Advanced Organophosphine Catalysis for Scalable Nitrogen Heterocycle Production in Pharmaceutical Manufacturing
Advanced Organophosphine Catalysis for Scalable Nitrogen Heterocycle Production in Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex nitrogen-containing scaffolds, which serve as the backbone for countless bioactive molecules. Patent CN1166646C introduces a groundbreaking synthetic strategy that addresses long-standing challenges in the production of nitrogen heterocyclic compounds, specifically piperazines and morpholines. This technology leverages the unique reactivity of electron-poor alkynes reacting with nucleophiles possessing double nucleophilic centers, facilitated by an organophosphine catalyst. Unlike conventional routes that often rely on harsh conditions or precious metal catalysts, this invention offers a streamlined pathway operating under mild, neutral conditions. For R&D directors and process chemists, this represents a significant opportunity to simplify synthetic routes for key intermediates used in antiviral, cardiovascular, and neurological therapies. The versatility of this method allows for the generation of diverse structural analogues by simply varying the alkyne or the nucleophilic partner, providing a powerful tool for library synthesis and process development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
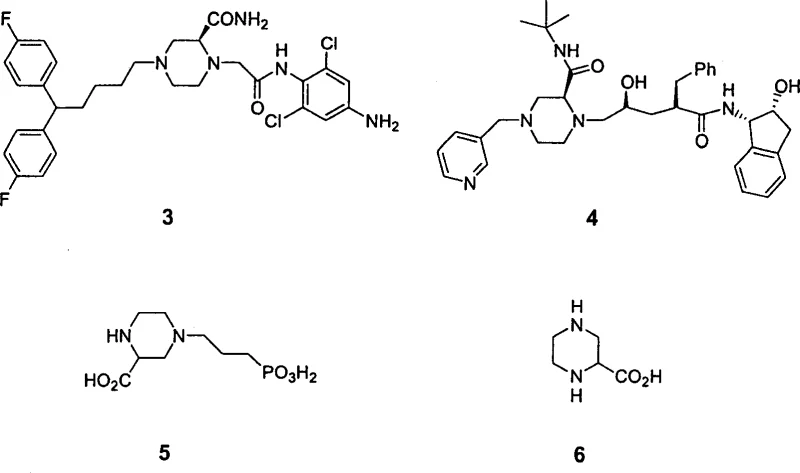
Historically, the synthesis of critical intermediates like 2-piperazine carboxylic acid (Compound 6) has been fraught with inefficiencies and safety concerns. The traditional industrial route typically involves the hydrogenation of aromatic precursors, such as 2-pyrazine carboxylic acid, using heterogeneous catalysts like palladium on carbon (Pd/C). This approach necessitates high-pressure hydrogenation equipment, posing significant safety risks and requiring specialized infrastructure that increases capital expenditure. Furthermore, the precursor 2-pyrazine carboxylic acid itself is often derived from pyrazine through multi-step sequences involving alkylation and oxidation, which are characterized by severe reaction conditions and notoriously low yields. The reliance on heavy metal catalysts also introduces the risk of metal contamination in the final product, necessitating costly and time-consuming purification steps to meet stringent regulatory limits for residual metals in pharmaceutical ingredients. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks in the supply chain for essential drug intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN1166646C circumvents these obstacles by employing a completely different mechanistic paradigm based on organocatalysis. By utilizing electron-poor alkynes, such as substituted propiolates or butynones, and reacting them with readily available diamines or amino alcohols, the process constructs the heterocyclic ring in a single, efficient step. The use of organic phosphines, such as triphenylphosphine or tributylphosphine, as catalysts eliminates the need for transition metals entirely, thereby removing the burden of metal scavenging from the downstream processing. The reaction proceeds smoothly at temperatures ranging from room temperature to 130°C, with optimal results often observed between 60°C and 130°C, significantly reducing energy consumption compared to high-pressure hydrogenation. This mildness not only preserves sensitive functional groups but also minimizes the formation of degradation byproducts, leading to cleaner reaction profiles and simplified isolation procedures. As illustrated in specific embodiments, this approach successfully generates protected piperazine and morpholine derivatives with excellent efficiency.
Mechanistic Insights into Organophosphine-Catalyzed Cyclization
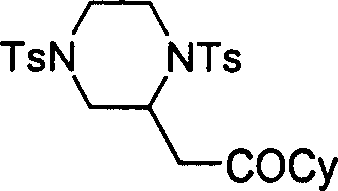
The core of this innovation lies in the activation of the electron-deficient alkyne by the nucleophilic phosphine catalyst, which initiates a cascade of addition reactions. The mechanism likely involves the initial attack of the phosphine on the beta-carbon of the alkyne, generating a zwitterionic intermediate that is highly susceptible to nucleophilic attack. The double-center nucleophile, which contains two reactive sites such as amine groups or an amine and a hydroxyl group, then engages with this activated species. 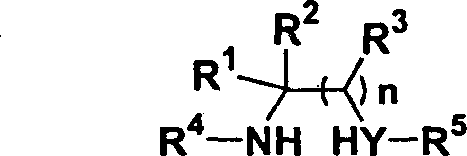 The first nucleophilic center attacks the activated alkyne, followed by an intramolecular cyclization involving the second nucleophilic center to close the ring and release the phosphine catalyst. This catalytic cycle ensures that only stoichiometric amounts of the valuable alkyne and nucleophile are consumed, while the catalyst turns over multiple times. The ability to tune the electronic properties of the alkyne via the electron-withdrawing group E (such as ester or acyl groups) allows for precise control over the reaction rate and regioselectivity. This level of mechanistic control is crucial for minimizing side reactions, such as polymerization of the alkyne or oligomerization of the nucleophile, which are common pitfalls in uncatalyzed thermal reactions.
The first nucleophilic center attacks the activated alkyne, followed by an intramolecular cyclization involving the second nucleophilic center to close the ring and release the phosphine catalyst. This catalytic cycle ensures that only stoichiometric amounts of the valuable alkyne and nucleophile are consumed, while the catalyst turns over multiple times. The ability to tune the electronic properties of the alkyne via the electron-withdrawing group E (such as ester or acyl groups) allows for precise control over the reaction rate and regioselectivity. This level of mechanistic control is crucial for minimizing side reactions, such as polymerization of the alkyne or oligomerization of the nucleophile, which are common pitfalls in uncatalyzed thermal reactions.
From an impurity control perspective, the neutral nature of the reaction conditions is a distinct advantage over acid or base-catalyzed alternatives. Acidic conditions can lead to the deprotection of sensitive groups like tosylates or the hydrolysis of ester functionalities, while basic conditions might promote elimination reactions or racemization of chiral centers. By maintaining a neutral pH throughout the synthesis, the integrity of the protecting groups (e.g., tosyl, benzyl) and the stereochemistry of the starting materials are preserved. This results in a crude product with a significantly reduced impurity profile, facilitating easier purification via standard techniques like recrystallization or column chromatography. The patent data indicates that yields generally range from 66% to 99%, demonstrating the robustness of this method across a variety of substrates. For quality assurance teams, this consistency translates to more predictable batch outcomes and reduced waste generation, aligning with green chemistry principles.
How to Synthesize Nitrogen Heterocycles Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The process begins with the selection of an appropriate solvent; while the patent lists a wide array of options including benzene, toluene, THF, and DMF, acetonitrile is frequently highlighted for its effectiveness in dissolving both the catalyst and the nucleophile. It is imperative to use dried solvents to prevent the hydrolysis of the electron-poor alkyne or the deactivation of the phosphine catalyst by moisture. The molar ratio of reactants is another critical variable, with the patent recommending a ratio of electron-poor alkyne to nucleophile to catalyst of approximately 0.8-1.2:1:0.1-0.3. Deviating significantly from these ratios may result in incomplete conversion or difficulty in separating excess starting materials. For detailed operational protocols, including specific workup procedures and purification strategies, please refer to the standardized guide below.
- Prepare the reaction mixture by dissolving an organic phosphine catalyst (e.g., triphenylphosphine) and a double-center nucleophile (e.g., N,N'-ditosylethylenediamine) in a dry organic solvent such as acetonitrile.
- Heat the solution to a temperature between 60°C and 130°C to ensure complete dissolution and activation of the nucleophilic species before introducing the alkyne substrate.
- Add the electron-poor alkyne (e.g., substituted propiolate or butynone) to the reaction system, maintain heating until TLC indicates consumption of starting materials, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this organophosphine-catalyzed technology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift away from precious metal catalysts like palladium directly impacts the raw material cost structure, as organic phosphines are significantly cheaper and more abundant than noble metals. Furthermore, the elimination of high-pressure hydrogenation steps reduces the dependency on specialized reactor vessels, allowing for production in standard glass-lined or stainless steel reactors that are more commonly available in multipurpose chemical plants. This flexibility enhances supply chain resilience by enabling manufacturing across a broader network of contract manufacturing organizations (CMOs) without the need for costly facility upgrades. The mild reaction conditions also contribute to improved safety profiles, potentially lowering insurance premiums and reducing the regulatory burden associated with handling hazardous high-pressure gases.
- Cost Reduction in Manufacturing: The economic impact of replacing heavy metal catalysts with organic phosphines cannot be overstated, as it removes the entire unit operation dedicated to metal scavenging and recovery. Traditional processes often require specialized resins or filtration steps to reduce metal residues to parts-per-million levels, which adds both material costs and processing time. By avoiding these steps entirely, the overall cost of goods sold (COGS) is substantially reduced. Additionally, the high yields reported (up to 99%) mean that less raw material is wasted per kilogram of product produced, further driving down the effective cost per unit. The simplicity of the workup, often involving just solvent removal and chromatography or recrystallization, minimizes labor hours and utility consumption, contributing to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted alkynes and protected diamines, are commodity chemicals that are widely available from multiple global suppliers. This contrasts with specialized aromatic precursors that may have limited sources or long lead times. By utilizing a platform technology that accepts a broad range of commercially available building blocks, manufacturers can mitigate the risk of supply disruptions caused by single-source dependencies. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures due to specification drifts. This reliability ensures a steady flow of intermediates to downstream API synthesis lines, preventing costly production stoppages.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of exothermic hazards associated with high-pressure hydrogenation and the use of non-toxic organic catalysts. The reaction can be safely performed in batch or continuous flow modes, offering flexibility for different production volumes. From an environmental standpoint, the reduction in heavy metal waste simplifies effluent treatment and disposal, helping facilities meet increasingly stringent environmental regulations. The use of common organic solvents that can be easily recovered and recycled further enhances the sustainability profile of the process. These factors combined make the technology highly attractive for companies aiming to reduce their environmental footprint while maintaining high production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrogen heterocycle synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding the nuances of catalyst selection, substrate scope, and downstream processing is essential for successfully integrating this technology into existing manufacturing workflows. We encourage technical teams to review these details closely when evaluating the feasibility of this route for their specific target molecules.
Q: What are the primary advantages of this organophosphine method over traditional hydrogenation?
A: Unlike traditional methods that require harsh conditions and expensive heavy metal catalysts like Pd/C for hydrogenating pyrazine carboxylic acid, this patented process operates under mild, neutral conditions (60-130°C) using inexpensive organic phosphines, eliminating the need for high-pressure equipment and complex metal removal steps.
Q: Can the synthesized heterocycles be converted into active pharmaceutical ingredients?
A: Yes, the patent explicitly demonstrates that products like P13 can be readily transformed into critical drug intermediates such as 2-piperazine carboxylic acid (Compound 6), which serves as a precursor for major therapeutics including Draflazine, Crixivan, and CPP.
Q: What types of solvents are compatible with this catalytic system?
A: The method exhibits broad solvent tolerance, successfully utilizing both polar and non-polar dried solvents including acetonitrile, N,N-dimethylformamide (DMF), toluene, tetrahydrofuran, and dichloromethane, allowing for flexible process optimization based on solubility requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen Heterocycle Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthetic routes in the competitive landscape of pharmaceutical intermediate production. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are well-equipped to leverage the organophosphine catalysis technology described in CN1166646C to deliver high-purity nitrogen heterocyclic compounds that meet your stringent purity specifications. Our state-of-the-art rigorous QC labs are capable of performing comprehensive impurity profiling and residual metal analysis, guaranteeing that every batch conforms to the highest industry standards. Whether you require custom synthesis of novel analogues or large-scale supply of established intermediates, our infrastructure is designed to support your project goals with speed and precision.
We invite you to collaborate with us to explore how this advanced synthetic methodology can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data for our available inventory or to discuss route feasibility assessments for your target molecules. Let us be your partner in driving innovation and efficiency in your chemical supply chain.
