Advanced One-Pot Tandem Synthesis of Dipyrrole Derivatives for Commercial Pharmaceutical Manufacturing
Introduction to Novel Dipyrrole Synthesis Technology
The pharmaceutical industry is constantly seeking robust and scalable pathways to access complex heterocyclic scaffolds, particularly those with proven biological significance. Patent CN102351772A introduces a groundbreaking methodology for the tandem synthesis of dipyrrole and its derivatives through a highly efficient one-pot process. This technology addresses the critical need for streamlined manufacturing of 1,3'-dipyrrole cores, which serve as essential precursors for a vast array of ocean natural products and bioactive molecules. Unlike traditional approaches that often suffer from harsh conditions and low atom economy, this invention utilizes a tandem reaction between trisubstituted alkenes and isocyanides to construct the dipyrrole framework with remarkable precision. The structural versatility of the resulting compounds, as illustrated by the general formula (I), allows for extensive functionalization, making this route highly attractive for medicinal chemistry campaigns focused on antibacterial and antitumor agents.
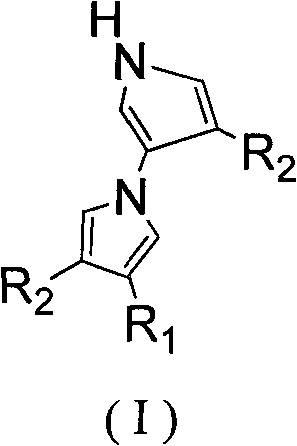
Furthermore, the strategic design of this synthesis ensures that the final products possess inherent biological activities, including strong cytotoxicity against human cancer cells and efficacy against methicillin-resistant Staphylococcus aureus (MRSA). By establishing a direct link between simple, commercially available starting materials and high-value pharmacological targets, this patent represents a significant leap forward in process chemistry. For R&D directors and procurement specialists alike, understanding the nuances of this transformation is key to securing a reliable supply chain for next-generation therapeutic intermediates. The ability to generate such complex architectures in a single operational step not only accelerates discovery timelines but also lays the foundation for cost-effective commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,3'-dipyrrole derivatives has been plagued by synthetic inefficiencies that hinder large-scale adoption. Conventional methodologies often rely on multi-step sequences involving sensitive intermediates, requiring rigorous protection and deprotection strategies that drastically increase material costs and processing time. Many existing reports describe the use of microwave-assisted synthesis or other complicated compound methods which, while effective on a milligram scale, present formidable challenges when transitioning to kilogram or ton-scale manufacturing. These traditional routes frequently demand harsh reaction conditions, including extreme temperatures or pressures, which pose safety risks and require specialized equipment that increases capital expenditure. Additionally, the reliance on difficult-to-obtain raw materials in older synthetic pathways creates supply chain bottlenecks, leading to inconsistent availability and volatile pricing for the final intermediates. The cumulative effect of these limitations is a high barrier to entry for the commercial production of dipyrrole-based active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast, the method disclosed in CN102351772A revolutionizes the landscape by employing a simple, one-pot tandem reaction that bypasses the need for isolation of intermediates. This novel approach leverages the reactivity of trisubstituted alkenes, represented by formula (II), and isocyanides, specifically p-toluenesulfonyl methyl isocyanide (TosMIC) represented by formula (III), to directly forge the dipyrrole skeleton. 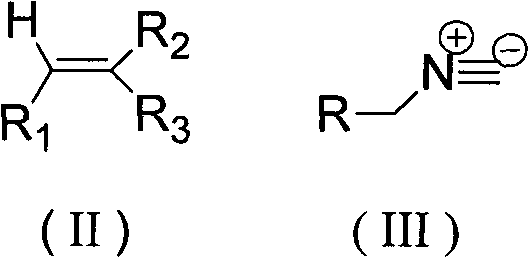
The brilliance of this strategy lies in its operational simplicity; the reaction proceeds smoothly under mild conditions, typically ranging from 0°C to 60°C, using readily available organic solvents and base catalysts. By eliminating the need for transition metals or exotic reagents, this process significantly reduces the impurity profile associated with heavy metal contamination, a critical parameter for regulatory compliance in pharma manufacturing. The one-pot nature of the reaction means that the entire transformation occurs in a single vessel, minimizing solvent usage, reducing waste generation, and lowering the overall environmental footprint. This shift from complex, multi-step protocols to a streamlined tandem process offers a clear pathway for cost reduction in pharmaceutical intermediate manufacturing, ensuring that high-quality dipyrrole derivatives can be produced with greater efficiency and reliability.
Mechanistic Insights into Base-Catalyzed Tandem Cyclization
The core of this technological advancement is the base-catalyzed tandem cyclization mechanism, which orchestrates the convergence of the alkene and isocyanide components into the final dipyrrole structure. The reaction initiates with the deprotonation of the acidic methylene group in the isocyanide (TosMIC) by the base catalyst, generating a nucleophilic carbanion species. This activated nucleophile then attacks the electron-deficient double bond of the trisubstituted alkene, triggering a cascade of intramolecular cyclizations. The choice of base is pivotal; the patent highlights the effectiveness of both inorganic bases like sodium hydride (NaH), potassium carbonate (K2CO3), and cesium carbonate (Cs2CO3), as well as organic bases such as DBU and triethylamine (TEA). Each base offers distinct kinetic profiles, allowing process chemists to fine-tune the reaction rate and selectivity based on the specific electronic nature of the substituents R1, R2, and R3. The mechanistic pathway ensures high regioselectivity, favoring the formation of the 1,3'-linkage essential for the desired biological activity.
From an impurity control perspective, this mechanism is exceptionally clean. Because the reaction avoids radical pathways or high-energy intermediates often seen in thermal cyclizations, the formation of side products is minimized. The use of mild temperatures (0°C to 60°C) further suppresses decomposition pathways that could lead to polymeric byproducts or tar formation. Moreover, the workup procedure described—quenching with saturated ammonium chloride followed by extraction and washing with sodium carbonate—effectively removes residual base and polar impurities. This results in a crude product of high purity, which can be further refined to stringent specifications using standard column chromatography or recrystallization. For quality assurance teams, this predictable impurity profile simplifies analytical method development and validation, ensuring that the final API intermediate meets the rigorous standards required for clinical and commercial use.
How to Synthesize Dipyrrole Derivatives Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and reaction parameters to maximize yield and purity. The patent outlines a robust protocol where the molar ratio of the alkene to the isocyanide is optimized between 1.0:0.8 and 1.0:2.5, with a preferred range of 1.0:1.0 to 1.3 to ensure complete conversion of the limiting reagent. The solvent volume is equally critical, recommended at 3 to 5 mL per mmol of the alkene substrate to maintain optimal concentration for the tandem cyclization. Common solvents such as acetonitrile, THF, and DMF have shown excellent performance, providing a balance between solubility and reaction kinetics. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-efficiency process in a pilot or production setting.
- Mix trisubstituted alkene and isocyanide (such as TosMIC) in an organic solvent like acetonitrile or THF under nitrogen protection.
- Add a base catalyst (e.g., NaH, K2CO3, or Cs2CO3) and stir at temperatures between 0°C and 60°C for 0.5 to 24 hours.
- Quench the reaction with saturated ammonium chloride, extract with dichloromethane, wash, dry, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis technology translates into tangible strategic advantages that extend beyond mere technical feasibility. The elimination of transition metal catalysts is a primary driver for cost optimization, as it removes the necessity for expensive metal scavengers and the associated validation steps required to prove low residual metal levels in the final product. This simplification of the downstream processing significantly lowers the cost of goods sold (COGS) and reduces the burden on waste management systems. Furthermore, the use of commodity chemicals like TosMIC and simple alkenes ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialty chemical markets. The mild reaction conditions also imply lower energy consumption, contributing to a more sustainable and economically viable manufacturing process.
- Cost Reduction in Manufacturing: The streamlined one-pot process drastically reduces the number of unit operations required, effectively cutting labor costs and facility occupancy time. By avoiding the use of precious metal catalysts, the process eliminates a major cost center and the complex purification steps associated with metal removal. The high atom economy of the tandem reaction ensures that a larger proportion of the starting mass is converted into the desired product, minimizing raw material waste. Additionally, the ability to use common, low-cost solvents like acetonitrile or ethyl acetate further drives down the variable costs of production, making the final dipyrrole intermediates highly competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as substituted alkenes and TosMIC ensures that the supply chain is resilient against disruptions. Unlike processes dependent on bespoke or hard-to-source reagents, this method allows for rapid scaling by leveraging existing global chemical infrastructure. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without significant re-validation, ensuring continuity of supply for downstream API manufacturers. This reliability is crucial for maintaining inventory levels and meeting the just-in-time delivery demands of major pharmaceutical clients.
- Scalability and Environmental Compliance: The mild temperature range (0°C to 60°C) and ambient pressure conditions make this process inherently safer and easier to scale from laboratory to commercial production. The absence of hazardous reagents and the generation of minimal waste align perfectly with modern green chemistry principles and strict environmental regulations. The simplified workup procedure reduces the volume of organic waste solvents requiring disposal, lowering environmental compliance costs. This scalability ensures that the technology can support the transition from clinical trial materials to multi-ton commercial production without the need for fundamental process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dipyrrole synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, optimization, and application potential. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this one-pot dipyrrole synthesis method?
A: The method offers mild reaction conditions (0-60°C), avoids expensive transition metal catalysts, and simplifies the workflow into a single pot, significantly reducing operational complexity and waste generation compared to traditional multi-step microwave methods.
Q: Which catalysts and solvents are compatible with this process?
A: The process is highly versatile, utilizing common inorganic and organic bases such as NaH, K2CO3, Cs2CO3, DBU, or TEA. Compatible solvents include acetonitrile, THF, DMF, and various chlorinated hydrocarbons, allowing for flexible process optimization.
Q: What are the biological applications of the synthesized dipyrrole derivatives?
A: The resulting 1,3'-dipyrrole derivatives exhibit significant antibacterial activity against MRSA and potent antitumor properties, making them valuable intermediates for developing novel oncology and anti-infective pharmaceutical agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dipyrrole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the one-pot tandem synthesis described in CN102351772A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into a robust, GMP-compliant commercial process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We are committed to delivering dipyrrole derivatives with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and potency.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our dipyrrole portfolio and to discuss route feasibility assessments that can accelerate your project timelines. Together, we can drive efficiency and innovation in the manufacturing of next-generation antitumor and antibacterial agents.
