Advanced Purification Technology for 1,2-Benzisothiazole-3-Ketone Intermediates
Introduction to Advanced BIT Purification Technologies
The landscape of fine chemical manufacturing is constantly evolving, driven by the dual imperatives of environmental sustainability and product quality enhancement. A pivotal development in this sector is detailed in patent CN103204823A, which introduces a superior method for purifying 1,2-benzisothiazole-3-ketone (BIT), a critical intermediate widely utilized in the production of biocides and sanitizers. This innovation addresses the significant limitations of conventional acid-base purification techniques by employing a sophisticated recrystallization strategy using water-soluble polar proton solvents mixed with water. By shifting away from harsh chemical treatments, this methodology not only drastically reduces the generation of saline wastewater but also transforms the physical morphology of the final product from a hazardous fine powder into a safe, snow-white granular solid. For industry leaders seeking a reliable agrochemical intermediate supplier, understanding this technological leap is essential for optimizing supply chains and ensuring regulatory compliance.
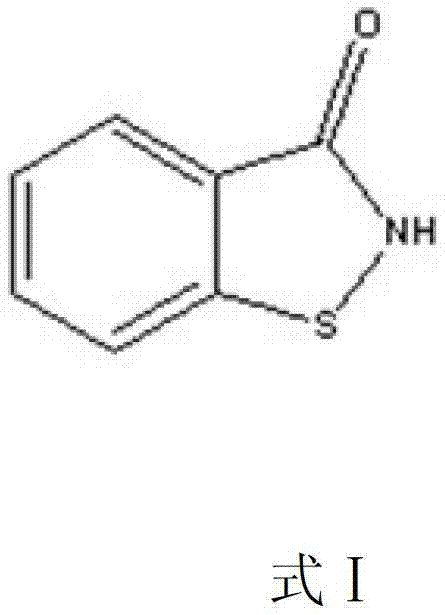
The structural integrity and purity of 1,2-benzisothiazole-3-ketone are paramount for its efficacy as a biocidal agent, necessitating rigorous purification standards that traditional methods often struggle to meet without excessive environmental cost. The patent outlines a process where the careful selection of solvents, such as methanol or ethanol in combination with water, creates an ideal thermodynamic environment for crystal growth. This approach ensures that the resulting material possesses high content and minimal impurity profiles, satisfying the demanding requirements of downstream pharmaceutical and agrochemical applications. As we delve deeper into the technical specifics, it becomes clear that this method represents a significant stride towards greener chemistry and operational excellence in the production of high-value heterocyclic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of 1,2-benzisothiazole-3-ketone has relied heavily on acid-base extraction methods, which involve dissolving the crude product in liquid caustic soda and subsequently precipitating it with hydrochloric acid. While effective in isolating the compound, this traditional approach suffers from severe drawbacks that impact both economic efficiency and environmental stewardship. The primary issue is the generation of substantial volumes of saline wastewater, which imposes a heavy burden on waste treatment facilities and increases the overall cost of production due to disposal fees and water consumption. Furthermore, the physical form of the product obtained through acid-base precipitation is typically a fine powder, which poses significant occupational health risks; the dust generated can be easily inhaled, causing severe irritation to the human respiratory tract and necessitating expensive containment and ventilation systems.
The Novel Approach
In stark contrast, the novel purification method described in the patent utilizes a solvent engineering approach that completely bypasses the need for acidic or alkaline reagents, thereby eliminating the root cause of saline waste generation. By leveraging the solubility characteristics of 1,2-benzisothiazole-3-ketone in mixed solvent systems comprising water-soluble polar proton solvents and water, the process achieves high-purity isolation through controlled recrystallization. This shift not only aligns with modern green chemistry principles by reducing pollution but also fundamentally alters the physical properties of the output. The resulting product is a snow-white granular crystal rather than a fine dust, which dramatically improves operational safety by preventing the formation of inhalable airborne particles. This transformation in morphology also facilitates easier drying and handling, making the material ideally suited for subsequent anhydrous processing steps without the complications associated with hygroscopic powders.
Mechanistic Insights into Solvent-Mediated Recrystallization
The core mechanism driving the success of this purification strategy lies in the precise manipulation of solubility parameters and supersaturation levels within the solvent mixture. When 1,2-benzisothiazole-3-ketone is dissolved in a heated mixture of a polar proton solvent like methanol and water, the system reaches a state of complete solvation where impurities are either kept in solution or removed via activated carbon decolorization. Upon controlled cooling, the solubility of the target compound decreases, leading to nucleation and crystal growth. The presence of water in the organic solvent matrix acts as an anti-solvent to some degree, modulating the rate of crystallization to favor the formation of larger, more regular granular structures rather than rapid, chaotic precipitation of fine particles. This controlled growth kinetics is crucial for excluding impurities from the crystal lattice, as slower growth allows the molecules to arrange themselves into a more ordered and pure structure, effectively rejecting foreign species that do not fit the lattice geometry.
Furthermore, the choice of specific solvents such as methanol, ethanol, or acetic acid allows for fine-tuning the polarity of the medium to match the dipole moment of the BIT molecule, ensuring maximum recovery and purity. The patent specifies optimal mass ratios between the solvent, water, and the crude material, which are critical for maintaining the correct degree of supersaturation throughout the cooling phase. If the cooling is too rapid or the solvent ratio is incorrect, the system might revert to forming undesirable micro-crystals or oils. However, by adhering to the specified temperature gradients—dissolving at 60-80°C and cooling slowly to 10-25°C—the process ensures a thermodynamic pathway that favors the most stable crystalline form. This mechanistic control is what enables the production of material with HPLC purity exceeding 99.85%, a benchmark that is difficult to achieve with less controlled precipitation methods.
How to Synthesize 1,2-Benzisothiazole-3-ketone Efficiently
Implementing this advanced purification protocol requires strict adherence to the defined process parameters to ensure consistent quality and yield. The procedure begins with the preparation of the recrystallization solvent, followed by the dissolution of the crude BIT under heated conditions to ensure homogeneity. Decolorization steps are integrated to remove colored impurities, after which the solution is subjected to a controlled cooling regime to induce crystallization. The final isolation involves filtration and drying to obtain the free-flowing granular product. For R&D teams looking to adopt this methodology, the following guide outlines the critical operational steps derived directly from the patent data to facilitate seamless technology transfer and scale-up.
- Mix water-soluble polar proton solvents (such as methanol or ethanol) with water to create a recrystallization solvent system.
- Dissolve crude 1,2-benzisothiazole-3-ketone in the solvent mixture by heating to 40-120°C, followed by decolorization and filtration.
- Cool the filtrate to 0-35°C with stirring to induce crystallization, then filter and dry the resulting granular crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel purification technology offers tangible strategic benefits that extend beyond mere technical specifications. The elimination of acid and alkali reagents translates directly into a simplified raw material inventory, reducing the complexity of sourcing and storing hazardous chemicals. Moreover, the drastic reduction in wastewater volume significantly lowers the operational expenditure associated with effluent treatment and environmental compliance, providing a clear pathway for cost reduction in fine chemical manufacturing. The transition from a dusty powder to a granular product also mitigates the need for extensive dust control infrastructure, further lowering capital and operational costs related to worker safety and facility maintenance.
- Cost Reduction in Manufacturing: The removal of acid-base neutralization steps eliminates the consumption of bulk chemicals like caustic soda and hydrochloric acid, directly reducing raw material costs. Additionally, the avoidance of large-scale saline wastewater generation means that facilities can operate with reduced load on their effluent treatment plants, leading to substantial savings in utility and waste disposal expenses. The high yield and purity achieved in a single recrystallization step also minimize the need for re-processing or multiple purification cycles, thereby enhancing overall process efficiency and throughput.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents such as methanol and ethanol ensures that the supply chain remains robust and resilient against fluctuations in specialty chemical availability. The granular nature of the final product improves flowability and packaging efficiency, reducing the risk of caking or degradation during storage and transport. This physical stability ensures that the material arrives at the customer's site in optimal condition, reducing the likelihood of rejection or quality disputes and fostering a more reliable supply partnership.
- Scalability and Environmental Compliance: The process is inherently scalable, as the principles of recrystallization are well-understood and easily adapted from laboratory to industrial scales without complex engineering changes. By significantly reducing the environmental footprint through the elimination of saline waste and hazardous dust, manufacturers can more easily comply with increasingly stringent global environmental regulations. This proactive approach to sustainability not only future-proofs the production facility but also enhances the brand value of the end products in markets that prioritize green chemistry and responsible sourcing.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this purification method for their specific applications, we have compiled a set of frequently asked questions based on the patent's technical disclosures. These inquiries address common concerns regarding process safety, product quality, and operational parameters, providing a clear overview of what stakeholders can expect when integrating this technology into their production lines. Understanding these details is crucial for assessing the potential impact on current manufacturing workflows and quality assurance protocols.
Q: What are the advantages of this purification method over traditional acid-base methods?
A: Unlike traditional methods that generate large amounts of saline wastewater and produce dusty powders, this novel recrystallization process eliminates the need for acid and alkali, significantly reducing pollution. Furthermore, it yields a snow-white granular product that minimizes respiratory hazards associated with inhaling fine BIT powder.
Q: What purity levels can be achieved with this recrystallization technique?
A: The optimized solvent system allows for the production of high-quality 1,2-benzisothiazole-3-ketone with HPLC purity exceeding 99.85%. The process effectively removes impurities while maintaining a high yield, making it suitable for applications requiring stringent quality specifications.
Q: Is the final product suitable for anhydrous applications?
A: Yes, the granular crystals produced by this method are free of crystallization water and are easy to dry. This makes the material particularly advantageous for downstream production processes that require strictly anhydrous conditions, facilitating easier handling and integration into synthesis workflows.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Benzisothiazole-3-ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the synthesis of effective biocides and pharmaceutical agents. Our expertise as a CDMO partner allows us to leverage advanced purification technologies, such as the solvent-based recrystallization method described herein, to deliver 1,2-benzisothiazole-3-ketone that meets the most rigorous industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material regardless of their volume requirements. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch for identity, assay, and impurity profiles, guaranteeing performance in your final formulations.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of superior purification strategies. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced methods can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-performance chemical intermediates.
