Advanced Synthesis of N-CBZ-L-Histidine: Enhancing Purity and Scalability for Global Peptide Manufacturing
The landscape of peptide synthesis relies heavily on the availability of high-quality protected amino acids, where N-carbobenzoxy-L-histidine serves as a critical building block for numerous therapeutic agents. A recent technological breakthrough detailed in patent CN112898204B introduces a refined synthesis method that addresses long-standing challenges in purity and process operability associated with histidine protection. This innovation is particularly significant for R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers who can deliver consistent quality at scale. By replacing traditional reagents with N-benzyloxycarbonyl-succinimide and utilizing ammonia gas for selective deprotection, the process eliminates the formation of viscous intermediates and minimizes the risk of racemization. This report analyzes the technical merits of this approach, demonstrating how it aligns with the industry's demand for cost reduction in API manufacturing and enhanced supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-carbobenzoxy-L-histidine has been plagued by significant technical hurdles when employing benzyl chloroformate as the primary protecting reagent. The conventional one-step protection method often results in the inevitable generation of double-CBZ byproducts and other impurities, leading to difficult purification processes and suboptimal yields. Furthermore, the reaction mixture tends to become extremely viscous, creating a sticky intermediate that hinders uniform stirring and causes impurity encapsulation which cannot be easily washed away. Even rigorous recrystallization efforts often fail to meet the stringent purity standards required for polypeptide synthesis. Additionally, the subsequent removal of the imidazolyl protecting group using strong bases like potassium hydroxide or sodium ethoxide poses a severe risk of damaging the optical rotation of the product due to partial deprotection of the alpha-amino group.
The Novel Approach
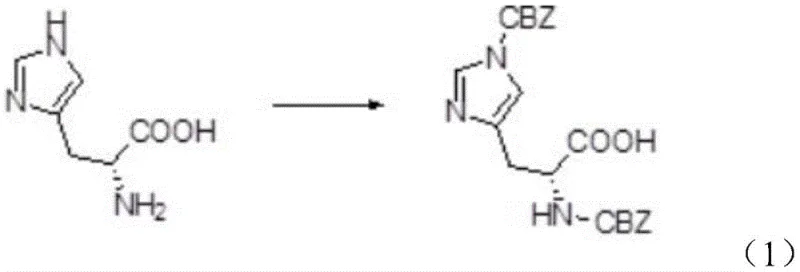
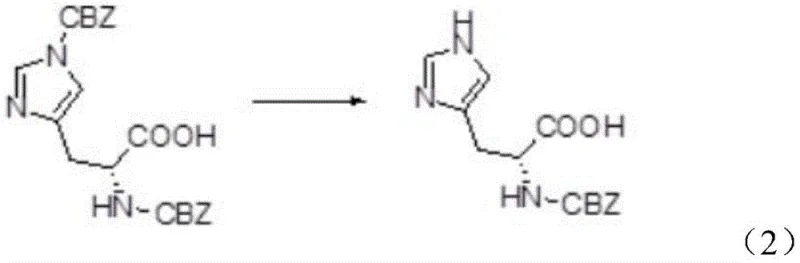
The novel methodology presented in the patent data offers a transformative solution by utilizing N-benzyloxycarbonyl-succinimide (Z-OSu) to replace benzyl chloroformate in the initial protection step. This substitution fundamentally alters the physical properties of the reaction intermediate, preventing the formation of the problematic sticky mass and ensuring a cleaner reaction profile with fewer unnecessary byproducts. Consequently, the intermediate bis-benzyloxycarbonyl-L-histidine possesses such high quality that it can proceed directly to the second reaction step without intermediate purification, drastically simplifying the workflow. In the deprotection phase, the creative adoption of ammonia gas replaces harsh alkaline conditions, providing a mild environment that selectively removes the imidazolyl carbobenzoxy group while preserving the alpha-amino protection. This strategic shift not only improves the overall yield but also guarantees the preservation of the product's optical activity, making it ideal for high-purity amino acid derivatives production.
Mechanistic Insights into Selective Deprotection and Catalysis
The core of this synthesis lies in the precise control of chemoselectivity during both the protection and deprotection phases. In the first step, the reaction between L-histidine and N-benzyloxycarbonyl-succinimide is facilitated by an organic base and a phase transfer catalyst, such as a quaternary ammonium salt. The catalyst plays a pivotal role in enhancing the solubility of the reagents and promoting the double CBZ protection on both the alpha-amino and imidazolyl groups efficiently. The use of specific molar ratios, typically ranging from 1:2 to 1:3 for the histidine to Z-OSu, ensures complete conversion while minimizing waste. The organic base, whether triethylamine or DIPEA, acts not only as a proton scavenger but also aids in dissolving the succinimide derivative, creating a homogeneous reaction environment that prevents local hotspots which could lead to side reactions.
In the second step, the mechanism shifts to a selective nucleophilic attack by ammonia on the carbobenzoxy group attached to the imidazole ring. Unlike strong alkalis which indiscriminately attack ester and carbamate bonds leading to racemization, ammonia gas operates under mild thermal conditions, typically between 10°C and 40°C. This selectivity is crucial because the imidazolyl CBZ group is slightly more labile than the alpha-amino CBZ group under these specific ammonolysis conditions. The reaction proceeds to cleave the imidazole protection, releasing benzyl carbamate as a stable byproduct which precipitates out or can be recovered from the filtrate. This mechanistic pathway ensures that the chiral center at the alpha-carbon remains untouched, preserving the specific optical rotation required for biological activity in the final peptide drug substance.
How to Synthesize N-CBZ-L-Histidine Efficiently
Implementing this synthesis route requires careful attention to reagent addition rates and temperature control to maximize the benefits of the novel chemistry. The process begins with the dispersion of the succinimide reagent in a solvent like dichloromethane, followed by the sequential addition of the base and catalyst before introducing the histidine. Maintaining the reaction temperature during the exothermic addition of Z-OSu is critical to prevent degradation. Once the bis-protected intermediate is formed, the workup involves a simple acid wash to remove excess base, avoiding complex isolation steps. The subsequent introduction of ammonia gas must be controlled to ensure saturation without excessive pressure buildup, allowing the selective deprotection to proceed to completion. For a detailed breakdown of the standardized operating procedures and safety protocols, please refer to the technical guide below.
- React L-histidine with N-benzyloxycarbonyl-succinimide (Z-OSu) in the presence of an organic base and phase transfer catalyst to form bis-benzyloxycarbonyl-L-histidine.
- Adjust pH and wash the reaction mixture to remove residual bases without isolating the intermediate.
- Introduce ammonia gas into the reaction solution to selectively remove the imidazolyl carbobenzoxy group, yielding high-purity N-CBZ-L-histidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthesis method represents a significant opportunity for cost reduction in API manufacturing and operational streamlining. The elimination of the intermediate purification step reduces solvent consumption and labor hours, directly impacting the cost of goods sold. Furthermore, the ability to recover high-purity benzyl carbamate as a byproduct adds a layer of economic efficiency, as this material can be recycled or sold, offsetting raw material costs. The robustness of the process against viscosity issues means that reactor turnover times are improved, enhancing the overall throughput of the manufacturing facility without requiring capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The replacement of benzyl chloroformate with N-benzyloxycarbonyl-succinimide eliminates the need for extensive purification of sticky intermediates, which traditionally consumes large volumes of solvents and energy. By enabling a telescoped process where the intermediate is not isolated, the manufacturing cycle time is significantly shortened, leading to substantial cost savings in utilities and labor. Additionally, the recovery of benzyl carbamate byproduct provides an internal value stream that further optimizes the economic balance of the production run.
- Enhanced Supply Chain Reliability: The use of milder reaction conditions and less hazardous reagents improves the safety profile of the manufacturing process, reducing the risk of unplanned shutdowns due to safety incidents. The high quality of the intermediate ensures consistent batch-to-batch performance, minimizing the variability that often disrupts supply schedules. This reliability allows for more accurate forecasting and inventory management, ensuring that downstream peptide synthesis operations receive their critical raw materials on time.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the handling of viscous masses that are difficult to manage in large-scale reactors. The simplified workup procedure reduces the volume of wastewater generated, aligning with stricter environmental regulations and lowering waste disposal costs. The ability to operate at near-ambient temperatures during the deprotection step also reduces the energy footprint of the facility, supporting sustainability goals while maintaining high production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived directly from the patent specifications and experimental data to provide clarity on process feasibility. Understanding these details is essential for technical teams evaluating the integration of this method into their existing production lines.
Q: Why is ammonia gas preferred over potassium hydroxide for deprotection in this synthesis?
A: Ammonia gas provides mild reaction conditions that selectively remove the imidazolyl protecting group without affecting the alpha-amino CBZ group, thereby preventing racemization and preserving optical rotation.
Q: What are the advantages of using N-benzyloxycarbonyl-succinimide over benzyl chloroformate?
A: Using N-benzyloxycarbonyl-succinimide avoids the formation of sticky intermediates common with benzyl chloroformate, significantly simplifying purification and improving overall product quality.
Q: Can the byproducts from this synthesis be recovered?
A: Yes, the process generates benzyl carbamate as a high-purity byproduct during the deprotection step, which can be recovered and reused, contributing to cost efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-CBZ-L-Histidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity protected amino acids play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like the one described in CN112898204B are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-CBZ-L-histidine meets the exacting standards required for pharmaceutical applications. Our commitment to quality ensures that your peptide synthesis projects proceed without interruption due to raw material inconsistencies.
We invite you to collaborate with us to leverage these technological advancements for your specific supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value for your organization.
