Revolutionizing Rubber Antiozonant Production: A Deep Dive into Novel Triazine Synthesis
The global demand for high-performance rubber antiozonants continues to drive innovation in fine chemical synthesis, particularly for heavy-duty applications such as agricultural and truck tires. Patent CN113429361B introduces a groundbreaking preparation method for 2,4,6-tris(N-1,4-dimethylpentyl p-phenylenediamine)-1,3,5-triazine, commonly known in the industry as TAPDT. This specific chemical architecture is critical for preventing rubber aging caused by heat, ozone, and mechanical fatigue, addressing the persistent issue of discoloration and migration found in traditional alkylaryl anti-aging agents. Unlike conventional methods that struggle with selectivity and purification, this novel protocol utilizes a unique formamide-based intermediate strategy to achieve superior reaction control. By shifting the synthetic paradigm from direct amine alkylation to a triazine-formamide coupling sequence, the technology offers a robust solution for manufacturers seeking to enhance product stability while optimizing production costs. This report analyzes the technical merits and commercial implications of this patented route for international procurement and R&D teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of TAPDT and similar antiozonants has relied heavily on the direct reaction of p-phenylenediamine with ketones, a process fraught with significant chemical engineering challenges. As documented in prior art such as US4794135, this traditional one-step reaction requires precise control over the ketone-to-amine ratio, often necessitating a large excess of amine to drive conversion. This excess creates a downstream bottleneck, as the unreacted aniline derivatives must be recovered from the mixture, a process that is both energy-intensive and technically difficult due to similar boiling points. Furthermore, the direct alkylation pathway frequently generates N'-di(dimethylpentyl) p-phenylenediamine, known as anti-aging agent 4030, as a persistent by-product that compromises the purity profile of the final triazine. These impurities not only affect the performance of the rubber compound but also complicate the isolation process, leading to lower overall yields and increased waste generation that negatively impacts the cost structure of manufacturing.
The Novel Approach
In stark contrast to the legacy methodologies, the process disclosed in CN113429361B employs a stepwise construction of the triazine core using 2,4,6-tris(formamido)-1,3,5-triazine as a pivotal intermediate. This strategic shift allows for the utilization of cyanuric chloride and formamide, which are small molecules with minimal steric hindrance, facilitating a highly selective nucleophilic substitution reaction at low temperatures. By establishing the triazine skeleton early in the synthesis using formamide groups, the method effectively bypasses the formation of difficult-to-remove dialkylated impurities associated with direct amine usage. The subsequent conversion of the formamido groups to the target N-1,4-dimethylpentyl p-phenylenediamine moieties is achieved through a controlled coupling with p-chloronitrobenzene followed by catalytic hydrogenation. This modular approach not only simplifies the operational workflow but also dramatically improves the selectivity and yield of the reaction, ensuring a cleaner product profile that meets the stringent quality requirements of modern tire manufacturing without the need for complex purification trains.
Mechanistic Insights into Formamide-Mediated Triazine Construction
The core innovation of this technology lies in the initial formation of the 2,4,6-tris(formamido)-1,3,5-triazine intermediate, a reaction that leverages the high nucleophilicity of formamide under basic conditions. In this step, cyanuric chloride reacts with formamide and sodium carbonate in an aqueous-organic biphasic system, typically maintained at temperatures between -5°C and 5°C to prevent hydrolysis of the chloride groups. The mechanism involves the sequential displacement of chlorine atoms on the triazine ring by the nitrogen of the formamide, driven by the neutralization of the generated hydrochloric acid by the carbonate base. This low-temperature protocol is critical for maintaining the integrity of the triazine ring and ensuring that the formamido groups are installed with high fidelity, setting the stage for the subsequent transformation steps. The use of formamide as a protecting and activating group is particularly advantageous because it avoids the steric bulk associated with larger amine molecules, allowing for complete substitution of all three chlorine atoms without the formation of partially substituted by-products.
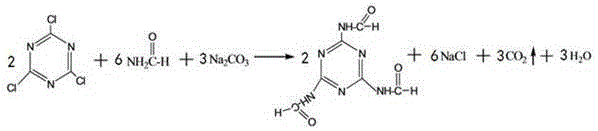
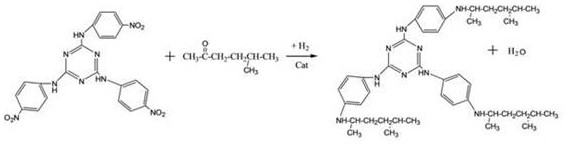
Following the formation of the formamido intermediate, the synthesis proceeds through a coupling reaction with p-chloronitrobenzene, followed by a critical reductive alkylation step that defines the final physical properties of the antiozonant. The intermediate reacts with p-chloronitrobenzene to form 2,4,6-tris(4-nitrophenyl)-1,3,5-triazine, which then undergoes catalytic hydrogenation in the presence of methyl isoamyl ketone. This reductive alkylation is a sophisticated transformation where the nitro groups are reduced to amines in situ, which immediately react with the ketone to form the secondary amine linkage, all within a single high-pressure reactor setup. The choice of catalyst, such as palladium-carbon or platinum-carbon, and the control of hydrogen pressure at approximately 0.2 MPa are essential for driving this reaction to completion while minimizing the formation of over-reduced or side-reaction products. This integrated reduction-alkylation mechanism ensures that the bulky 1,4-dimethylpentyl groups are attached precisely to the para-position of the phenyl rings, resulting in a molecule with the optimal molecular weight and migration resistance required for high-performance tire applications.
How to Synthesize TAPDT Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions and stoichiometry to maximize the benefits of the novel intermediate strategy. The process begins with the preparation of the formamido-triazine in an aqueous slurry, followed by extraction into an organic solvent like toluene, which can be carried forward directly or isolated depending on plant configuration. The subsequent coupling and reduction steps are performed in standard pressure reactors, making the technology highly adaptable to existing fine chemical infrastructure without the need for specialized exotic equipment. For detailed operational parameters, including specific temperature ramps, catalyst loading rates, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical control points for ensuring batch-to-batch consistency.
- React cyanuric chloride with formamide and sodium carbonate at low temperature to form the 2,4,6-tris(formamido) intermediate.
- Couple the formamido intermediate with p-chloronitrobenzene under reflux conditions to generate the nitro-triazine precursor.
- Perform catalytic hydrogenation reduction alkylation using methyl isoamyl ketone to finalize the TAPDT structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this formamide-based synthesis route offers substantial advantages in terms of raw material security and overall cost structure optimization. The primary feedstocks, cyanuric chloride and formamide, are commodity chemicals produced on a massive global scale, ensuring a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized amine intermediates. By eliminating the need for p-phenylenediamine in the initial steps, manufacturers can avoid the price fluctuations and supply constraints associated with this specific aromatic amine, thereby stabilizing the cost of goods sold over the long term. Furthermore, the high selectivity of the reaction means that less raw material is wasted on by-product formation, directly translating to improved atom economy and reduced expenditure on waste disposal and environmental compliance measures. This efficiency allows for a more competitive pricing model for the final antiozonant product while maintaining healthy margins for the producer.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers manufacturing costs by removing the need for complex distillation columns required to recover excess amines in traditional methods. The ability to operate at moderate temperatures and pressures reduces energy consumption, while the high yield minimizes the cost per kilogram of the active ingredient. Additionally, the use of recoverable solvents and heterogeneous catalysts further enhances the economic viability of the process, allowing for substantial cost savings that can be passed down the supply chain to end-users.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals rather than custom-synthesized intermediates drastically reduces the risk of supply disruptions. The simplified process flow shortens the production cycle time, enabling manufacturers to respond more quickly to changes in market demand and reduce lead times for customers. This agility is crucial for maintaining continuous production schedules in the tire and rubber industry, where downtime can be extremely costly, ensuring that customers receive their orders consistently and without delay.
- Scalability and Environmental Compliance: The chemistry is inherently scalable, having been demonstrated effectively from laboratory bench scales to multi-ton commercial production batches without loss of efficiency. The reduction in by-products and the use of less hazardous reagents contribute to a cleaner environmental profile, making it easier for facilities to meet increasingly strict global environmental regulations. This compliance advantage reduces the regulatory burden on the supply chain and future-proofs the manufacturing asset against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the value proposition for potential partners. Understanding these details is essential for R&D and procurement teams evaluating the feasibility of integrating this material into their supply chains.
Q: How does the formamide route improve impurity profiles compared to traditional PPD methods?
A: The formamide route avoids the use of excess p-phenylenediamine, which is difficult to recover and often leads to N,N'-di-substituted impurities. By using small-molecule formamide, steric hindrance is minimized, resulting in higher selectivity and easier purification of the final triazine product.
Q: What are the scalability advantages of this specific triazine synthesis pathway?
A: The process utilizes cheap and readily available raw materials like cyanuric chloride and formamide. The reaction conditions are moderate, and the intermediate can be used directly in solution or isolated, offering flexibility for large-scale continuous manufacturing without complex distillation steps.
Q: Does this method reduce the environmental footprint of antiozonant production?
A: Yes, the method significantly reduces by-product formation and eliminates the need for recovering high-boiling-point amines. The simplified workflow and higher atom economy contribute to reduced waste generation and lower energy consumption during the purification phases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TAPDT Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patented routes like CN113429361B into reliable commercial reality. Our engineering teams have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial volume is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. This commitment to quality ensures that the TAPDT supplied meets the exacting performance requirements of global tire manufacturers, providing the durability and stability necessary for modern rubber applications.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency. By partnering with us, you gain access to a secure source of high-performance antiozonants backed by a commitment to continuous improvement and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
