Advanced Vanadium-Catalyzed Synthesis of Dextro-Rabeprazole for Commercial Scale-Up
Advanced Vanadium-Catalyzed Synthesis of Dextro-Rabeprazole for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies for the synthesis of chiral proton pump inhibitors, particularly Dextro-Rabeprazole, due to its superior pharmacokinetic profile compared to its racemic counterpart. Patent CN108623564B introduces a groundbreaking preparation method that leverages a vanadium metal center coordinated with a specific tetradentate organic ligand to achieve exceptional enantioselectivity. This innovation addresses the longstanding challenges associated with the asymmetric oxidation of prochiral thioethers, offering a pathway that combines high optical purity with operational simplicity. By shifting away from traditional titanium-based systems, this technology provides a distinct advantage in minimizing peroxide-related impurities, a critical factor for regulatory compliance in API manufacturing. The method demonstrates remarkable efficiency, achieving yields nearing 90% with enantiomeric excess values exceeding 99.6%, setting a new benchmark for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer sulfoxides like Dextro-Rabeprazole has relied heavily on chiral resolution techniques or early-generation asymmetric oxidation protocols. Chiral resolution, while conceptually straightforward, suffers from an inherent theoretical yield limit of 50%, necessitating the recycling or disposal of the unwanted enantiomer, which drastically inflates production costs and waste generation. Furthermore, traditional asymmetric oxidation methods, such as those utilizing titanium tetraisopropoxide and diethyl tartrate, often struggle with catalyst stability and the formation of stubborn peroxide impurities. These impurities not only complicate the purification process but can also pose safety risks during scale-up due to their potential instability. The requirement for large amounts of specialized bases and the sensitivity of titanium catalysts to moisture further exacerbate the operational complexity, making these conventional routes less attractive for modern, cost-sensitive supply chains.
The Novel Approach
The novel approach detailed in the patent data utilizes a vanadium acetylacetonate precursor complexed with a chiral tetradentate ligand, creating a highly active and selective catalytic species. This system operates effectively in common organic solvents like toluene at controlled low temperatures, specifically between -10°C and 0°C, which are easily achievable in standard industrial reactors. Unlike previous methods that might require cryogenic conditions or exotic reagents, this vanadium-catalyzed route maintains high conversion rates while suppressing the over-oxidation to sulfones. The use of cumene hydroperoxide as the oxidant in this specific coordination environment ensures that the oxygen transfer is highly directed, resulting in minimal byproduct formation. This streamlined process eliminates the need for complex resolution steps, thereby doubling the potential yield from the starting thioether and significantly reducing the overall material throughput required for a given batch size.
Mechanistic Insights into Vanadium-Catalyzed Asymmetric Oxidation
The core of this technological advancement lies in the precise coordination chemistry between the vanadium metal center and the chiral tetradentate ligand. The ligand, synthesized from salicylaldehyde and chiral dinaphthalene diamine, creates a rigid chiral pocket around the vanadium atom. This steric environment is crucial for differentiating the two faces of the prochiral sulfur atom in the thioether substrate. When the oxidant approaches the metal center, the bulky naphthalene groups of the ligand direct the attack to a specific trajectory, ensuring that the oxygen atom is transferred exclusively to form the desired R-configuration. This high degree of stereocontrol is what allows the process to achieve enantiomeric excess values greater than 99.6%, effectively rendering downstream chiral purification unnecessary. The stability of the vanadium-ligand complex also prevents the dissociation of the chiral inducer during the reaction, maintaining consistent selectivity throughout the entire batch cycle.
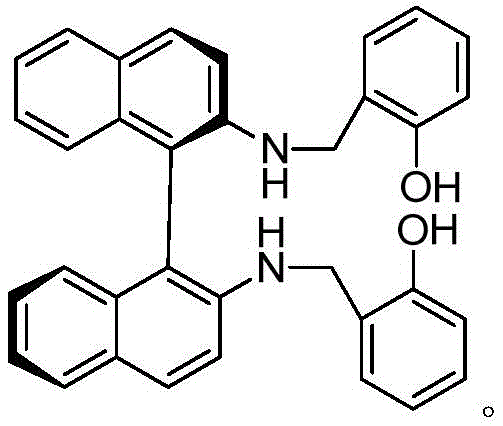
Another critical mechanistic advantage of this system is its ability to suppress the formation of sulfone impurities, which result from the over-oxidation of the desired sulfoxide product. In many oxidation reactions, once the sulfoxide is formed, it remains susceptible to further oxidation, leading to a mixture that is difficult to separate. However, the electronic properties of the vanadium complex in this specific formulation appear to deactivate towards the sulfoxide product once the initial oxidation is complete. This kinetic selectivity ensures that the concentration of the sulfone impurity remains exceptionally low, often below 0.1% as demonstrated in the experimental data. Controlling this specific impurity is vital because sulfone derivatives can be structurally similar to the API and difficult to remove via standard crystallization, potentially compromising the final drug quality.
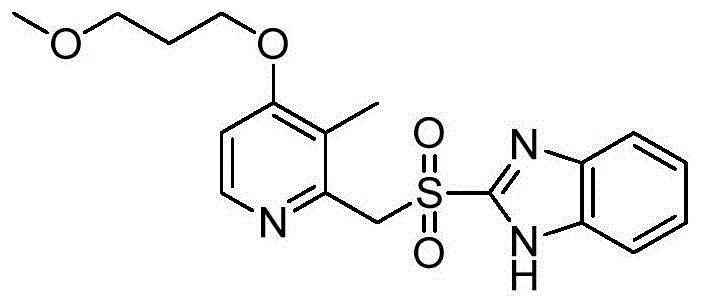
How to Synthesize Dextro-Rabeprazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-efficiency results in a laboratory or pilot plant setting. The process begins with the in situ generation of the active catalyst, followed by the controlled addition of the substrate and oxidant under strict thermal regulation. Maintaining the reaction temperature within the narrow window of -10°C to 0°C is paramount for maximizing enantioselectivity and minimizing side reactions. The workup procedure involves a simple quenching with sodium thiosulfate to neutralize excess oxidant, followed by crystallization steps that leverage the high purity of the crude reaction mixture. For a comprehensive understanding of the specific stoichiometric ratios, solvent choices, and timing required to replicate this success, please refer to the detailed standardized synthesis steps provided in the guide below.
- Prepare the catalyst by mixing vanadium acetylacetonate and the chiral tetradentate ligand in toluene at 20-30°C.
- Add the prochiral thioether intermediate and diisopropylethylamine, then cool the mixture to -10 to 0°C.
- Slowly add cumene hydroperoxide as the oxidant while maintaining low temperature to ensure high enantioselectivity and minimize sulfone impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this vanadium-catalyzed methodology represents a significant opportunity to optimize the cost structure and reliability of the Dextro-Rabeprazole supply chain. By transitioning from resolution-based methods to direct asymmetric synthesis, manufacturers can effectively double the yield from the same amount of starting material, which directly translates to substantial raw material cost savings. The elimination of the resolution step also removes the need for additional solvents and processing time associated with separating enantiomers, further driving down the cost of goods sold. Moreover, the high purity of the crude product reduces the burden on quality control laboratories and minimizes the risk of batch failures due to out-of-specification impurity profiles, ensuring a more predictable and stable supply of the intermediate.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the dramatic improvement in atom economy and yield efficiency. Traditional resolution methods inherently waste half of the produced material, whereas this asymmetric oxidation converts nearly all the starting thioether into the desired dextro-isomer. Additionally, the catalyst system uses relatively inexpensive vanadium salts compared to precious metal catalysts, and the ligand can be synthesized from readily available precursors. The reduction in purification steps, driven by the low levels of sulfone and unreacted thioether, means less solvent consumption and lower energy usage for distillation and drying, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of raw materials. The process operates at mild sub-zero temperatures that do not require specialized cryogenic equipment, making it adaptable to a wider range of manufacturing facilities globally. The reagents involved, such as cumene hydroperoxide and vanadium acetylacetonate, are commodity chemicals with stable supply lines, reducing the risk of bottlenecks associated with exotic or proprietary catalysts. This accessibility ensures that production schedules can be maintained consistently without the delays often caused by the sourcing of specialized chiral auxiliaries or the logistical complexities of recycling unwanted enantiomers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles by reducing waste generation. The high selectivity means fewer byproducts end up in the waste stream, lowering the costs and complexities associated with effluent treatment. The process is inherently scalable because the exothermic nature of the oxidation is managed effectively by the controlled addition of the oxidant at low temperatures, a parameter that is easily managed in large-scale reactors with standard cooling jackets. This controllability mitigates safety risks associated with runaway reactions, facilitating a smoother transition from pilot scale to commercial tonnage production while meeting stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this vanadium-catalyzed synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is essential for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. The answers reflect the specific advantages of the tetradentate ligand system and its impact on final product quality and manufacturing efficiency.
Q: Why is the Vanadium-Tetradentate Ligand system superior to traditional Titanium catalysts?
A: The Vanadium-based system described in CN108623564B offers superior stability and enantioselectivity compared to traditional Titanium-tartrate complexes. It significantly reduces the formation of difficult-to-remove peroxide impurities and operates effectively under milder conditions, simplifying the downstream purification process.
Q: How does this method control the formation of sulfone impurities?
A: By strictly controlling the reaction temperature between -10°C and 0°C and utilizing the specific steric environment of the tetradentate ligand, the method prevents over-oxidation. This ensures that the sulfone impurity levels remain below 0.1%, drastically reducing the burden on final crystallization steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids complex chiral resolution steps which typically cap yields at 50%, and instead utilizes a direct asymmetric oxidation with high conversion rates (>99%) and excellent ee values (>99%), making it highly efficient for multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dextro-Rabeprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the vanadium-catalyzed oxidation described in CN108623564B to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and purity observed in the lab are faithfully reproduced at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Dextro-Rabeprazole we supply meets the highest international standards for safety and efficacy. Our commitment to technological excellence allows us to offer partners a supply chain that is both resilient and cost-competitive.
We invite you to collaborate with us to leverage these manufacturing advancements for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this novel catalytic route. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your supply of this critical proton pump inhibitor intermediate is secure, compliant, and economically optimized for the long term.
