Advanced Oxfendazole Manufacturing: Reducing Impurity B via Controlled Oxidation
Advanced Oxfendazole Manufacturing: Reducing Impurity B via Controlled Oxidation
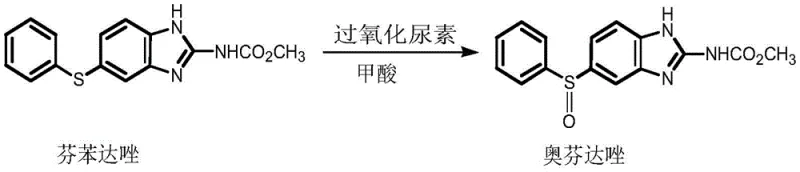
In the competitive landscape of veterinary pharmaceutical intermediates, the ability to control impurity profiles is the definitive marker of a superior manufacturing process. The recent disclosure in patent CN110903247B introduces a transformative approach to the synthesis of oxfendazole, a broad-spectrum anthelmintic agent widely used in animal health. This patent details a preparation method that fundamentally alters the oxidation step by replacing traditional liquid hydrogen peroxide with urea peroxide. For R&D directors and procurement specialists alike, this shift represents a critical opportunity to enhance product quality while mitigating the safety risks associated with violent exothermic reactions. By leveraging this technology, manufacturers can achieve a significant reduction in Oxfendazole Impurity B, a persistent peroxidation byproduct that often complicates regulatory compliance and downstream purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
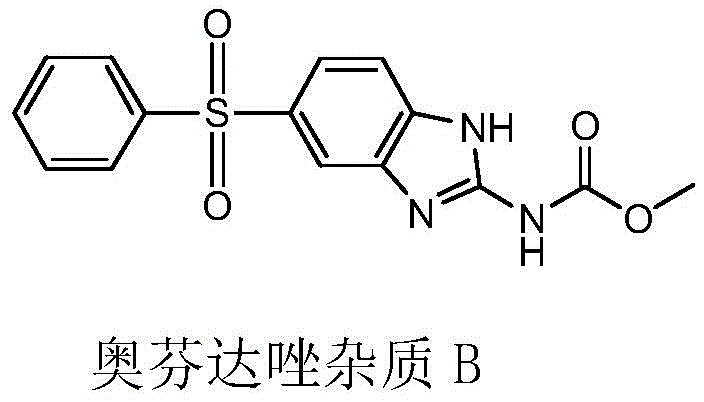
Traditionally, the industrial synthesis of oxfendazole from fenbendazole has relied heavily on liquid hydrogen peroxide as the primary oxidant. While chemically effective, this conventional route suffers from inherent thermodynamic instability. The oxidation of the sulfide group to the sulfoxide is highly exothermic, and without precise control, the reaction can easily proceed to the sulfone stage or generate complex peroxidation byproducts. The most notorious of these is Oxfendazole Impurity B, a structural analog that arises from over-oxidation or radical side reactions. In standard processes, the rapid release of oxygen from liquid peroxide creates localized hot spots within the reactor, driving these unwanted side reactions and resulting in a crude product burdened with difficult-to-remove impurities. This not only lowers the overall yield but also necessitates extensive and costly recrystallization steps to meet pharmacopoeial standards.
The Novel Approach
The methodology outlined in CN110903247B offers a sophisticated solution to these thermal challenges by utilizing urea peroxide (carbamide peroxide) as a solid-state oxidant. Unlike its liquid counterpart, urea peroxide decomposes gradually in the presence of formic acid, releasing atomic oxygen in a controlled, steady stream. This "slow-release" mechanism effectively dampens the reaction kinetics, preventing the sharp temperature spikes that drive the formation of Impurity B. The result is a much milder reaction environment that favors the selective formation of the sulfoxide bond while suppressing over-oxidation. Furthermore, the use of formic acid as both solvent and catalyst enhances the solubility of the starting material, ensuring a homogeneous reaction mixture that further contributes to consistent product quality and reduced batch-to-batch variability.
Mechanistic Insights into Urea Peroxide-Mediated Oxidation
To fully appreciate the technical merit of this process, one must examine the mechanistic role of urea peroxide in acidic media. Upon dissolution in formic acid, the urea-hydrogen peroxide adduct dissociates, making hydrogen peroxide available for reaction. However, the key differentiator is the rate of availability. The hydrogen bonding network within the urea peroxide crystal lattice requires energy to break, which naturally throttles the release of the oxidant. In the context of electrophilic oxidation of the sulfur atom in fenbendazole, this ensures that the concentration of active oxidizing species never exceeds the threshold required for rapid, uncontrolled propagation. Consequently, the reaction proceeds through a stable transition state that minimizes the generation of free radicals responsible for forming the peroxidation impurity shown in the structural analysis. This kinetic control is paramount for achieving the high purity levels demanded by modern veterinary drug regulations.
Furthermore, the workup procedure described in the patent leverages this chemical stability to simplify purification. By terminating the reaction with sodium sulfite, any residual oxidant is safely quenched without generating gas evolution hazards common in peroxide quenching. The subsequent decolorization with activated carbon and pH-adjusted crystallization allows for the efficient removal of trace organic contaminants. The patent data indicates that this refined workflow consistently delivers oxfendazole with Impurity B levels well below 1%, often hovering around 0.92% to 0.96%, and unreacted fenbendazole (Impurity A) below 0.3%. This level of control demonstrates a robust understanding of the interplay between reaction thermodynamics and crystallization dynamics, providing a reliable blueprint for high-quality API intermediate production.
How to Synthesize Oxfendazole Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to replicate the success seen in the patent examples. The process begins with the dissolution of fenbendazole in formic acid, followed by the controlled addition of the urea peroxide solution. Maintaining the reaction temperature between 25°C and 45°C during the addition phase is critical to preserving the selectivity of the oxidation. Following the reaction, the quenching and crystallization steps are designed to maximize recovery while locking in the purity profile. For detailed operational parameters and specific equipment recommendations, please refer to the standardized synthesis guide below.
- Dissolve fenbendazole in formic acid at 45-60°C and cool to 25-45°C before adding urea peroxide solution.
- Slowly add urea peroxide aqueous solution over 2-4 hours while maintaining temperature control to ensure mild oxidation.
- Terminate reaction with sodium sulfite, decolorize with activated carbon, and crystallize by adjusting pH to 4.5-5.0.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this urea peroxide-based oxidation method translates directly into tangible operational efficiencies and risk mitigation. The shift away from hazardous liquid peroxides reduces the regulatory burden associated with the storage and transport of dangerous goods, simplifying logistics and lowering insurance premiums. Moreover, the enhanced safety profile of the reaction minimizes the risk of thermal runaway incidents, protecting both personnel and capital equipment. From a cost perspective, the significant reduction in Impurity B means that less material is lost to mother liquors during purification, effectively increasing the usable yield per batch without requiring additional raw material inputs. This efficiency gain is a powerful driver for margin improvement in high-volume manufacturing scenarios.
- Cost Reduction in Manufacturing: The elimination of violent exotherms reduces the need for expensive cryogenic cooling systems and specialized pressure-rated reactors. By operating under milder conditions, facilities can utilize standard glass-lined or stainless steel equipment, significantly lowering capital expenditure requirements. Additionally, the higher selectivity of the reaction reduces the consumption of solvents and adsorbents needed for downstream purification, leading to substantial savings in variable production costs and waste disposal fees.
- Enhanced Supply Chain Reliability: Urea peroxide is a stable, solid commodity chemical that is easier to source and store than concentrated liquid hydrogen peroxide, which can degrade over time. This stability ensures a consistent supply of high-quality oxidant, reducing the risk of batch failures due to reagent variability. The robustness of the process also means that production schedules are less likely to be disrupted by safety incidents or extended reaction times caused by thermal management issues, ensuring on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The mild nature of this oxidation process makes it inherently scalable from pilot plant to commercial tonnage without the exponential increase in safety risks often seen with peroxide chemistry. The reduced formation of toxic byproducts and the use of formic acid, which can be recovered or neutralized relatively easily, aligns with increasingly stringent environmental regulations. This green chemistry approach not only future-proofs the manufacturing site against regulatory changes but also enhances the corporate sustainability profile, a key factor for multinational partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel oxidation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for upgrading existing production lines.
Q: Why is urea peroxide preferred over hydrogen peroxide for oxfendazole synthesis?
A: Urea peroxide acts as a solid carrier for hydrogen peroxide, releasing active oxygen slowly and steadily. This prevents the violent exothermic reactions associated with liquid hydrogen peroxide, thereby significantly reducing the formation of over-oxidized byproducts like Impurity B.
Q: What is the typical purity profile achieved with this new method?
A: According to patent data, this method consistently achieves oxfendazole with Impurity B levels below 1% (often around 0.92-0.96%) and unreacted fenbendazole (Impurity A) below 0.3%, meeting European Pharmacopoeia standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for domestic industrial production due to its milder reaction conditions, higher safety profile, and simplified post-reaction processing compared to traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxfendazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes like the urea peroxide oxidation method requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of this oxidation chemistry is maintained at every volume. Our facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of oxfendazole meets the highest international standards, including the low impurity profiles achievable through this patented method.
We invite you to collaborate with us to leverage this technology for your veterinary drug portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global anthelmintic market.
