Advanced Asymmetric Oxidation for High-Purity Proton Pump Inhibitor Intermediates
The pharmaceutical industry's relentless pursuit of high-purity Proton Pump Inhibitors (PPIs) has long been constrained by the complexities of chiral synthesis. Patent CN102603714B introduces a transformative methodology for synthesizing sulfinyl compounds with a single optical configuration, addressing the critical need for efficient, scalable production of key API intermediates like Esomeprazole and Lansoprazole. This technology leverages a novel Chiral Titanium complex to drive asymmetric oxidation, effectively converting prochiral thioethers into their corresponding chiral sulfoxides with exceptional stereoselectivity. Unlike traditional resolution techniques that discard half of the material, this direct asymmetric approach maximizes atom economy and operational efficiency. For R&D directors and procurement specialists, understanding this shift from resolution to direct asymmetric synthesis is paramount for optimizing supply chains and reducing the cost of goods sold in the competitive gastroenterology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure sulfinyl compounds relied heavily on classical resolution techniques or early-generation asymmetric oxidation methods that suffered from significant drawbacks. Conventional resolution, as noted in prior art like CN101391993, necessitates the use of substantial quantities of expensive resolving agents to separate enantiomers, inherently capping the theoretical yield at 50% unless dynamic kinetic resolution is employed. Furthermore, these processes involve loaded down with trivial details purification procedures, including multiple recrystallizations that consume vast amounts of solvent and time. Even alternative asymmetric oxidation methods, such as those disclosed in WO2009066321, often utilize chiral ligands with insufficient stereoselectivity, resulting in low optical yields that mandate repeated purification cycles to meet pharmacopeial standards. These inefficiencies translate directly into higher manufacturing costs, increased environmental waste, and prolonged lead times, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to meet global demand.
The Novel Approach
The methodology described in CN102603714B represents a paradigm shift by employing a highly specific Chiral Titanium complex to facilitate asymmetric oxidation with superior control. By utilizing a ligand derived from 5-[(R)-2'-hydroxypropionyl]amino-1-hydroxyethyl pyrazole complexed with titanium(IV), the system creates a rigid chiral environment that directs the oxidant specifically to one face of the sulfur atom. This precision eliminates the need for downstream resolution, allowing for the direct isolation of the desired enantiomer in high purity. The process operates under mild conditions, typically between -20°C and 40°C, using common organic solvents like ethyl acetate or toluene, which simplifies solvent recovery and waste management. The result is a streamlined workflow that not only boosts overall yield to over 76% but also delivers enantiomeric excess values exceeding 99%, effectively removing the burden of extensive purification from the production schedule.
Mechanistic Insights into Chiral Titanium-Catalyzed Asymmetric Oxidation
The core of this technological breakthrough lies in the unique coordination chemistry of the Chiral Titanium complex. The ligand, synthesized from D-alpha-hydroxypropionic acid ethyl ester and 5-amino-1-(2-hydroxyethyl) pyrazole, acts as a bidentate chelator that locks the titanium center into a specific geometry. When this complex interacts with the prochiral thioether substrate, it forms a transient intermediate where the sulfur atom is positioned precisely for oxygen transfer from the oxidant, such as metachloroperbenzoic acid (mCPBA) or cumene hydroperoxide. The steric bulk of the ligand shields one face of the sulfur, ensuring that oxidation occurs exclusively from the exposed face, thereby generating the sulfinyl group with the desired absolute configuration. This mechanism avoids the formation of racemic mixtures at the source, fundamentally altering the impurity profile of the reaction crude.
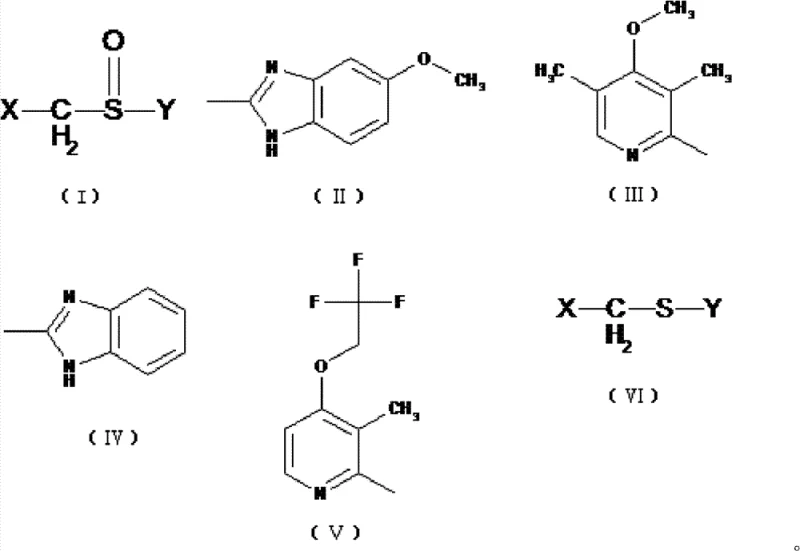
Furthermore, the presence of a catalytic amount of water plays a crucial role in the activation of the titanium species, facilitating the formation of the active oxidizing cluster. The reaction proceeds through a well-defined transition state that minimizes side reactions such as over-oxidation to sulfones, which are common impurities in sulfoxide synthesis. By controlling the stoichiometry of the oxidant (1.5 to 3 molar parts) and maintaining strict temperature protocols during the addition phase, the process ensures that the kinetic pathway favors the formation of the chiral sulfoxide. This level of mechanistic control is essential for R&D teams aiming to validate the robustness of the process for regulatory filings, as it demonstrates a clear understanding of Critical Process Parameters (CPPs) and their impact on Critical Quality Attributes (CQAs).
How to Synthesize Chiral Sulfinyl Compounds Efficiently
Implementing this synthesis route requires careful attention to the preparation of the chiral ligand and the sequential addition of reagents to maintain the integrity of the catalytic cycle. The process begins with the dissolution of the prochiral thioether in a suitable solvent, followed by the introduction of the pre-formed Chiral Titanium complex at low temperatures to prevent thermal degradation of the active species. Subsequent addition of the oxidant must be controlled to manage the exotherm and ensure high selectivity. The workup procedure involves a unique basification step using sodium hydroxide in acetone or 2-butanone, which facilitates the precipitation of the final product as a stable salt, such as Esomeprazole sodium. Detailed standardized synthetic steps see the guide below for precise operational parameters.
- Dissolve prochiral thioether in an organic solvent such as ethyl acetate or toluene, add a catalytic amount of water, and cool the solution to a temperature range of 10 to -20°C.
- Add the Chiral Titanium complex (formed from titanium isopropylate and a chiral pyrazole ligand), react briefly, then introduce the oxidant (mCPBA or cumene hydroperoxide) while maintaining low temperatures.
- Quench the reaction with aqueous ammonia, adjust pH to neutral with acetic acid, extract with dichloromethane, and crystallize the final product from acetone or 2-butanone after basification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric oxidation technology offers profound strategic advantages beyond mere technical elegance. By eliminating the resolution step, manufacturers can drastically reduce the consumption of raw materials, particularly the expensive resolving agents and the additional solvents required for their removal. This reduction in material intensity directly correlates to a significant decrease in the cost of goods sold, allowing for more competitive pricing strategies in the generic pharmaceutical market. Moreover, the simplified workflow reduces the number of unit operations, which in turn lowers energy consumption and labor costs associated with extended processing times. The ability to achieve high purity in fewer steps also means faster batch turnover, enhancing the overall agility of the supply chain to respond to market fluctuations.
- Cost Reduction in Manufacturing: The elimination of resolution agents and the reduction of purification cycles lead to substantial cost savings. Traditional methods often suffer from yield losses due to the inherent 50% limit of resolution or the losses incurred during repeated recrystallizations of low-selectivity oxidation products. In contrast, this direct asymmetric method achieves high yields (reported up to 76% and potentially higher upon optimization) and high optical purity in a single pass. This efficiency means that less starting material is required to produce the same amount of API intermediate, optimizing the utilization of capital and resources. Additionally, the use of commodity solvents like ethyl acetate and toluene, rather than specialized or hazardous solvents, further drives down operational expenditures and simplifies waste disposal logistics.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes significantly to supply continuity. Because the reaction tolerates a reasonable range of temperatures and uses stable reagents, the risk of batch failure due to minor process deviations is minimized. High enantiomeric excess values (above 99%) achieved directly from the reactor reduce the dependency on downstream polishing steps that can often become bottlenecks. This reliability ensures that delivery schedules for critical PPI intermediates can be met consistently, mitigating the risk of stockouts for downstream API manufacturers. The scalability of the method, demonstrated by its suitability for industrial production, assures partners that supply can be ramped up from pilot scale to multi-ton commercial production without fundamental changes to the chemistry.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns with green chemistry principles by improving atom economy and reducing waste generation. The avoidance of large quantities of resolving agents reduces the solid waste burden, while the streamlined process lowers the volume of wastewater and solvent emissions. This compliance is increasingly vital as global regulations on pharmaceutical manufacturing tighten. The process is designed for commercial scale-up of complex pharmaceutical intermediates, utilizing standard reactor equipment and straightforward workup procedures like filtration and crystallization. This ease of scale-up ensures that the technology can be transferred seamlessly from the laboratory to the plant, securing a long-term, sustainable supply of high-quality chiral building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What enantiomeric excess (ee) can be achieved with this titanium-catalyzed method?
A: According to patent CN102603714B, this method achieves exceptionally high stereoselectivity, with reported enantiomeric excess values reaching up to 99.8% for Esomeprazole sodium and 99.4% for Lansoprazole sodium without the need for repeated purification cycles.
Q: How does this process improve upon traditional resolution methods?
A: Traditional resolution requires large amounts of resolving agents and involves tedious purification steps that lower overall yield. This asymmetric oxidation method bypasses resolution entirely, utilizing a chiral catalyst to directly generate the desired single optical configuration, significantly simplifying the workflow and reducing waste.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production. It utilizes common organic solvents like ethyl acetate and toluene, operates at manageable temperatures (-20°C to 50°C), and achieves high overall yields (over 76% in examples), making it economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulfinyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced chiral synthesis methods requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify enantiomeric excess and impurity profiles, guaranteeing that every batch of Esomeprazole or Lansoprazole intermediate meets the highest global standards. Our commitment to quality assurance ensures that your supply chain remains uninterrupted and compliant with all regulatory requirements.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product's competitiveness in the global marketplace.
