Advanced 2-Step Synthesis of 2-Chloro-5-Aminopyrimidine for Commercial Scale-Up
Introduction to Patent CN101219997A
The pharmaceutical industry constantly seeks more efficient pathways for constructing complex heterocyclic scaffolds, and patent CN101219997A presents a transformative approach to synthesizing 2-chloro-5-aminopyrimidine, a critical building block for various bioactive molecules. This intellectual property discloses a robust methodology that drastically condenses the synthetic timeline from a cumbersome six-step sequence into a highly efficient two-step process. By leveraging 5-nitrouracil as a readily available starting material, the invention circumvents the need for hazardous halogenation and nitration steps typically associated with traditional furan-based routes. The technical breakthrough lies not only in the reduction of unit operations but also in the strategic selection of reagents that enhance safety profiles and environmental compliance. For R&D directors and process chemists, this patent represents a significant opportunity to optimize impurity profiles and boost overall throughput without compromising on the structural integrity of the final pyrimidine derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-5-aminopyrimidine has been plagued by inefficiencies inherent to multi-step linear syntheses starting from 2-furancarboxylic acid. As illustrated in the reaction scheme below, the conventional pathway requires six distinct chemical transformations, beginning with the dangerous bromination of the furan ring followed by nitrosation and cyclization. 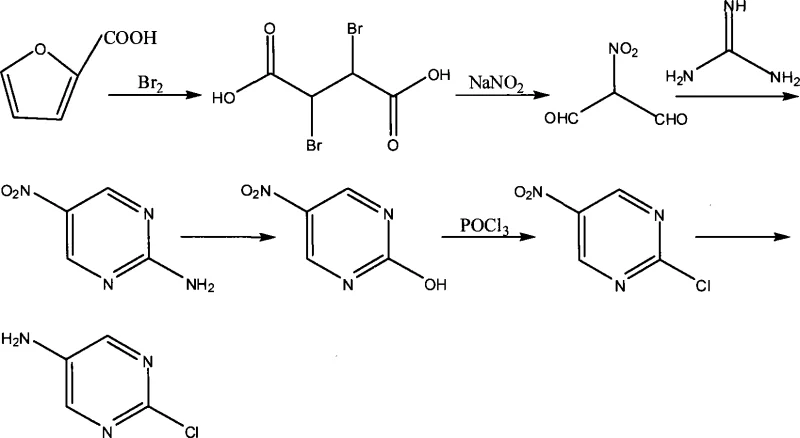 This legacy route necessitates the handling of liquid bromine and sodium nitrite, reagents known for their high toxicity and potential to generate hazardous waste streams that complicate regulatory compliance. Furthermore, each additional step in the sequence introduces opportunities for yield loss and the accumulation of difficult-to-remove impurities, ultimately resulting in a low gross production rate that is economically unsustainable for large-scale manufacturing. The reliance on such a fragmented process also extends lead times and increases the capital expenditure required for equipment and containment systems capable of managing corrosive and toxic intermediates.
This legacy route necessitates the handling of liquid bromine and sodium nitrite, reagents known for their high toxicity and potential to generate hazardous waste streams that complicate regulatory compliance. Furthermore, each additional step in the sequence introduces opportunities for yield loss and the accumulation of difficult-to-remove impurities, ultimately resulting in a low gross production rate that is economically unsustainable for large-scale manufacturing. The reliance on such a fragmented process also extends lead times and increases the capital expenditure required for equipment and containment systems capable of managing corrosive and toxic intermediates.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent utilizes a convergent strategy that initiates with 5-nitrouracil, a stable and commercially abundant precursor. This innovative route collapses the synthesis into two primary stages: an initial chlorination to form 2,4-dichloro-5-nitropyrimidine, followed by a selective reduction that simultaneously installs the amine functionality and removes the unwanted chlorine atom at the 4-position. 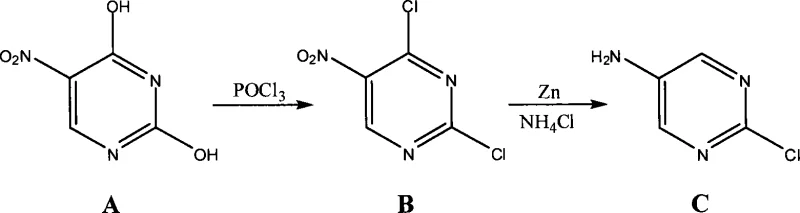 By bypassing the construction of the pyrimidine ring from scratch, the process eliminates the most hazardous steps of the traditional method, specifically avoiding the use of elemental bromine. The simplicity of this approach allows for easier purification protocols, often requiring only extraction and recrystallization to achieve high purity standards. This streamlined workflow not only accelerates the time-to-market for downstream API production but also aligns with green chemistry principles by reducing the overall E-factor and minimizing the consumption of auxiliary materials.
By bypassing the construction of the pyrimidine ring from scratch, the process eliminates the most hazardous steps of the traditional method, specifically avoiding the use of elemental bromine. The simplicity of this approach allows for easier purification protocols, often requiring only extraction and recrystallization to achieve high purity standards. This streamlined workflow not only accelerates the time-to-market for downstream API production but also aligns with green chemistry principles by reducing the overall E-factor and minimizing the consumption of auxiliary materials.
Mechanistic Insights into POCl3-Mediated Chlorination and Zinc Reduction
The first stage of this synthesis involves a vigorous chlorination reaction where 5-nitrouracil is treated with phosphorus oxychloride (POCl3) in the presence of an organic base such as triethylamine or diisopropylethylamine. Mechanistically, the organic base acts as a proton scavenger, facilitating the nucleophilic attack of the pyrimidine oxygen atoms on the phosphorus center, which subsequently activates the ring for chloride substitution. The reaction conditions, typically maintained between 30°C and 105°C, ensure complete conversion to the 2,4-dichloro-5-nitropyrimidine intermediate while minimizing thermal degradation. The choice of base is critical; bulky amines like DIPEA can help moderate the exotherm and improve the solubility of the intermediate species, leading to cleaner reaction profiles and higher isolated yields ranging from 88% to 95% in optimized embodiments.
The second stage is a masterpiece of chemoselectivity, utilizing zinc dust in a weakly acidic aqueous environment to effect a dual transformation. In this step, the zinc serves as a reducing agent that targets both the nitro group at the 5-position and the chlorine atom at the 4-position, while remarkably leaving the chlorine at the 2-position intact. The weakly acidic medium, provided by ammonium chloride or acetic acid, protonates the intermediate species and facilitates the electron transfer from the zinc surface. This specific reactivity pattern is governed by the electronic differences between the 2 and 4 positions on the pyrimidine ring, where the 4-chloro group is more susceptible to nucleophilic displacement or reduction under these specific conditions. This single-pot reduction-dechlorination strategy eliminates the need for separate protection and deprotection steps, significantly simplifying the downstream processing and workup procedures.
How to Synthesize 2-Chloro-5-Aminopyrimidine Efficiently
To implement this synthesis effectively, operators must strictly control the stoichiometry of phosphorus oxychloride and the organic base during the initial chlorination to prevent the formation of poly-chlorinated byproducts. The subsequent reduction step requires careful monitoring of temperature and zinc addition rates to manage hydrogen evolution and ensure complete conversion without over-reduction of the pyrimidine ring. Detailed standard operating procedures regarding solvent selection for extraction and recrystallization are essential to maximize recovery and purity.
- Chlorinate 5-nitrouracil with phosphorus oxychloride and an organic base at 30-105°C to form 2,4-dichloro-5-nitropyrimidine.
- Reduce the intermediate using zinc dust in a weakly acidic aqueous solution (e.g., ammonium chloride or acetic acid) at 30-100°C.
- Filter, extract with organic solvent, and recrystallize to obtain high-purity 2-chloro-5-aminopyrimidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to this 2-step methodology offers profound economic benefits driven primarily by the drastic reduction in processing time and raw material complexity. By eliminating four synthetic steps, manufacturers can significantly reduce labor costs, utility consumption, and the depreciation burden on reactor assets, leading to a much lower cost of goods sold (COGS). The avoidance of specialized containment required for liquid bromine further lowers capital investment barriers, making the production facility more versatile and safer to operate. Additionally, the use of 5-nitrouracil, a commodity chemical with a robust global supply chain, mitigates the risk of raw material shortages that often plague custom-synthesized starting materials.
- Cost Reduction in Manufacturing: The consolidation of the synthesis into two high-yielding steps inherently drives down manufacturing costs by reducing the cumulative loss of material associated with isolation and purification at each stage. Eliminating the need for expensive and hazardous reagents like liquid bromine and sodium nitrite removes significant waste disposal costs and regulatory compliance fees. The simplified workflow also reduces the demand for solvents and energy, contributing to substantial operational expenditure savings that can be passed down to the customer or reinvested in R&D.
- Enhanced Supply Chain Reliability: Relying on 5-nitrouracil as the feedstock ensures a stable supply chain, as this material is produced in large volumes for various agricultural and pharmaceutical applications, unlike niche intermediates that may have single-source suppliers. The shorter production cycle time means that inventory turnover is faster, allowing for more responsive fulfillment of customer orders and reduced working capital tied up in work-in-progress inventory. This agility is crucial for maintaining continuity of supply in the face of market fluctuations or unexpected demand surges.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the use of extremely hazardous reagents that require specialized, small-batch handling equipment, allowing for straightforward scale-up from pilot plants to multi-ton commercial reactors. The reduction in toxic waste generation aligns with increasingly stringent environmental regulations, reducing the liability and permitting hurdles associated with chemical manufacturing. Furthermore, the ability to recycle solvents and auxiliary materials like phosphorus oxychloride derivatives enhances the sustainability profile of the operation, appealing to eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and quality expectations for potential partners considering this technology for their supply chain.
Q: What are the primary advantages of the 5-nitrouracil route over the furan-carboxylic acid route?
A: The 5-nitrouracil route reduces the synthesis from six steps to just two, eliminating the use of highly toxic reagents like liquid bromine and sodium nitrite, thereby significantly improving safety and overall yield.
Q: How is selectivity achieved during the reduction step to preserve the 2-chloro group?
A: Selectivity is controlled by using zinc dust in a weakly acidic aqueous medium, which facilitates the reduction of the nitro group and the removal of the 4-chloro substituent while retaining the 2-chloro group intact.
Q: Is the starting material 5-nitrouracil commercially viable for large-scale production?
A: Yes, 5-nitrouracil is a commercially available commodity chemical with a stable supply chain, making it a cost-effective and reliable starting material for industrial manufacturing compared to custom-synthesized precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Aminopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2-chloro-5-aminopyrimidine meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence allows us to deliver consistent quality while optimizing the cost structure of your supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, reliability, and mutual growth.
