Advanced Green Synthesis of Pyridoimidazole Derivatives for Commercial Antitumor Drug Manufacturing
The pharmaceutical industry is constantly seeking efficient, sustainable pathways to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN113149984B discloses a groundbreaking green synthesis method for pyridoimidazole derivatives, a class of nitrogen-containing heterocycles with profound significance in oncology. This technology addresses the critical need for environmentally benign manufacturing processes by utilizing molecular oxygen as a terminal oxidant and inexpensive copper salts as catalysts. The core innovation lies in the direct oxidative coupling of substituted 3-indoleacetic acids with pyridoimidazole compounds, bypassing the multi-step sequences traditionally required to construct these fused ring systems. 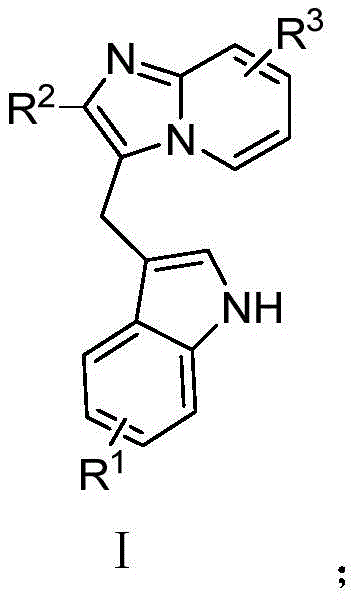 As illustrated in the general structure, the versatility of this method allows for extensive structural diversification, enabling the rapid generation of libraries for structure-activity relationship (SAR) studies. For R&D directors and process chemists, this represents a paradigm shift from linear, waste-intensive syntheses to convergent, atom-economical strategies that align with modern regulatory and sustainability mandates.
As illustrated in the general structure, the versatility of this method allows for extensive structural diversification, enabling the rapid generation of libraries for structure-activity relationship (SAR) studies. For R&D directors and process chemists, this represents a paradigm shift from linear, waste-intensive syntheses to convergent, atom-economical strategies that align with modern regulatory and sustainability mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyridoimidazole frameworks has been plagued by significant synthetic inefficiencies that hinder commercial viability. Traditional routes often rely on the use of stoichiometric amounts of hazardous oxidants, such as hypervalent iodine reagents or heavy metal salts, which generate substantial quantities of toxic waste and pose severe safety risks during scale-up. Furthermore, conventional methodologies frequently necessitate harsh reaction conditions, including extreme temperatures or strongly acidic environments, which can lead to the degradation of sensitive functional groups and result in poor overall yields. The reliance on multi-step protocols not only increases the consumption of organic solvents but also complicates the purification landscape, requiring rigorous chromatographic separations to remove persistent metal residues and side products. These factors collectively drive up the cost of goods sold (COGS) and extend the lead time for producing high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain for potential antitumor drug candidates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a copper-catalyzed decarboxylative coupling strategy that fundamentally simplifies the synthetic landscape. By employing molecular oxygen—an abundant, non-toxic, and cost-effective oxidant—the process eliminates the environmental burden associated with chemical oxidants while maintaining high reaction efficiency. The use of readily available substituted 3-indoleacetic acids and pyridoimidazole precursors allows for a direct, one-step assembly of the target scaffold under mild heating conditions. 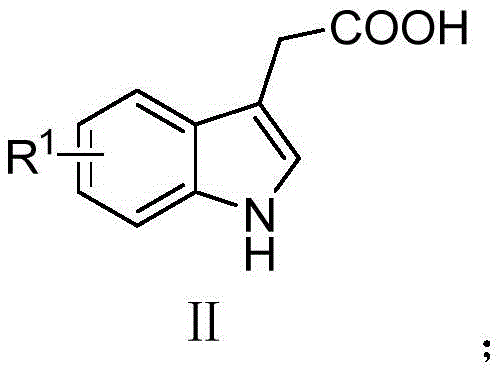
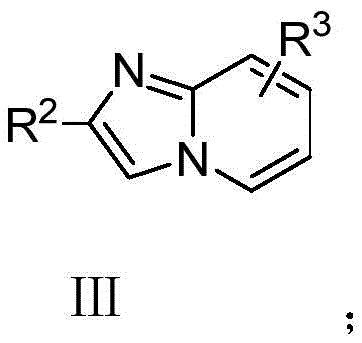 This convergence not only reduces the number of unit operations but also significantly minimizes solvent usage and energy consumption. The robustness of this method is evidenced by its tolerance to a wide range of substituents, including halogens, alkyl groups, and alkoxy moieties, ensuring that diverse chemical space can be explored without compromising reaction performance or product integrity.
This convergence not only reduces the number of unit operations but also significantly minimizes solvent usage and energy consumption. The robustness of this method is evidenced by its tolerance to a wide range of substituents, including halogens, alkyl groups, and alkoxy moieties, ensuring that diverse chemical space can be explored without compromising reaction performance or product integrity.
Mechanistic Insights into Copper-Catalyzed Decarboxylative Coupling
The mechanistic pathway of this transformation offers fascinating insights into the role of the copper catalyst in facilitating radical generation and bond formation. Under the optimized reaction conditions, the copper catalyst, specifically copper acetate, interacts with the substituted 3-indoleacetic acid to promote a single-electron transfer process. This interaction triggers the decarboxylation of the indoleacetic acid moiety, generating a reactive indole-methyl radical intermediate. This radical species is highly nucleophilic and subsequently attacks the electron-deficient position on the substituted pyridoimidazole ring system. The resulting intermediate undergoes a rapid elimination and aromatization sequence to restore aromaticity, yielding the final pyridoimidazole derivative. The choice of solvent plays a pivotal role in stabilizing these transient radical species; polar aprotic solvents like DMF or DMSO are particularly effective in solvating the ionic copper species and facilitating the diffusion of oxygen, thereby sustaining the catalytic cycle.
From an impurity control perspective, the mild nature of this oxidative coupling is advantageous for maintaining high product purity. Unlike harsh oxidative conditions that might over-oxidize sensitive indole or pyridine rings, the copper-oxygen system operates with sufficient selectivity to target the specific C-H activation site adjacent to the carboxyl group. This selectivity minimizes the formation of polymeric byproducts or over-oxidized species that are common in traditional methods. Furthermore, the use of a homogeneous copper catalyst ensures uniform reaction kinetics throughout the batch, reducing the likelihood of localized hot spots that could degrade the product. The subsequent workup involving simple extraction and column chromatography is highly effective because the reaction mixture contains fewer inorganic salts compared to methods using stoichiometric oxidants, streamlining the isolation of the active pharmaceutical ingredient (API) intermediate.
How to Synthesize Pyridoimidazole Derivatives Efficiently
Implementing this synthesis protocol requires careful attention to the molar ratios of reactants and the specific reaction atmosphere to maximize yield and reproducibility. The patent specifies that a molar ratio of indoleacetic acid to pyridoimidazole compound between 1:1 and 1:1.5 is optimal, with a slight excess of the pyridoimidazole partner helping to drive the reaction to completion. The catalyst loading is typically kept low, around 10 mol%, which is economically favorable for large-scale production. The reaction is conducted under an oxygen atmosphere, which can be supplied via an oxygen balloon or a continuous flow of oxygen gas, ensuring a constant supply of the oxidant throughout the 6 to 8-hour heating period. Detailed standardized synthesis steps see the guide below.
- Mix substituted 3-indoleacetic acid and substituted pyridoimidazole compounds in a solvent such as DMF or DMSO.
- Add a copper catalyst, preferably copper acetate, and introduce oxygen as the oxidant into the reaction system.
- Heat the mixture to facilitate the decarboxylative coupling reaction, followed by extraction and purification to obtain the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this green synthesis method translates into tangible strategic advantages that extend beyond mere technical feasibility. The shift towards a catalytic process using earth-abundant metals and atmospheric oxygen drastically alters the cost structure of manufacturing these complex heterocycles. By removing the dependency on precious metal catalysts or exotic oxidizing agents, the raw material costs are significantly reduced, providing a buffer against market volatility in reagent pricing. Moreover, the simplified downstream processing means that less time and resources are dedicated to purification, effectively increasing the throughput of the manufacturing facility without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants like hypervalent iodine salts represents a major cost saving opportunity. In traditional processes, these reagents are not only costly to purchase but also expensive to dispose of safely due to their hazardous nature. By replacing them with oxygen, the process converts a significant cost center into a negligible operational expense. Additionally, the use of copper acetate, a commodity chemical, instead of palladium or rhodium complexes, further lowers the catalyst cost profile. The high atom economy of the decarboxylative coupling ensures that the majority of the starting material mass is incorporated into the final product, minimizing waste disposal fees and maximizing the yield per kilogram of input.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically substituted 3-indoleacetic acids and pyridoimidazoles, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. This abundance ensures a stable supply chain capable of meeting fluctuating demand schedules. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, enhancing batch-to-b consistency. For supply chain planners, this reliability translates to more accurate forecasting and reduced safety stock requirements, as the risk of production delays due to reagent shortages or failed batches is substantially mitigated.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with strong chemical oxidants. The use of oxygen gas can be easily managed in standard stainless steel reactors equipped with gas sparging systems, facilitating a seamless transition from kilo-lab to multi-ton scales. From an environmental compliance standpoint, the process generates minimal hazardous waste, aligning with increasingly stringent global regulations on industrial emissions and effluent discharge. This "green" credential not only simplifies permitting processes but also enhances the corporate sustainability profile, which is becoming a key differentiator in securing contracts with major pharmaceutical partners who prioritize responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the practical aspects of adopting this method for industrial applications. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using oxygen as an oxidant in this synthesis?
A: Using oxygen eliminates the need for expensive and toxic chemical oxidants like hypervalent iodine salts, significantly reducing waste generation and raw material costs while adhering to green chemistry principles.
Q: Why is copper acetate preferred over other copper catalysts for this reaction?
A: Experimental data indicates that copper acetate provides the highest yield compared to other copper salts like bromide or chloride, likely due to its optimal solubility and coordination properties in polar aprotic solvents like DMF.
Q: How does this method simplify the purification process?
A: The reaction produces minimal byproducts under mild conditions, allowing for high-purity isolation through simple extraction and column chromatography, avoiding complex recrystallization or distillation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridoimidazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green synthesis technology in accelerating the development of antitumor therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyridoimidazole derivatives delivered meets the highest international standards. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our dedicated technical team is committed to maintaining the integrity of your IP while optimizing the process for maximum yield.
We invite you to collaborate with us to leverage this advanced synthetic route for your next-generation oncology programs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-purity intermediates and bring your life-saving medications to patients faster and more sustainably.
