Advanced Ionic Liquid Catalysis for Commercial Scale-up of 2-Amino-5,6-Dichlorobenzothiazole
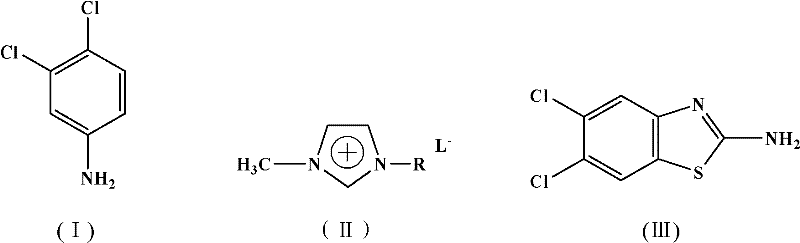
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for critical heterocyclic intermediates, particularly those serving as scaffolds for next-generation therapeutics. Patent CN101857579B introduces a significant technological advancement in the production of 2-amino-5,6-dichlorobenzothiazole, a vital building block for antifungal, antitumor, and anti-infective agents such as the HQ-495 class of medicines. This patent discloses a novel one-pot synthesis method that leverages the unique properties of acidic ionic liquids to replace traditional, environmentally burdensome solvents and catalysts. By integrating the reaction medium and catalytic function into a single reusable component, this technology addresses the growing demand for green chemistry solutions without compromising on yield or product quality. For R&D directors and procurement specialists, understanding this shift from conventional bromination techniques to ionic liquid-mediated cyclization is crucial for securing a sustainable supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiazole derivatives like 2-amino-5,6-dichlorobenzothiazole has relied heavily on traditional thiocyanation and cyclization protocols that present significant operational and environmental challenges. Conventional routes often utilize elemental bromine or harsh mineral acids, which are highly corrosive and pose severe safety risks during handling and storage. These methods typically generate substantial quantities of spent acid and hazardous waste streams, necessitating complex and costly downstream treatment processes to meet environmental compliance standards. Furthermore, the separation of the final product from the reaction mixture in traditional systems can be inefficient, often leading to product loss or the retention of difficult-to-remove impurities. The reliance on stoichiometric amounts of hazardous reagents also drives up the raw material costs and complicates the supply chain due to strict regulatory controls on such chemicals.
The Novel Approach
In stark contrast, the methodology described in patent CN101857579B utilizes a sophisticated acidic ionic liquid system, specifically designed imidazolium-based salts, to facilitate the transformation of 3,4-dichloroaniline into the target benzothiazole. This approach fundamentally alters the reaction landscape by employing the ionic liquid as a dual-function medium that acts simultaneously as the solvent and the acid catalyst. The process operates effectively within a moderate temperature range of 80°C to 110°C, avoiding the extreme conditions often required by older methods. A key feature of this novel approach is the recyclability of the ionic liquid; after the product is extracted, the remaining ionic phase can be recovered and reused, drastically minimizing waste generation. This one-pot strategy not only simplifies the operational workflow but also enhances the overall atom economy of the synthesis, making it a superior choice for modern, eco-conscious manufacturing facilities.
Mechanistic Insights into Acidic Ionic Liquid-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the specific interaction between the acidic ionic liquid and the reactants, which facilitates a smooth cyclization pathway. The ionic liquid, characterized by an organic cation such as an imidazolium ring substituted with sulfonic acid groups and an inorganic or organic anion, provides a highly polar and acidic environment. This environment promotes the activation of 3,4-dichloroaniline and ammonium thiocyanate, enabling the formation of the intermediate thiourea species which subsequently undergoes oxidative cyclization in the presence of N-bromosuccinimide (NBS). The acidic protons or Lewis acidic sites within the ionic liquid structure stabilize the transition states, lowering the activation energy required for the ring closure. This mechanistic efficiency ensures that the reaction proceeds rapidly and selectively towards the desired 2-amino-5,6-dichlorobenzothiazole structure, minimizing the formation of side products that typically plague less controlled acidic environments.
Furthermore, the impurity profile of the final product is significantly improved due to the homogeneous nature of the catalysis and the specific solvation properties of the ionic liquid. Unlike heterogeneous catalysts that may suffer from mass transfer limitations or active site poisoning, the ionic liquid ensures uniform contact between all reactant molecules. This uniformity prevents localized hotspots of high acidity that could lead to over-halogenation or decomposition of the sensitive amino group. The result is a crude product with high inherent purity, often exceeding 98% content as demonstrated in the patent examples, which reduces the burden on subsequent purification steps such as recrystallization or column chromatography. For quality control teams, this translates to a more consistent and reliable specification sheet for every batch produced.
How to Synthesize 2-Amino-5,6-Dichlorobenzothiazole Efficiently
The practical implementation of this synthesis involves a straightforward charging sequence where 3,4-dichloroaniline, ammonium thiocyanate, and N-bromosuccinimide are introduced directly into the acidic ionic liquid reactor. The molar ratios are carefully balanced, typically ranging from 1:1 to 1:1.5 for the reactants relative to the aniline, ensuring complete conversion while minimizing excess reagent waste. The reaction progress is conveniently monitored using thin-layer chromatography (TLC) with a hexane-acetone solvent system, allowing operators to determine the precise endpoint when the starting material spot disappears. Following the reaction, a simple extraction protocol using toluene separates the organic product from the ionic liquid phase, which remains in the aqueous raffinate for recovery. The detailed standardized synthesis steps, including specific workup procedures and recycling protocols, are outlined in the guide below.
- Charge 3,4-dichloroaniline, ammonium thiocyanate, and N-bromosuccinimide into an acidic ionic liquid reactor.
- Maintain the reaction mixture at a temperature between 80°C and 110°C until TLC monitoring indicates complete consumption of the starting material.
- Perform post-treatment by extracting the reaction liquid with toluene, washing with water, and recovering the reusable ionic liquid from the raffinate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-based technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of volatile organic solvents and the reduction of hazardous waste streams directly correlate to lower operational expenditures related to waste disposal and environmental compliance. Additionally, the ability to recycle the expensive ionic liquid catalyst multiple times creates a closed-loop system that insulates the manufacturing process from fluctuations in raw material pricing. This stability is crucial for long-term supply contracts where cost predictability is a primary requirement for multinational pharmaceutical clients seeking reliable partners.
- Cost Reduction in Manufacturing: The dual functionality of the ionic liquid eliminates the need to purchase separate solvents and acid catalysts, thereby consolidating raw material costs into a single, recoverable input. By removing the necessity for complex neutralization and waste treatment steps associated with spent mineral acids, the overall processing time and utility consumption are significantly decreased. This streamlined workflow allows for a substantial reduction in the cost of goods sold (COGS), providing a competitive pricing advantage in the global market for fine chemical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: The simplicity of the one-pot operation reduces the risk of batch failures caused by multi-step transfer errors or equipment contamination. Since the ionic liquid is non-volatile and thermally stable, storage and handling requirements are less stringent compared to corrosive liquid acids or gaseous reagents, ensuring a safer and more continuous production schedule. This robustness guarantees consistent delivery timelines for downstream customers who rely on just-in-time inventory models for their API manufacturing lines.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions tighten globally, this green synthesis route positions manufacturers favorably for future audits and inspections. The drastic reduction in "three wastes" (waste gas, waste water, and solid residue) simplifies the permitting process for capacity expansion. Consequently, scaling this process from pilot plant quantities to multi-ton commercial production can be achieved with minimal additional investment in pollution control infrastructure, facilitating rapid response to surges in market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-amino-5,6-dichlorobenzothiazole using this patented ionic liquid method. These insights are derived directly from the experimental data and beneficial effects described in the source intellectual property, providing clarity on performance metrics and operational feasibility. Understanding these details helps stakeholders evaluate the suitability of this technology for their specific supply chain requirements.
Q: What are the primary advantages of using acidic ionic liquids in this synthesis?
A: The acidic ionic liquid serves a dual function as both the reaction medium and the catalyst, eliminating the need for additional volatile organic solvents and separate acid catalysts. Furthermore, the ionic liquid can be recovered and reused multiple times, significantly reducing waste generation and operational costs compared to traditional methods.
Q: What purity levels can be achieved with this patented method?
A: According to the experimental data in patent CN101857579B, the synthesized 2-amino-5,6-dichlorobenzothiazole consistently achieves a content purity of greater than 98%, with melting points aligning closely with literature values (178-180°C).
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the one-pot nature of the reaction simplifies operation and equipment requirements. The ability to recycle the ionic liquid catalyst and the reduction in hazardous 'three wastes' make this method highly scalable and compliant with modern environmental regulations for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5,6-Dichlorobenzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications and advanced agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every shipment of 2-amino-5,6-dichlorobenzothiazole meets the exacting standards required by top-tier pharmaceutical companies worldwide.
We invite you to collaborate with us to leverage this advanced green synthesis technology for your upcoming projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us become your strategic partner in chemical innovation.
