Advanced Catalyst-Free Reduction of Nitrosobenzene Compounds for Commercial Scale Aniline Production
Introduction to Advanced Nitrosobenzene Reduction Technology
The synthesis of aniline derivatives represents a cornerstone operation in the production of active pharmaceutical ingredients (APIs), agrochemicals, and functional dyes. Traditionally, converting nitroso or nitro groups into amino functionalities has been a bottleneck due to harsh conditions and purification challenges. However, the technological breakthrough detailed in patent CN115960002A introduces a paradigm shift in this domain. This patent discloses a highly efficient method for reducing nitrosobenzene compounds utilizing hydrazine hydrate as a reducing agent within an alkaline aqueous system. Unlike conventional catalytic hydrogenation, this approach operates under remarkably mild conditions, typically ranging from 0°C to 30°C, and crucially, it functions without the necessity for additional transition metal catalysts. This innovation not only streamlines the synthetic route but also addresses critical environmental and safety concerns associated with traditional reduction methodologies.
The core transformation involves the conversion of Compound 1, which contains one or more nitroso groups attached to an aromatic ring, into Compound 2, where these groups are reduced to amino groups. This general reaction scheme is fundamental to generating a vast array of chemical building blocks used globally. By leveraging hydrazine hydrate, the reaction generates nitrogen gas as the primary by-product, which naturally evolves from the reaction mixture, thereby driving the equilibrium forward and simplifying the downstream processing. For global procurement managers and R&D directors seeking a reliable pharmaceutical intermediates supplier, understanding this catalyst-free mechanism is vital for assessing long-term supply chain stability and cost structures.
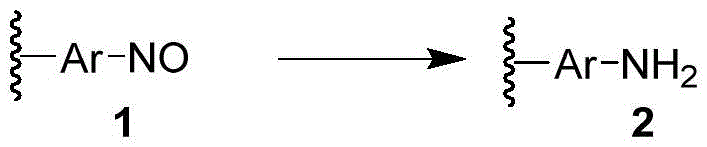
Furthermore, the versatility of this method extends to a wide range of substituted aromatic systems. The patent explicitly covers substrates where the aromatic rings may be substituted with various functional groups such as alkyl, alkoxy, hydroxyl, and amino groups, as well as complex bridged structures involving oxygen, sulfur, or nitrogen linkers. This broad substrate scope ensures that the technology is applicable to the commercial scale-up of complex polymer additives and specialty chemicals, not just simple anilines. The ability to tolerate diverse functional groups without requiring protective group strategies significantly enhances the step economy of multi-step syntheses, making it an attractive option for process chemists aiming to optimize overall yield and reduce waste.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the existing landscape of industrial organic synthesis, the preparation of aniline compounds predominantly relies on three established methods: iron powder reduction, phenol ammoniation, and nitrobenzene catalytic hydrogenation. Each of these legacy technologies carries significant baggage that impacts both operational expenditure and environmental compliance. The iron powder reduction method, while historically common, is notoriously inefficient; it consumes massive amounts of energy and generates substantial quantities of iron sludge, creating a severe pollution burden that requires costly waste treatment protocols. Moreover, the separation of the organic product from the inorganic residue is often difficult and leads to product loss, directly affecting the final yield and purity profile required for high-grade applications.
Alternatively, the phenol ammoniation method and nitrobenzene catalytic hydrogenation method, while offering cleaner profiles than iron reduction, introduce their own set of economic and safety hurdles. These processes invariably require the use of expensive noble metal catalysts such as palladium or platinum, or reactive metals like Raney nickel. The reliance on these catalysts not only inflates the raw material costs but also necessitates rigorous post-reaction purification steps to remove trace metal residues, which is a critical quality attribute for pharmaceutical intermediates. Furthermore, catalytic hydrogenation typically demands high-temperature and high-pressure reaction conditions, imposing strict safety requirements on the reactor infrastructure and increasing the capital expenditure for manufacturing facilities. These factors collectively contribute to higher production costs and longer lead times, challenging the efficiency of the global fine chemical intermediates supply chain.
The Novel Approach
The novel approach presented in patent CN115960002A effectively dismantles these barriers by introducing a hydrazine hydrate-mediated reduction in an alkaline aqueous medium. This method operates at near-ambient temperatures (0-30°C) and atmospheric pressure, drastically reducing the energy footprint and safety risks associated with high-pressure hydrogenation. The elimination of transition metal catalysts is perhaps the most significant advantage; it removes the need for expensive catalyst loading and the subsequent, often tedious, metal scavenging steps. This results in a cleaner reaction profile where the primary by-product is nitrogen gas, which harmlessly vents from the system, leaving behind a solution rich in the desired aniline product.
Additionally, the use of water as the primary solvent, specifically dilute sodium hydroxide solutions (1wt%-5wt%), aligns perfectly with green chemistry principles. Water is non-flammable, non-toxic, and inexpensive, contrasting sharply with the volatile organic solvents often required in other reduction protocols. The patent highlights that when the target aniline product is a solid at room temperature, it can be isolated simply by cooling the reaction mixture and filtering, bypassing the need for energy-intensive distillation or complex extraction procedures. This simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it shortens the batch cycle time and minimizes solvent consumption. For supply chain heads, this robustness means fewer variables that could cause batch failures, ensuring a more consistent and reliable flow of materials.
Mechanistic Insights into Hydrazine-Mediated Reduction
The mechanistic pathway of this reduction involves the nucleophilic attack of hydrazine on the electrophilic nitrogen of the nitroso group, facilitated by the alkaline environment. The hydroxide ions in the solution likely assist in deprotonating the hydrazine, enhancing its nucleophilicity, and stabilizing intermediate species. As the reaction progresses, the nitrogen-nitrogen bond in hydrazine is cleaved, transferring hydrogen equivalents to the nitroso group to form the amine, while the remaining nitrogen atoms combine to form molecular nitrogen (N2). This evolution of gas provides a thermodynamic driving force that pushes the reaction to completion, often achieving yields exceeding 90% as demonstrated in the patent examples. The absence of metal surfaces prevents side reactions such as over-reduction or hydrogenolysis of sensitive functional groups like benzyl ethers or halides, which are common pitfalls in catalytic hydrogenation.
Impurity control is another critical aspect where this mechanism excels. In traditional metal-catalyzed reductions, trace metals can catalyze decomposition pathways or couple with the product to form difficult-to-remove impurities. By operating in a metal-free homogeneous (or biphasic with additives) system, the impurity profile is significantly simplified. The patent data indicates that liquid phase purities of the resulting aniline compounds can reach as high as 99.6% (as seen in Example 1 for Compound 2a) directly after filtration and drying. This high purity is achieved without the need for column chromatography on a large scale, which is a major cost driver. The tolerance for various substituents is also remarkable; the method successfully reduces nitroso groups on phenols, biphenyls, and diphenyl ethers, as illustrated by the diverse substrate library in the patent documentation.
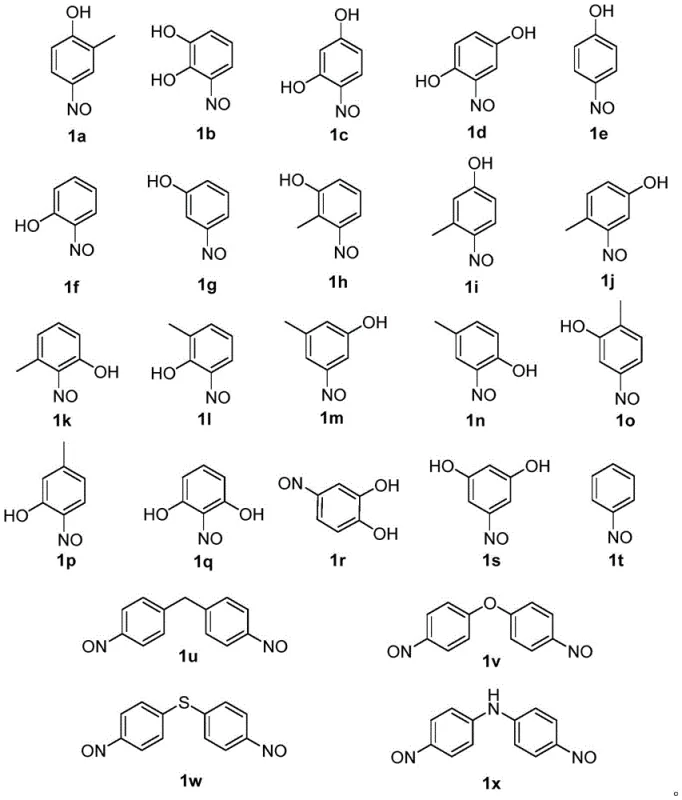
Specific examples from the patent underscore the robustness of this chemistry. For instance, the reduction of 4-nitroso-2-methylphenol (Compound 1a) to 4-amino-2-methylphenol (Compound 2a) was optimized to achieve an 87.1% yield with 98.0% purity by adding n-heptane as an additive (Example 4). The additive likely acts to extract the organic product or stabilize the interface, preventing over-oxidation or tar formation. Similarly, the reduction of 4,4'-dinitrosodiphenyl ether (Compound 1v) to the corresponding diamine (Compound 2v) proceeded with a 90.0% yield and 96.1% purity (Example 12). These results confirm that the method is scalable and effective for both mono- and di-substituted systems, providing a versatile tool for the synthesis of high-purity OLED material precursors and agrochemical intermediates.
How to Synthesize 4-Amino-2-Methylphenol Efficiently
To implement this technology in a pilot or production setting, the procedure generally involves dissolving or suspending the nitrosobenzene starting material in a dilute aqueous sodium hydroxide solution. An optional water-immiscible organic additive, such as n-heptane or tert-butyl methyl ether, may be introduced to improve mixing or product isolation. Hydrazine hydrate (typically 85% concentration) is then added dropwise while maintaining the temperature between 0°C and 20°C to control the exotherm. The mixture is stirred for a period ranging from 4 to 24 hours, depending on the specific substrate reactivity. Upon completion, if the product is a solid, it is isolated by cooling the mixture to 0-15°C and filtering. If the product remains in solution, standard extraction and workup procedures are applied. The detailed standardized synthesis steps for specific compounds are outlined below.
- Prepare the reaction system by mixing the nitrosobenzene compound (Compound 1) with an alkaline aqueous solvent, such as 1wt%-5wt% sodium hydroxide solution, and optionally add a water-immiscible organic additive like n-heptane.
- Cool the mixture to a temperature range of 0-20°C and slowly add hydrazine hydrate (85% concentration) while maintaining the reaction temperature between 0-30°C.
- Stir the reaction mixture for 4 to 24 hours, then isolate the resulting aniline compound (Compound 2) via filtration if solid, or extraction and purification if liquid, achieving high purity without heavy metal residues.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrazine-based reduction technology offers tangible strategic advantages beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the supply chain for raw materials and the reduction of dependency on critical minerals. By eliminating the need for palladium, platinum, or nickel catalysts, manufacturers are insulated from the volatile pricing and geopolitical supply risks associated with these precious metals. This shift not only stabilizes the cost of goods sold (COGS) but also removes the logistical complexity of sourcing and handling hazardous pyrophoric catalysts like Raney nickel. Consequently, this leads to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing in the final market.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the elimination of expensive catalysts and the reduction of energy consumption. Traditional hydrogenation requires high-pressure reactors and significant heating, whereas this method operates at ambient pressure and low temperatures (0-30°C), leading to significantly lower utility costs. Furthermore, the workup procedure is streamlined; since the by-product is nitrogen gas and the solvent is water, there is no need for complex solvent recovery systems or extensive wastewater treatment for heavy metals. The ability to isolate products via simple filtration for solid anilines further reduces processing time and labor costs, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Reliability in the supply of key intermediates is paramount for downstream drug manufacturers. This method enhances reliability by utilizing commodity chemicals—hydrazine hydrate and sodium hydroxide—which are widely available globally with stable supply chains. Unlike specialized catalysts that may have long lead times or single-source suppliers, the reagents for this process are easily procurable. Additionally, the mild reaction conditions reduce the wear and tear on manufacturing equipment, minimizing unplanned downtime for maintenance. The robustness of the reaction against varying substrate qualities ensures consistent batch-to-batch performance, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring on-time delivery to customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this technology mitigates those concerns effectively. The use of water as a solvent inherently reduces fire hazards compared to large-scale organic solvent usage. Moreover, the absence of heavy metal waste simplifies regulatory compliance and waste disposal, which is increasingly stringent in global manufacturing hubs. The process generates minimal hazardous waste, aligning with modern sustainability goals and reducing the environmental tax burden. This ease of scalability means that production can be ramped up from kilogram to multi-ton scales without significant re-engineering of the process, ensuring that supply can meet surging demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrosobenzene reduction technology. These answers are derived directly from the experimental data and specifications provided in patent CN115960002A, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: Does this reduction method require expensive transition metal catalysts?
A: No, the method described in patent CN115960002A utilizes hydrazine hydrate as the sole reducing agent in an alkaline environment, completely eliminating the need for costly palladium, platinum, or Raney nickel catalysts.
Q: What are the typical reaction conditions for this nitrosobenzene reduction?
A: The reaction proceeds under mild conditions, typically between 0°C and 30°C, using an aqueous sodium hydroxide solution as the solvent, which significantly reduces energy consumption compared to high-pressure hydrogenation methods.
Q: How does this method impact the purity of the final aniline product?
A: By avoiding transition metal catalysts, the process prevents metal contamination, and the by-product is nitrogen gas which escapes the system, allowing for the isolation of aniline derivatives with liquid phase purities exceeding 99% through simple filtration or crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-2-Methylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free hydrazine reduction technology described in patent CN115960002A for the production of high-value aniline derivatives. As a leading CDMO partner, we possess the technical expertise to adapt and optimize this proprietary route for your specific project needs. Our facilities are equipped to handle diverse synthetic pathways, and we bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key; therefore, our stringent purity specifications and rigorous QC labs ensure that every batch of 4-amino-2-methylphenol or related intermediates meets the highest international standards, free from the heavy metal contaminants often associated with traditional reduction methods.
We invite you to collaborate with us to leverage this advanced synthesis method for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient process. We encourage you to contact our technical procurement team today to request specific COA data for our aniline intermediates and to discuss route feasibility assessments tailored to your unique molecular targets. Let us help you secure a sustainable and cost-effective supply chain for your critical chemical building blocks.
