Advanced Manufacturing of 2-n-butyl-4-chloro-5-formylimidazole for Global Losartan Supply Chains
The global demand for Angiotensin II receptor blockers, particularly Losartan, necessitates a robust and sustainable supply chain for its critical precursors. Patent CN111592495A introduces a transformative preparation method for 2-n-butyl-4-chloro-5-formylimidazole, a pivotal intermediate in the synthesis of Losartan potassium. This innovation addresses long-standing inefficiencies in heterocyclic chemistry by shifting away from hazardous phosphorus-based reagents toward a greener, oxidative protocol. By leveraging valeraldehyde as a cost-effective starting material, this technology offers a streamlined three-step sequence involving cyclization, chlorination, and oxidation. For pharmaceutical manufacturers and procurement strategists, adopting this methodology represents a significant opportunity to optimize cost structures while enhancing environmental compliance. The following analysis details the technical merits and commercial viability of this advanced synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-n-butyl-4-chloro-5-formylimidazole has relied heavily on valeronitrile-derived pathways or the use of aggressive phosphorylating agents. Traditional protocols, such as those cited in US Patent 5,442,076 and US 5,606,072, typically employ phosphorus oxychloride (POCl3) in conjunction with DMF for Vilsmeier-Haack formylation. While chemically effective, these methods impose severe operational burdens on large-scale facilities. The generation of substantial quantities of phosphorus-containing wastewater creates a complex and expensive disposal challenge, often requiring specialized neutralization and treatment infrastructure. Furthermore, the handling of POCl3 poses significant safety risks due to its corrosive nature and reactivity with moisture, necessitating rigorous containment protocols that increase capital expenditure. Additionally, the separation and purification of the final product from phosphorus byproducts can be cumbersome, often leading to yield losses and extended cycle times that negatively impact overall plant throughput.
The Novel Approach
In stark contrast, the methodology disclosed in CN111592495A circumvents these bottlenecks by utilizing a completely different mechanistic strategy centered on oxidative formylation. Instead of introducing the formyl group via phosphorus reagents, this novel approach constructs the imidazole ring first using valeraldehyde and methylglyoxal, followed by selective chlorination and a final oxidation step using sodium persulfate. This shift eliminates the generation of toxic phosphorus waste entirely, replacing it with benign byproducts like sodium sulfate and hydrochloric acid, which are far easier to manage and potentially recoverable. The reaction conditions are notably milder, with key steps operating effectively at temperatures ranging from 15°C to 70°C, reducing energy consumption compared to high-temperature alternatives. This streamlined workflow not only simplifies the post-reaction workup but also enhances the safety profile of the manufacturing process, making it highly attractive for modern, regulation-compliant chemical production facilities seeking to minimize their environmental footprint.
Mechanistic Insights into Oxidative Formylation and Cyclization
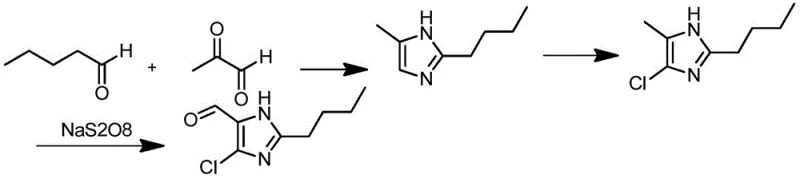
The core of this technological advancement lies in its elegant three-stage reaction sequence, which maximizes atom economy and minimizes side reactions. The process initiates with the cyclization of valeraldehyde and methylglyoxal in the presence of ammonia water. This condensation reaction efficiently constructs the 2-butyl-5-methylimidazole scaffold. The molar ratios are carefully optimized, typically employing a ratio of valeraldehyde to methylglyoxal to ammonia of approximately 1:1.2-1.6:2-3, ensuring complete conversion while suppressing oligomerization. Following ring closure, the intermediate undergoes electrophilic chlorination. Reagents such as sulfuryl chloride or trichloroisocyanuric acid are employed to introduce the chlorine atom at the 4-position of the imidazole ring. This step is critical for setting up the subsequent oxidation, and the patent highlights that this can be performed in common solvents like dichloromethane or methanol with high selectivity.
The final and most innovative step is the oxidation of the methyl group to a formyl group using sodium persulfate (Na2S2O8) catalyzed by transition metal salts like copper sulfate. This oxidative transformation occurs under relatively mild thermal conditions, typically maintained below 50°C to prevent over-oxidation to the carboxylic acid. The mechanism likely involves the generation of sulfate radical anions which abstract hydrogen from the methyl group, facilitating its conversion to the aldehyde functionality. This specific choice of oxidant is crucial; unlike chromic acid or other heavy metal oxidants, sodium persulfate leaves behind inorganic salts that do not contaminate the organic phase with toxic metals. The resulting crude product can be purified to ≥99% purity through simple crystallization techniques, such as dissolving in isopropanol and precipitating with water. This high level of purity is essential for downstream coupling reactions in API synthesis, ensuring that impurity profiles remain within strict regulatory limits.
How to Synthesize 2-n-butyl-4-chloro-5-formylimidazole Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process is designed to be telescoped where possible, reducing the need for intermediate isolation and drying, which significantly cuts down processing time. The initial cyclization can be conducted in a one-pot fashion, followed by direct extraction or solvent swap for the chlorination step. The final oxidation is exothermic and requires controlled addition of the oxidant to maintain thermal stability. Detailed standard operating procedures regarding stoichiometry, addition rates, and temperature ramps are critical for successful scale-up. Operators must ensure that the pH and solvent systems are compatible across the sequence to avoid emulsion formation during workups.
- Cyclize valeraldehyde and methylglyoxal with ammonia water at 15-70°C to obtain 2-butyl-5-methylimidazole.
- Perform chlorination on 2-butyl-5-methylimidazole using reagents like sulfuryl chloride or trichloroisocyanuric acid at 0-60°C.
- Oxidize the chlorinated intermediate using sodium persulfate and a copper catalyst at temperatures below 50°C to yield the final formyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent-covered methodology offers tangible strategic benefits beyond mere technical feasibility. The shift from specialized nitrile derivatives or hazardous phosphorus reagents to commodity chemicals like valeraldehyde and sodium persulfate fundamentally alters the cost structure of the intermediate. Valeraldehyde is a widely available bulk chemical with a stable supply chain, reducing the risk of raw material shortages that often plague niche pharmaceutical precursors. Furthermore, the elimination of phosphorus waste translates directly into reduced operational expenditures related to waste treatment and regulatory compliance. Facilities can avoid the high costs associated with treating phosphorus-laden effluent, which often requires precipitation and sludge disposal. This results in a cleaner, more sustainable manufacturing profile that aligns with increasingly stringent global environmental standards.
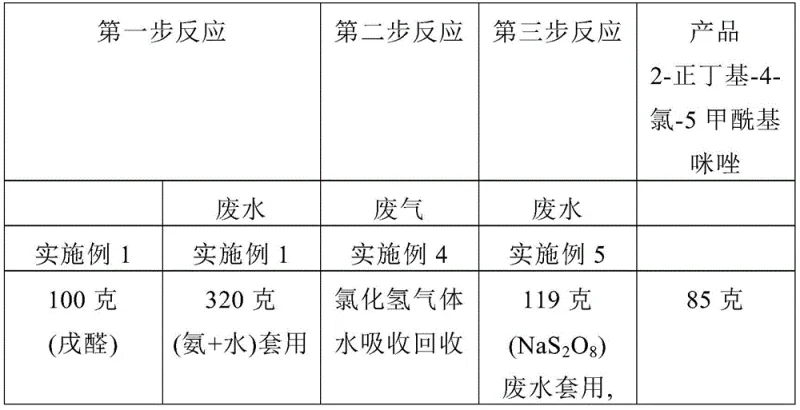
- Cost Reduction in Manufacturing: The economic advantage of this route is driven by the substitution of expensive and hazardous reagents with cost-effective alternatives. By removing the need for phosphorus oxychloride and the associated heavy metal catalysts often used in older oxidation methods, the bill of materials is significantly optimized. The simplified purification process, which relies on crystallization rather than complex chromatography or distillation, further reduces utility costs and solvent consumption. Additionally, the high yield reported in the patent embodiments, reaching up to 85% in the oxidation step, ensures that raw material input is efficiently converted to saleable product, minimizing waste-related financial losses.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for continuous API production. This method utilizes reagents that are standard stock items in the fine chemical industry, mitigating the risk of supply disruptions common with custom-synthesized intermediates. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, adds a layer of operational flexibility. In the event of a specific solvent shortage, the process can often be adapted to use alternative chlorinated or alcoholic solvents without compromising the reaction outcome. This flexibility ensures that production schedules remain intact even when facing minor logistical challenges in the raw material market.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this route is inherently safer for large-volume production. The absence of highly corrosive POCl3 reduces equipment maintenance costs and extends the lifespan of reactor vessels. Moreover, the byproducts generated, primarily sodium sulfate and hydrochloric acid, are manageable and potentially recoverable for other industrial uses, supporting a circular economy approach. This environmental stewardship not only reduces disposal fees but also enhances the corporate social responsibility profile of the manufacturing entity, a key factor for multinational pharmaceutical clients auditing their supply chains.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new synthesis technology is vital for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding yield consistency, impurity management, and regulatory alignment. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a transparent view of the process capabilities. Addressing these points early helps in forming a realistic implementation strategy and setting accurate expectations for project timelines and quality metrics.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: Unlike traditional methods relying on phosphorus oxychloride (POCl3) which generate hazardous phosphorus waste, this patented route utilizes sodium persulfate oxidation. This significantly reduces environmental impact, simplifies wastewater treatment, and lowers overall production costs by eliminating expensive heavy metal removal steps.
Q: What purity levels can be achieved with this manufacturing process?
A: The process allows for the production of crude products that can be further purified via crystallization. Specific embodiments in the patent demonstrate achieving purity levels of ≥99% through simple isopropanol dissolution and water precipitation, ensuring suitability for high-grade API synthesis.
Q: Is this process scalable for commercial industrial production?
A: Yes, the method is designed for industrial scalability. It uses readily available commodity chemicals like valeraldehyde and operates under mild temperature conditions (mostly below 70°C). The one-pot nature of several steps minimizes material transfer losses and enhances throughput efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-n-butyl-4-chloro-5-formylimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of pharmaceutical manufacturing. Our team of expert chemists has thoroughly analyzed the potential of Patent CN111592495A and is prepared to assist partners in scaling this technology from laboratory benchtop to full commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this oxidative method are fully realized in an industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-n-butyl-4-chloro-5-formylimidazole meets the exacting standards required for Losartan synthesis.
We invite forward-thinking pharmaceutical companies and contract manufacturers to collaborate with us on optimizing this supply chain. By leveraging our technical expertise, you can secure a stable source of high-quality intermediates while simultaneously reducing your environmental impact and production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative chemistry can drive value for your organization.
