Advanced Manufacturing of S-Pantoprazole Sodium: A Technical Breakthrough for Global Supply Chains
The pharmaceutical landscape for proton pump inhibitors (PPIs) continues to evolve, driven by the demand for higher efficacy and reduced metabolic variability. Patent CN103694222A introduces a transformative preparation method for S-pantoprazole sodium, the active S-enantiomer of pantoprazole, which offers superior bioavailability and pharmacokinetic profiles compared to its racemic counterpart. This technical disclosure outlines a robust three-step synthetic route that bypasses the limitations of traditional chiral separation or low-selectivity oxidation methods. By leveraging sodium bismuthate as a novel oxidizing agent in conjunction with a specific chiral bipyridine methyl amide ligand, the process achieves exceptional stereocontrol. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for high-value gastrointestinal therapeutics. The methodology not only simplifies operational complexity but also ensures the production of intermediates with chemical and optical purities exceeding 99.8%, addressing the stringent quality requirements of modern regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of S-pantoprazole has been plagued by inefficiencies inherent in racemic resolution or non-selective oxidation pathways. Traditional methods often rely on chiral separation techniques, such as preparative HPLC or enzymatic resolution, which theoretically cap the maximum yield at 50% for the desired enantiomer, leading to substantial material waste and inflated cost of goods sold (COGS). Furthermore, earlier asymmetric oxidation protocols frequently utilized vanadium-based catalysts, which pose significant environmental and safety challenges due to heavy metal toxicity and the complex downstream processing required to reduce residual metal levels to ppm specifications. These legacy processes often suffer from inconsistent enantioselectivity, requiring multiple recrystallization steps that further erode overall yield and extend production lead times. The reliance on harsh reaction conditions and expensive chiral auxiliaries in older patents has created a bottleneck for manufacturers seeking to scale production efficiently while maintaining compliance with increasingly rigorous impurity profiles.
The Novel Approach
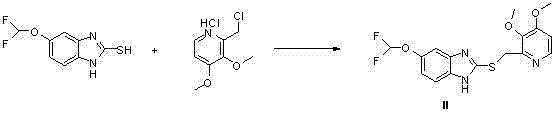
The methodology described in CN103694222A fundamentally reengineers the synthetic pathway by introducing a highly selective catalytic system centered on sodium bismuthate. This approach initiates with the condensation of 5-difluoro methoxy-2-mercapto-1H-benzimidazole and 2-chloromethyl-3,4-dimethoxy pyridine hydrochloride, establishing the core sulfide scaffold under mild alkaline conditions. Unlike conventional routes that struggle with over-oxidation to the sulfone or poor chiral induction, this novel protocol employs a titanium-chiral ligand complex that creates a sterically defined environment for oxygen transfer. The result is a direct, high-yielding transformation to the S-sulfoxide with minimal formation of the R-enantiomer or sulfone byproducts. This streamlined process eliminates the need for wasteful resolution steps and hazardous heavy metal catalysts, offering a cleaner, more sustainable, and economically viable alternative for the commercial manufacture of levpantoprazole intermediates.
Mechanistic Insights into Sodium Bismuthate Catalyzed Asymmetric Oxidation
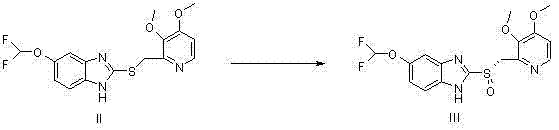
The core innovation of this patent lies in the mechanistic interplay between the titanium alkoxide, the chiral bipyridine methyl amide ligand, and the sodium bismuthate oxidant. In this catalytic cycle, the titanium center acts as a Lewis acid, coordinating with the chiral diamide ligand to form a rigid, chiral complex. This complex then activates the sulfide substrate, positioning it within a specific chiral pocket that dictates the trajectory of the incoming oxygen atom from the bismuthate species. The steric bulk and electronic properties of the N,N'-bis-(3-pyridylmethyl)-D(-)-tartrate diamide ligand are critical, as they effectively shield one face of the sulfur atom, ensuring that oxidation occurs exclusively from the desired direction to yield the S-configuration. This precise control minimizes the formation of the undesired R-enantiomer and prevents over-oxidation to the achiral sulfone, a common side reaction in non-catalyzed or poorly catalyzed systems. The use of sodium bismuthate is particularly advantageous as it serves as a stoichiometric oxygen donor that is compatible with the chiral titanium complex, maintaining high turnover and selectivity without degrading the ligand framework.
From an impurity control perspective, this mechanism offers distinct advantages for process chemistry teams. The high specificity of the chiral catalyst means that the crude reaction mixture contains significantly lower levels of stereoisomeric impurities compared to vanadium-catalyzed methods. This reduces the burden on downstream purification units, allowing for simpler crystallization protocols to achieve final purity specifications. Furthermore, the mild reaction temperature range of 50-60°C prevents thermal degradation of the sensitive benzimidazole and pyridine moieties, which can otherwise lead to complex decomposition byproducts that are difficult to purge. The compatibility of the reagents with common organic solvents like toluene and acetone facilitates efficient workup procedures, such as filtration and solvent exchange, ensuring that the final API intermediate meets the stringent residual solvent and heavy metal limits required for pharmaceutical registration. This robust mechanistic foundation provides a reliable platform for scaling up production while maintaining consistent quality attributes batch after batch.
How to Synthesize S-Pantoprazole Sodium Efficiently
The synthesis of S-pantoprazole sodium via this patented route is designed for operational simplicity and scalability, making it an ideal candidate for technology transfer. The process begins with the nucleophilic substitution of the mercapto-benzimidazole, followed by the critical asymmetric oxidation step which defines the stereochemistry of the final product. The final salification step converts the free acid sulfoxide into the stable sodium salt form required for formulation. Each stage has been optimized to maximize yield and minimize waste, utilizing readily available reagents and standard reactor configurations. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and crystallization parameters, are outlined in the guide below to assist process engineers in replicating these results.
- Condense 5-difluoro methoxy-2-mercapto-1H-benzimidazole with 2-chloromethyl-3,4-dimethoxy pyridine hydrochloride in the presence of sodium hydroxide to form the sulfide intermediate.
- Perform catalytic asymmetric oxidation of the sulfide intermediate using sodium bismuthate as the oxidant and a chiral bipyridine methyl amide ligand with titanium alkoxide.
- React the resulting S-pantoprazole sulfoxide with sodium hydroxide to form the final S-pantoprazole sodium salt through crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling strategic benefits that extend beyond simple technical metrics. The elimination of expensive and toxic transition metal catalysts, such as vanadium, directly translates to a reduction in raw material costs and waste disposal expenses. Additionally, the high yield and selectivity of the process mean that less starting material is required to produce the same amount of active pharmaceutical ingredient, effectively lowering the cost per kilogram of the final output. The use of mild reaction conditions reduces energy consumption and lowers the safety risks associated with high-pressure or high-temperature operations, contributing to a more resilient and sustainable manufacturing footprint. These factors combined create a supply chain that is not only more cost-effective but also less vulnerable to regulatory scrutiny regarding environmental impact and worker safety.
- Cost Reduction in Manufacturing: The replacement of traditional chiral separation methods with direct asymmetric synthesis drastically improves atom economy, effectively doubling the theoretical yield compared to resolution techniques. By avoiding the use of precious metal catalysts and reducing the number of purification steps, the overall cost of goods is significantly lowered. The high efficiency of the sodium bismuthate oxidation system minimizes the loss of valuable intermediates, ensuring that the production process remains economically competitive even at large scales. Furthermore, the simplified workup procedures reduce solvent consumption and processing time, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents, such as sodium bismuthate and common organic solvents, mitigates the risk of supply disruptions often associated with specialized or proprietary catalysts. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in temperature or mixing rates. This reliability ensures consistent delivery timelines for downstream API manufacturers, reducing the need for excessive safety stock and enabling a more lean inventory management strategy. The ability to source raw materials from multiple suppliers further strengthens the supply chain against geopolitical or market volatility.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-kilogram batches with consistent results, paving the way for ton-scale commercial production. The absence of heavy metal contaminants simplifies the environmental compliance process, as wastewater treatment is less complex and costly. The use of greener oxidants and the reduction of hazardous waste generation align with global sustainability goals, enhancing the corporate social responsibility profile of the manufacturing entity. This environmental compatibility facilitates faster regulatory approvals in markets with strict ecological standards, accelerating time-to-market for new generic or branded formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this S-pantoprazole sodium synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for stakeholders evaluating the potential for licensing or adopting this process within their existing manufacturing portfolios.
Q: What are the advantages of using sodium bismuthate over traditional vanadium catalysts?
A: Sodium bismuthate offers superior chiral selectivity and avoids the toxicity and difficult removal processes associated with heavy metal catalysts like vanadium, leading to higher purity and safer processing.
Q: What optical purity can be achieved with this novel method?
A: The process consistently achieves an enantiomeric excess (ee) value greater than 99.8%, ensuring high-quality API intermediates suitable for strict regulatory standards.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction temperatures (20-60°C) and common solvents, making it highly scalable and cost-effective for commercial manufacturing without requiring extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Pantoprazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering S-pantoprazole sodium and related intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to implement advanced chiral synthesis technologies, such as the sodium bismuthate oxidation method, positions us as a strategic partner for pharmaceutical companies seeking to optimize their supply chains and reduce costs without compromising quality.
We invite you to collaborate with us to explore how this innovative synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this superior manufacturing process. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a reliable, high-performance supply of S-pantoprazole sodium for your global operations.
