Scaling Diindolylmethane Derivative Production with Acid-Modified Zeolite Catalysis
Scaling Diindolylmethane Derivative Production with Acid-Modified Zeolite Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for synthesizing critical intermediates like diindolylmethane derivatives, which serve as pivotal scaffolds in anticancer agents and functional materials. A groundbreaking approach detailed in patent CN102153500B introduces a continuous flow synthesis strategy that fundamentally shifts the production paradigm from inefficient batch processing to high-throughput fixed-bed reactor technology. This innovation leverages acid-modified H-beta molecular sieves as a heterogeneous catalyst, offering a sustainable alternative to traditional Lewis acid systems. By integrating this advanced catalytic system, manufacturers can achieve superior product quality while drastically simplifying the downstream purification workflow. The technical implications of this patent extend far beyond mere yield improvements; it represents a strategic alignment with green chemistry principles that modern supply chains demand. For R&D directors and procurement specialists alike, understanding the mechanistic advantages of this zeolite-catalyzed route is essential for optimizing the sourcing of reliable diindolylmethane derivative suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diindolylmethane derivatives has been plagued by significant operational inefficiencies and environmental hazards inherent to batch processing techniques. Traditional protocols often rely on homogeneous Lewis acids such as lithium perchlorate (LiClO4) or corrosive mineral acids, which necessitate complex quenching and neutralization steps that generate substantial aqueous waste streams. Furthermore, many established methods utilize volatile and toxic chlorinated solvents like methylene dichloride, posing severe health risks to operators and complicating regulatory compliance regarding solvent residues in final API intermediates. Alternative approaches employing ionic liquids, while effective in the lab, suffer from prohibitive costs and difficult recovery processes that render them economically unviable for large-scale commercial manufacturing. These batch methods typically exhibit long reaction times ranging from one to five hours, coupled with inconsistent product quality due to poor heat and mass transfer characteristics. Consequently, the cumulative cost of waste disposal, solvent recovery, and extended reactor occupancy time creates a significant burden on the overall cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN102153500B utilizes a continuous fixed-bed reactor system packed with acid-modified H-beta zeolite, effectively eliminating the drawbacks of homogeneous catalysis. This novel approach allows for the seamless mixing of indole and substituted benzaldehydes in environmentally benign lower alcohols, such as ethanol or methanol, which are pumped through the catalyst bed at controlled flow rates between 0.5 and 2 mL/min. The heterogeneous nature of the zeolite catalyst ensures that the reaction proceeds with high selectivity and minimal byproduct formation, while the continuous flow regime guarantees uniform residence time and temperature control. This transition to continuous processing not only shortens the overall process time significantly but also facilitates the easy separation of the catalyst from the reaction mixture, allowing for reuse over extended periods exceeding 100 hours. The implementation of this technology enables the commercial scale-up of complex pharmaceutical intermediates with unprecedented efficiency and safety profiles.
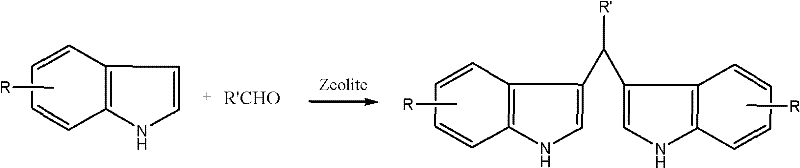
Mechanistic Insights into Zeolite-Catalyzed Electrophilic Substitution
The core of this technological advancement lies in the precise modification of the H-beta molecular sieve, which serves as a solid superacid catalyst driving the electrophilic substitution reaction between the indole nucleus and the carbonyl carbon of the aldehyde. The patent specifies that treating the H-beta sieve with acid solutions—ranging from mineral acids like hydrochloric and sulfuric acid to organic acids like oxalic and tartaric acid—enhances the surface acidity and optimizes the pore structure for reactant diffusion. This acid modification step, involving stirring at 30-55°C followed by calcination at 500-600°C, generates a high density of active Bronsted acid sites that activate the aldehyde towards nucleophilic attack by the electron-rich C3 position of the indole ring. The use of a molar excess of indole (3.5 to 5 equivalents relative to the aldehyde) is a critical mechanistic parameter that suppresses the formation of polymeric byproducts and drives the equilibrium towards the desired bis-indolyl methane product. By maintaining reaction temperatures between 80°C and 120°C within the fixed bed, the system achieves an optimal balance between reaction kinetics and catalyst stability, ensuring that the delicate indole structure is preserved without undergoing thermal degradation.
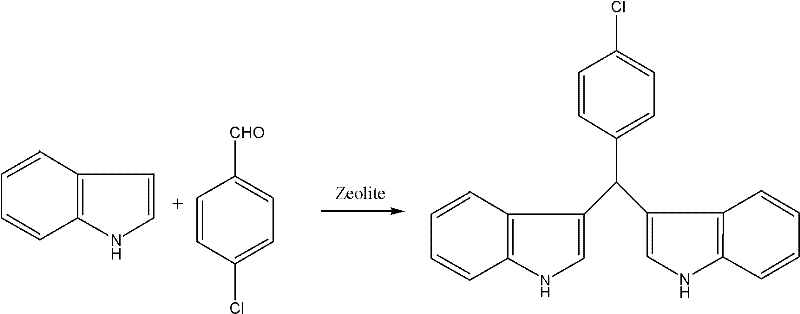
Furthermore, the impurity profile of the resulting diindolylmethane derivatives is exceptionally clean due to the shape-selective properties of the zeolite framework, which restricts the formation of bulky side products that cannot fit within the catalyst pores. This intrinsic selectivity reduces the burden on downstream purification units, such as crystallization or chromatography, thereby lowering the overall cost of goods sold. The versatility of this catalytic system is evidenced by its ability to accommodate various substituents on the benzaldehyde ring, including electron-withdrawing groups like chlorine and nitro groups, without compromising yield or reaction rate. For instance, the synthesis of 4-chlorophenyl-diindole methyl hydride proceeds with yields exceeding 94%, demonstrating the robustness of the catalyst against deactivation by halogenated substrates. This level of control over the reaction pathway is paramount for R&D teams focused on developing high-purity OLED material or pharmaceutical intermediates where trace impurities can dictate the success of clinical trials.
How to Synthesize Diindolylmethane Derivatives Efficiently
Implementing this continuous synthesis route requires careful attention to catalyst preparation and reactor engineering to maximize throughput and product consistency. The process begins with the activation of the H-beta molecular sieve, followed by its packing into a tubular reactor capable of withstanding temperatures up to 120°C. Operators must ensure that the feed solution, comprising indole and the chosen aldehyde dissolved in ethanol or methanol, is degassed and pumped at a steady rate to prevent channeling within the catalyst bed. The patent provides a clear roadmap for scaling this reaction from laboratory gram-scale experiments to multi-kilogram production campaigns, emphasizing the simplicity of the workup procedure which involves merely concentrating the effluent and adding water to precipitate the pure product. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Prepare the catalyst by treating H-beta molecular sieve with acid solution (0.8-1.2 mol/L), followed by drying and calcination at 500-600°C.
- Load the modified catalyst into a fixed-bed reactor and pump a mixture of indole and aldehyde (molar ratio 3.5-5: 1) in lower alcohol at 0.5-2 mL/min.
- Collect the effluent, concentrate, add water to precipitate the product, then filter and dry to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this zeolite-catalyzed continuous flow process offers transformative benefits for procurement managers and supply chain heads tasked with securing reliable sources of fine chemical intermediates. The elimination of expensive and hazardous reagents such as ionic liquids and perchlorate salts directly translates to substantial cost savings in raw material procurement and waste management budgets. Moreover, the use of common, low-toxicity solvents like ethanol simplifies the logistics of solvent storage and recovery, reducing the regulatory burden associated with handling chlorinated hydrocarbons. The continuous nature of the process ensures a steady, uninterrupted output of material, mitigating the risks of supply disruptions that are common with batch-based manufacturing where equipment turnaround time can be significant. This reliability is crucial for maintaining the continuity of downstream drug substance production schedules and meeting tight delivery windows for global clients.
- Cost Reduction in Manufacturing: The shift to a heterogeneous catalyst system removes the need for costly catalyst removal and neutralization steps, significantly streamlining the production workflow. By avoiding the use of precious metal catalysts or expensive ionic liquids, the variable cost per kilogram of product is drastically reduced, enhancing the overall margin profile for high-volume contracts. Additionally, the extended lifetime of the zeolite catalyst, which remains active for over 100 hours of continuous operation, minimizes the frequency of catalyst replacement and the associated downtime costs. This economic efficiency makes the process highly competitive for the commercial scale-up of complex polymer additives and pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: Continuous flow manufacturing inherently offers greater predictability in output rates compared to batch processing, allowing supply chain planners to forecast inventory levels with higher accuracy. The robustness of the acid-modified zeolite against poisoning by substrate impurities ensures that the production line remains stable even when facing variations in raw material quality. This resilience reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and emergency orders. Furthermore, the simplified process design reduces the dependency on specialized operator skills, making it easier to replicate the manufacturing process across different facilities if redundancy is required.
- Scalability and Environmental Compliance: The fixed-bed reactor configuration is inherently scalable, allowing for capacity expansion simply by increasing the reactor diameter or numbering up parallel units without re-optimizing the chemistry. This scalability is paired with a significantly reduced environmental footprint, as the process generates less hazardous waste and consumes less energy per unit of product due to improved thermal efficiency. Compliance with increasingly stringent environmental regulations is thus achieved not through end-of-pipe treatments, but through the intrinsic design of the synthesis route itself. This proactive approach to sustainability aligns perfectly with the corporate social responsibility goals of major multinational corporations seeking green chemistry partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this method for their specific product portfolios. We encourage technical teams to review these points closely when assessing potential process transfers or vendor qualifications.
Q: What is the operational lifetime of the acid-modified zeolite catalyst?
A: According to the patent data, the acid-modified H-beta zeolite catalyst demonstrates exceptional stability, capable of continuous operation for over 100 hours without significant loss of activity, ensuring consistent production cycles.
Q: How does this method improve environmental compliance compared to traditional routes?
A: This process replaces hazardous solvents like methylene chloride and expensive, difficult-to-recover ionic liquids with lower alcohols like ethanol. Additionally, the heterogeneous catalyst eliminates the need for complex post-reaction neutralization and heavy metal removal steps.
Q: Can this continuous flow method handle substituted benzaldehydes effectively?
A: Yes, the method is highly versatile. Experimental data confirms high yields (over 93%) for various substrates including 4-chlorobenzaldehyde and 2-nitrobenzaldehyde, demonstrating robust tolerance to electronic and steric variations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diindolylmethane Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global fine chemicals market. Our team of expert chemists has extensively evaluated the zeolite-catalyzed continuous flow process described in CN102153500B and possesses the technical capability to implement this route on an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to verify the identity and purity of every batch, guaranteeing that the diindolylmethane derivatives we supply are free from the impurities associated with older batch technologies.
We invite procurement leaders and R&D directors to collaborate with us to leverage these technological advancements for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient synthesis route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your unique molecular requirements. Let us help you optimize your supply chain and accelerate your time to market with our superior manufacturing capabilities.
