Scalable Metal-Free Synthesis of Aryl Phosphonates for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Aryl Phosphonates for Advanced Pharmaceutical Intermediates
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, safer, and more cost-effective synthetic methodologies. Patent CN108586527B introduces a groundbreaking approach to synthesizing aryl phosphonate compounds, a class of molecules pivotal in medicinal chemistry and material science. Unlike traditional routes that rely on harsh conditions or toxic transition metals, this invention leverages visible light photocatalysis using inexpensive organic dyes. For R&D directors and procurement specialists seeking a reliable aryl phosphonate supplier, this technology represents a paradigm shift towards sustainable manufacturing. The core innovation lies in the direct coupling of aryl hydrazines with phosphite esters under mild, metal-free conditions, effectively bypassing the limitations of classical nucleophilic substitutions.
This novel pathway not only simplifies the operational complexity but also addresses critical purity concerns inherent in pharmaceutical intermediate production. By eliminating the need for palladium or copper catalysts, the process inherently reduces the risk of heavy metal contamination, a major regulatory hurdle in drug development. Furthermore, the use of readily available starting materials and ambient reaction conditions suggests a substantial potential for cost reduction in pharmaceutical intermediates manufacturing. As we delve deeper into the technical specifics, it becomes clear that this method offers a robust solution for the commercial scale-up of complex organophosphorus compounds.
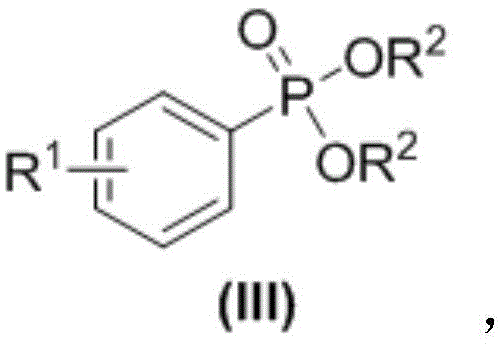
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl phosphonates has been fraught with significant chemical and economic challenges. The classic Michaelis-Arbuzov reaction, while effective for alkyl phosphonates, fails miserably with aryl halides due to the inability of the aromatic ring to undergo SN2 substitution. Consequently, chemists have been forced to rely on transition-metal-catalyzed cross-coupling reactions, typically employing expensive palladium or copper complexes. These methods not only inflate the raw material costs but also introduce severe downstream processing burdens. The removal of trace metal residues to meet ppm-level specifications requires additional purification steps, such as scavenger treatments or extensive chromatography, which drastically lower overall yield and increase waste generation. Moreover, these reactions often demand rigorous exclusion of oxygen and moisture, necessitating specialized equipment and inert gas supplies that further escalate operational expenditures.
The Novel Approach
In stark contrast, the methodology disclosed in CN108586527B utilizes a visible-light-driven radical mechanism that operates efficiently in an open system exposed to air. By employing organic dyes like Eosin Y or Eosin B as photosensitizers, the reaction initiates at room temperature (25-45°C) without any external heating or cooling beyond ambient control. This approach fundamentally alters the economic model of production by removing the dependency on precious metals entirely. The reaction tolerates a wide array of functional groups, including halogens and trifluoromethyl groups, which are often sensitive to harsh thermal conditions or strong bases used in older protocols. This tolerance expands the chemical space accessible to process chemists, allowing for the late-stage functionalization of complex drug candidates without protecting group strategies.

Mechanistic Insights into Visible Light Photocatalytic Coupling
The mechanistic elegance of this transformation lies in the photo-induced single electron transfer (SET) process facilitated by the organic dye. Upon irradiation with visible light, the photosensitizer enters an excited state, acting as a potent redox mediator. It interacts with the aryl hydrazine substrate to generate a reactive aryl radical species through the extrusion of nitrogen gas. This highly reactive intermediate then attacks the phosphorus center of the phosphite ester, forming the crucial carbon-phosphorus bond. The presence of a catalytic amount of base, such as triethylene diamine or triethylamine, plays a vital role in neutralizing acidic byproducts and regenerating the catalytic cycle. This radical pathway avoids the high-energy oxidative addition steps required in palladium catalysis, thereby lowering the activation energy barrier and enabling the reaction to proceed under such mild thermal conditions.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for high-purity aryl phosphonate production. Since no transition metals are introduced, the impurity profile is significantly cleaner, dominated primarily by organic byproducts that are easier to separate than metal complexes. The reaction's selectivity is governed by the stability of the generated aryl radicals, which minimizes side reactions such as homocoupling or over-oxidation. Detailed analysis of the substrate scope reveals that both electron-rich and electron-deficient aryl hydrazines participate effectively. For instance, substrates bearing strong electron-withdrawing groups like trifluoromethyl maintain high reactivity, demonstrating the robustness of the radical intermediate against electronic deactivation. This consistency ensures batch-to-batch reproducibility, a key metric for supply chain reliability.
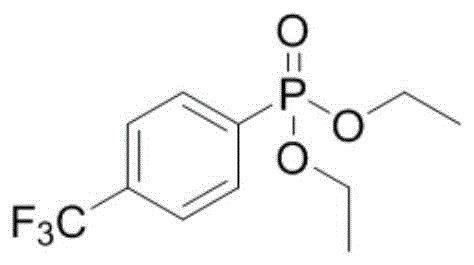
How to Synthesize Diethyl Phenylphosphonate Efficiently
The practical implementation of this synthesis is straightforward and designed for ease of adoption in standard laboratory or pilot plant settings. The protocol involves mixing the aryl hydrazine and phosphite ester in a polar aprotic solvent like acetonitrile, followed by the addition of the organic base and the eosin dye. The mixture is simply stirred under white light irradiation, eliminating the need for specialized high-pressure reactors or cryogenic cooling systems. This simplicity translates directly into reduced capital expenditure for manufacturing facilities. For detailed operational parameters regarding stoichiometry, light intensity, and workup procedures, please refer to the standardized guide below.
- Combine aryl hydrazine, phosphite ester, solvent (acetonitrile/DMF), organic base, and eosin photosensitizer in a reaction vessel.
- Stir the mixture in air under visible light irradiation at mild temperatures (25-45°C) for 1 to 6 hours.
- Isolate the target aryl phosphonate compound via standard silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this photocatalytic technology offers compelling strategic benefits beyond mere technical novelty. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as palladium prices are subject to significant market fluctuations. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents, leading to substantial cost savings in waste disposal and raw material usage. The ability to run reactions in air at near-ambient temperatures also lowers energy consumption significantly compared to refluxing conditions or cryogenic processes. These factors collectively contribute to a more resilient and cost-efficient supply chain for critical phosphorus-containing intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization here is the complete removal of expensive transition metal catalysts and the associated ligands. In traditional cross-coupling, catalyst loading and recovery can account for a significant portion of the COGS. By switching to an organic dye system, the direct material cost is drastically lowered. Additionally, the mild reaction conditions reduce energy overheads, and the simpler workup reduces labor and solvent costs. This creates a leaner manufacturing process that is less sensitive to raw material price volatility.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like aryl hydrazines and triethyl phosphite ensures a stable supply base, unlike specialized organometallic reagents which may have limited suppliers. The robustness of the reaction to air and moisture means that storage and handling requirements are less stringent, reducing the risk of batch failures due to environmental exposure. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery windows for downstream API manufacturers.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with increasingly strict environmental regulations regarding heavy metal discharge. Scaling up photocatalytic reactions is becoming increasingly feasible with modern flow chemistry technologies, allowing for safe and efficient production from kilograms to tons. The reduced toxicity profile of the reagents and the absence of metal waste simplify the environmental impact assessment, facilitating faster regulatory approvals for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential partners.
Q: Why is this metal-free method superior for pharmaceutical intermediates?
A: Traditional methods often rely on palladium or copper catalysts, which leave toxic metal residues requiring expensive removal steps. This patent utilizes an organic dye (Eosin Y/B), ensuring the final aryl phosphonate is free from heavy metal contamination, which is critical for API safety compliance.
Q: What are the typical reaction conditions for this synthesis?
A: The process operates under exceptionally mild conditions: temperatures between 25-45°C, atmospheric pressure (open to air), and visible light irradiation. This contrasts sharply with the high heat and inert atmospheres required by conventional coupling reactions.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the patent demonstrates excellent substrate scope. It successfully synthesizes derivatives containing electron-withdrawing groups like trifluoromethyl and chlorine, as well as electron-donating groups like methoxy and methyl, proving its versatility for complex molecule construction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Phosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free photocatalytic technology for the global pharmaceutical supply chain. Our team of expert process chemists is fully equipped to adapt and optimize this visible-light methodology for your specific aryl phosphonate targets. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities include rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets stringent purity specifications required by top-tier drug developers.
We invite you to collaborate with us to leverage this innovative synthesis route for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data for our existing phosphonate inventory or to discuss route feasibility assessments for novel compounds. Let us help you secure a sustainable and cost-effective supply of high-quality aryl phosphonates for your critical applications.
