterocycle. These reagents are dissolved in N-methylpyrrolidone, a polar aprotic solvent known for its ability to dissolve a wide range of organic compounds and stabilize radical intermediates. The reaction mixture is then placed in a quartz tube, which is transparent to the specific wavelength of blue light used, ensuring maximum photon flux reaches the reactants. Following the irradiation period, the workup involves a standard aqueous quench and organic extraction, making it compatible with existing infrastructure in most chemical manufacturing facilities. Detailed standardized synthesis steps follow below.
- Combine quinoxalinone substrate and thiophenol derivative in N-methylpyrrolidone solvent within a quartz reaction vessel.
- Irradiate the reaction mixture with blue light (5-9 Watts) at room temperature under air atmosphere for 20-36 hours.
- Quench the reaction with water, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic synthesis method translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of multiple synthetic steps inherently reduces the consumption of solvents, reagents, and energy, leading to a lower cost of goods sold (COGS). By avoiding the use of expensive transition metal catalysts or hazardous chlorinating agents, the process mitigates the risks associated with volatile raw material pricing and supply chain disruptions for specialty chemicals. Furthermore, the simplified workflow reduces the labor hours required for production and quality control testing, as fewer intermediate checkpoints are necessary. This efficiency gain allows manufacturers to respond more agilely to market demands, shortening the lead time from order placement to delivery of high-purity intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this protocol is the drastic reduction in unit operations. Traditional four-step syntheses incur costs related to heating, cooling, and isolation at every stage. By consolidating this into a single room-temperature reaction, energy expenditures are significantly minimized. Additionally, the avoidance of stoichiometric bases and chlorinating agents removes the cost burden of purchasing these reagents and treating the resulting acidic or saline waste streams. The high atom economy of the direct thiolation ensures that a greater proportion of the input mass is converted into the desired product, reducing waste disposal fees and maximizing raw material utilization efficiency.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, namely substituted quinoxalinones and thiophenols, are commodity chemicals available from multiple global suppliers. This diversity in sourcing prevents bottlenecks that often occur when a synthesis relies on a single-source custom intermediate. The robustness of the reaction conditions—specifically the tolerance to air and moisture—means that production is less susceptible to delays caused by stringent environmental controls or equipment failures associated with inert atmosphere processing. This resilience ensures a consistent and uninterrupted supply of critical pharmaceutical intermediates, safeguarding downstream drug production schedules against upstream volatility.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but advancements in flow chemistry and reactor design have made this increasingly viable. The low power requirement (5-9 Watts per small scale) suggests that the reaction is efficient, and scaling can be achieved through numbering up reactors rather than increasing vessel size, maintaining consistent light exposure. From an environmental standpoint, the process aligns with green chemistry metrics by operating at room temperature and using air as a benign oxidant. This reduces the carbon footprint of the manufacturing process and simplifies compliance with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this blue-light mediated synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of the method's capabilities and limitations for potential partners and licensees. Understanding these nuances is essential for integrating this chemistry into existing production pipelines effectively.
Q: What are the advantages of this blue-light method over traditional chlorination routes?
A: Traditional methods require harsh chlorination, hydrolysis, and alkylation steps involving excess bases and toxic reagents. This novel photocatalytic approach operates at room temperature under air, significantly reducing energy consumption and hazardous waste generation while simplifying the operational workflow.
Q: Is this synthesis method scalable for commercial production of pharmaceutical intermediates?
A: Yes, the protocol utilizes readily available starting materials and standard organic solvents like NMP and ethyl acetate. The absence of sensitive transition metal catalysts and the use of ambient temperature conditions make the process highly amenable to scale-up from laboratory to industrial manufacturing environments.
Q: What is the substrate scope regarding electron-withdrawing and electron-donating groups?
A: The method demonstrates excellent tolerance for diverse substituents. It successfully accommodates electron-withdrawing groups such as chloro, bromo, nitro, and trifluoromethyl, as well as electron-donating groups like methoxy and methyl on both the quinoxalinone ring and the thiophenol moiety, ensuring broad applicability for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Thioether Quinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light photocatalysis in modern pharmaceutical manufacturing. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial supply is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-thioether quinoxalinone meets the highest quality standards required for clinical and commercial drug applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthetic route for their pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a reliable and cost-effective supply chain for your critical intermediates.
