Advanced Manufacturing of Gliquidone Intermediates: A Safer, High-Yield Solvent-Based Cyclization Route
Introduction to Patent CN111825614B
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for critical antidiabetic agents like gliquidone. Patent CN111825614B introduces a transformative preparation method for the key gliquidone intermediate, specifically 4-[2-(3,4-dihydro-7-methoxy-1,3-dioxo-2(1H)-isoquinolinyl)ethyl]benzenesulfonamide. This innovation addresses the longstanding challenges associated with high-temperature solvent-free reactions and multi-step acid chloride protocols. By utilizing a sealed hydrophilic solvent system, the process achieves superior conversion rates while significantly mitigating the risks of equipment blockage and hazardous gas evolution. For global supply chain leaders, this technology represents a pivotal shift towards more sustainable and scalable manufacturing practices, ensuring a reliable supply of high-quality intermediates for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this isoquinoline derivative has relied on two primary methodologies, both of which present significant industrial drawbacks. The first approach, documented by R.A. Hill et al., involves a solvent-free melt reaction at extreme temperatures of 200°C. While this method avoids solvent usage, it necessitates specialized high-temperature circulation equipment and poses severe safety hazards due to the thermal stress on reactor materials. Furthermore, the reaction mixture tends to solidify immediately upon cooling below the melting point, causing catastrophic blockages in valves and pipelines, which complicates material transfer and increases downtime. The second conventional route involves a two-step activation using thionyl chloride to form an acid chloride intermediate, followed by amidation with triethylamine. This pathway generates substantial amounts of corrosive hydrogen chloride gas and volatile amine odors, creating complex waste gas treatment requirements and increasing the environmental footprint of the manufacturing facility.
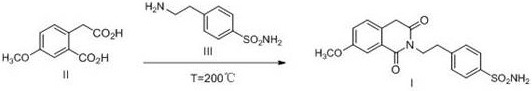
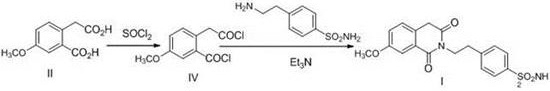
The Novel Approach
In stark contrast to these legacy methods, the novel process described in CN111825614B employs a one-pot cyclization strategy within a sealed reactor using hydrophilic solvents. By maintaining the reaction temperature between 150°C and 170°C, the system operates well below the dangerous thresholds of the solvent-free method while ensuring sufficient energy for cyclization. The presence of a polar solvent, such as water or lower alcohols, facilitates the initial formation of soluble salts between the reactants, ensuring a homogeneous reaction phase that prevents localized overheating. Upon completion, the product precipitates uniformly as the system cools, allowing for straightforward centrifugation or filtration without the risk of pipeline clogging. This streamlined approach not only simplifies the operational workflow but also enhances the overall safety profile by eliminating the need for corrosive activating agents and reducing the thermal load on production equipment.
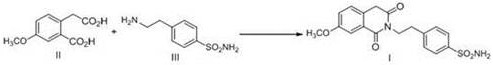
Mechanistic Insights into Sealed Solvent Cyclization
The core chemical mechanism driving this improved synthesis relies on the unique solvation properties of hydrophilic media under elevated pressure. When Compound II (the dicarboxylic acid derivative) and Compound III (the sulfonamide amine) are introduced into a polar solvent, they initially undergo a salification reaction. This salt formation is crucial because it dramatically increases the solubility of the reactants, creating a uniform solution that overcomes the mass transfer limitations often seen in heterogeneous melt reactions. As the temperature is raised to the optimal range of 150-170°C within the sealed vessel, the system undergoes intramolecular dehydration. The polar environment stabilizes the transition state, facilitating the nucleophilic attack of the amine nitrogen onto the carbonyl carbon, ultimately closing the six-membered imide ring to form the target isoquinoline structure. This mechanism ensures that the reaction proceeds to near-completion with minimal side products, as the solvent effectively suppresses polymerization or degradation pathways that might occur at higher, uncontrolled temperatures.
From an impurity control perspective, the choice of solvent plays a dual role as both a reaction medium and a purification agent. In traditional solvent-free processes, impurities are often trapped within the solidified melt, requiring energy-intensive recrystallization steps to achieve pharmaceutical-grade purity. However, in this novel sealed system, the solvent remains present throughout the cooling phase. As the temperature drops to room temperature, the solubility of the target intermediate decreases sharply, causing it to crystallize out of the solution. Crucially, most organic impurities and unreacted starting materials remain dissolved in the mother liquor due to their differing solubility profiles. This in-situ purification effect means that a simple filtration and washing step is sufficient to yield a product with purity levels exceeding 99%, drastically reducing the need for downstream chromatographic purification or multiple recrystallizations.
How to Synthesize Gliquidone Intermediate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins by charging the dicarboxylic acid and the sulfonamide amine into a pressure-rated reactor, followed by the addition of a hydrophilic solvent in an amount 5 to 10 times the mass of the acid component. The system is then sealed to prevent solvent loss and heated to a target temperature of 160°C, which has been identified as the optimal balance between reaction rate and selectivity. Monitoring via HPLC is recommended to confirm that the residual starting material falls below 1% before proceeding to the workup phase. Once the reaction is complete, the mixture is cooled gradually to ambient temperature to induce crystallization, followed by filtration and drying at 60°C. For a detailed breakdown of the specific operational parameters and safety checks required for this procedure, please refer to the standardized guide below.
- Charge Compound II and Compound III into a reaction kettle with a hydrophilic solvent (5-10x mass of Compound II) and seal the system.
- Heat the sealed mixture to 150-170°C to facilitate intramolecular dehydration and cyclization, monitoring until raw material conversion is complete.
- Cool the reaction mixture to room temperature, filter the precipitated solid, wash with solvent, and dry to obtain the pure intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound strategic advantages beyond mere technical feasibility. The elimination of thionyl chloride and triethylamine removes the need for specialized corrosion-resistant equipment and complex scrubbing systems for acidic and amine waste gases, leading to a significant reduction in capital expenditure and ongoing maintenance costs. Furthermore, the transition from a two-step activation process to a direct one-pot cyclization inherently shortens the production cycle time, thereby increasing the throughput capacity of existing manufacturing assets without the need for additional reactors. The ability to use common, recyclable solvents like water or ethanol further decouples the process from the volatility of specialized reagent markets, ensuring greater stability in raw material pricing and availability.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the reduction of auxiliary material consumption. By omitting the acid chloride activation step, manufacturers save on the direct costs of thionyl chloride and the base, while also avoiding the losses associated with multi-step transfers. Additionally, the high purity achieved directly from filtration minimizes the solvent and energy consumption typically required for extensive recrystallization procedures. The potential for solvent recovery and reuse in a closed-loop system further amplifies these savings, creating a leaner cost structure that enhances competitiveness in the global API market.
- Enhanced Supply Chain Reliability: Operational reliability is significantly improved by mitigating the risks of equipment failure and batch inconsistency. The lower operating temperature reduces thermal stress on reactor linings and seals, extending the lifespan of critical production assets and reducing unplanned downtime. Moreover, the avoidance of solidification issues ensures smooth material flow throughout the plant, preventing the bottlenecks that often plague solvent-free high-temperature processes. This operational stability translates into more predictable lead times and a consistent ability to meet delivery schedules, which is critical for maintaining uninterrupted supply chains for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the use of standard pressure reactors and benign solvents. The absence of hazardous gas evolution simplifies the environmental permitting process and reduces the burden on waste treatment facilities. The process aligns well with green chemistry principles by improving atom economy and reducing the E-factor through efficient solvent usage. This environmental compatibility not only future-proofs the manufacturing site against tightening regulations but also appeals to increasingly eco-conscious stakeholders and partners in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: Why is a sealed solvent system preferred over solvent-free melting for this intermediate?
A: Solvent-free methods require temperatures above 200°C to maintain fluidity, leading to safety risks and equipment blockage upon cooling. The sealed solvent system allows reaction at lower temperatures (150-170°C) and ensures the product remains dispersed for easy filtration.
Q: What hydrophilic solvents are compatible with this cyclization process?
A: The patent specifies water, methanol, ethanol, n-propanol, isopropanol, butanol, acetonitrile, dioxane, and tetrahydrofuran. Water and lower alcohols are particularly effective due to their polarity and ability to form soluble salt intermediates.
Q: How does this method improve purity compared to traditional acid chloride routes?
A: By avoiding thionyl chloride and triethylamine, this method eliminates corrosive byproducts and difficult-to-remove amine salts. The solvent acts as a recrystallization medium during cooling, yielding products with >99% purity directly after filtration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gliquidone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of gliquidone intermediate meets the highest quality standards required for pharmaceutical applications. Our commitment to technical excellence ensures that we can navigate the complexities of sealed reactor operations with precision and safety.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how this optimized synthesis method can enhance your operational efficiency and reduce total landed costs. Let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the antidiabetic therapeutic sector.
