Advanced Blue Organic Semiconductor Materials Based on 2,6-Di-Tert-Butylanthracene for High-Efficiency OLED Displays
The rapid evolution of the organic light-emitting diode (OLED) industry has placed immense pressure on material scientists to solve the persistent "blue gap" challenge, where blue emitters historically lag behind red and green counterparts in efficiency and operational stability. Patent CN112239414B addresses this critical bottleneck by disclosing a novel class of blue organic semiconductor materials based on a 2,6-di-tert-butylanthracene core. This technology represents a significant leap forward in molecular engineering, utilizing steric hindrance and aggregation-induced emission (AIE) mechanisms to overcome traditional fluorescence quenching issues. For R&D directors and procurement specialists in the display sector, this patent offers a robust pathway to high-purity OLED material production that balances performance with manufacturability. The disclosed compounds exhibit tunable energy levels and exceptional solid-state quantum yields, making them prime candidates for next-generation display panels and lighting applications where color purity and longevity are paramount.
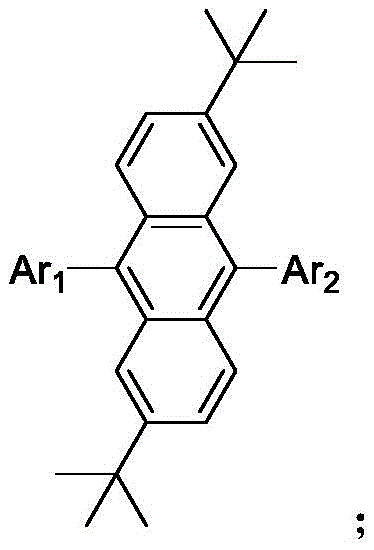
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anthracene-based blue emitters have long suffered from the Aggregation-Caused Quenching (ACQ) effect, a phenomenon where the planar rigid structure of the anthracene nucleus leads to strong intermolecular pi-pi interactions in the solid state. When these molecules aggregate in a thin film, their fluorescence is severely quenched, resulting in low external quantum efficiency (EQE) and poor device performance. Furthermore, conventional synthesis routes often involve harsh conditions or complex purification steps to achieve the necessary purity levels for electronic applications, which drives up costs and complicates supply chain logistics. The inability to effectively decouple molecular emission from aggregation effects has forced manufacturers to rely on complex host-guest doping systems, adding layers of complexity to device fabrication and increasing the risk of phase separation over time.
The Novel Approach
The innovation presented in this patent fundamentally alters the molecular landscape by introducing bulky tert-butyl groups at the 2 and 6 positions of the anthracene core. This strategic modification creates significant steric hindrance that physically prevents the molecules from packing too closely together, thereby suppressing the non-radiative decay pathways associated with ACQ. Additionally, the patent describes the attachment of various functional regulatory groups (Ar1 and Ar2) at the 9 and 10 positions, which can be tailored to introduce AIE characteristics. This dual approach ensures that the material not only maintains high-efficiency blue emission in solution but actually enhances its luminous efficiency in the solid film state. The result is a material system capable of achieving high-performance non-doped OLED devices, simplifying the overall device architecture while delivering superior brightness and stability.
Mechanistic Insights into Suzuki Cross-Coupling and Functionalization
The synthetic strategy relies on a modular three-step sequence that allows for precise control over the final molecular structure. The process begins with a Friedel-Crafts alkylation, where anthracene reacts with tert-butyl alcohol in trifluoroacetic acid (TFA) to install the critical steric bulk. This is followed by a selective bromination step using liquid bromine in dichloromethane, which activates the 9 and 10 positions for subsequent cross-coupling. The final and most versatile step is the Suzuki-Miyaura cross-coupling reaction, catalyzed by tetrakis(triphenylphosphine)palladium [Pd(PPh3)4] in the presence of potassium carbonate. This palladium-catalyzed transformation enables the attachment of diverse aryl or heteroaryl boronic acids, allowing chemists to fine-tune the HOMO-LUMO energy levels and emission wavelengths without altering the core stability provided by the tert-butyl groups.
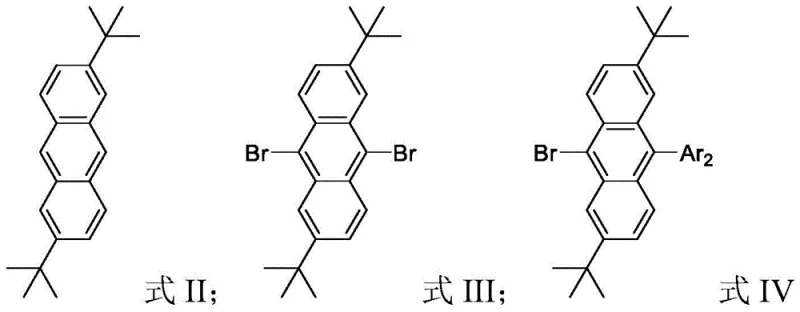
Impurity control is rigorously managed through the selection of mild reaction conditions and effective workup procedures. For instance, the bromination step is conducted at room temperature to prevent poly-bromination side reactions, ensuring high selectivity for the 9,10-dibromo intermediate. The subsequent Suzuki coupling utilizes a biphasic solvent system of toluene, ethanol, and water, which facilitates the dissolution of both organic substrates and inorganic bases while allowing for easy separation of the palladium catalyst residues. The patent reports isolated yields ranging from 60% to 92% across different derivatives, demonstrating the robustness of this catalytic cycle. By avoiding extreme temperatures or pressures, the process minimizes thermal degradation of the sensitive anthracene core, ensuring that the final electronic grade material meets the stringent purity specifications required for commercial OLED manufacturing.
How to Synthesize 2,6-Di-tert-butylanthracene Derivatives Efficiently
The preparation of these high-performance blue emitters follows a logical progression from commodity chemicals to specialized electronic materials. The initial alkylation establishes the steric framework, while the bromination provides the reactive handles necessary for diversification. The final coupling steps are where the specific optical properties are encoded into the molecule. Detailed operational parameters, including molar ratios and solvent compositions, are optimized to maximize yield and minimize waste. For a comprehensive guide on the specific stoichiometry and purification techniques required for laboratory or pilot-scale production, please refer to the standardized synthesis protocol below.
- Perform Friedel-Crafts alkylation of anthracene with tert-butyl alcohol in trifluoroacetic acid (TFA) under reflux to obtain 2,6-di-tert-butylanthracene.
- Conduct bromination of the alkylated anthracene using liquid bromine in dichloromethane (DCM) at room temperature to yield the 9,10-dibromo intermediate.
- Execute Suzuki cross-coupling reaction between the dibromo intermediate and arylboronic acids using Pd(PPh3)4 catalyst in a toluene/ethanol/water solvent system at 110°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthetic route outlined in this patent offers distinct advantages over legacy methods that rely on scarce or hazardous reagents. The starting materials, anthracene and tert-butyl alcohol, are commodity chemicals available in bulk quantities from multiple global suppliers, reducing the risk of raw material shortages. The reliance on standard palladium catalysis rather than exotic transition metals means that the process can be easily scaled using existing infrastructure in fine chemical manufacturing plants. This compatibility with established industrial practices significantly lowers the barrier to entry for commercial production, ensuring a stable and continuous supply of high-quality blue emitters for downstream device manufacturers.
- Cost Reduction in Manufacturing: The elimination of complex doping requirements in the final device architecture translates to substantial cost savings in panel fabrication. By utilizing materials that perform efficiently in non-doped configurations, manufacturers can reduce the number of deposition layers and simplify the vacuum evaporation process. Furthermore, the high reaction yields reported in the patent (up to 92% for certain derivatives) indicate an atom-economical process that minimizes raw material waste. The use of recyclable solvents like toluene and ethanol further contributes to a lower cost of goods sold (COGS) compared to processes requiring chlorinated solvents or cryogenic conditions.
- Enhanced Supply Chain Reliability: The robustness of the Suzuki coupling reaction ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term supply contracts. Since the synthesis does not depend on proprietary catalysts or unstable intermediates, the risk of production delays due to reagent instability is significantly mitigated. The ability to synthesize both symmetric and asymmetric derivatives using the same core intermediate provides flexibility; if demand shifts for a specific emission wavelength, the production line can be adapted quickly without requiring a complete overhaul of the upstream synthesis of the anthracene backbone.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (reflux conditions around 110°C) and atmospheric pressure, which simplifies the engineering requirements for large-scale reactors. This inherently safer profile reduces the capital expenditure needed for specialized high-pressure equipment. Additionally, the aqueous workup steps facilitate the removal of inorganic salts and metal residues, aiding in the production of ultra-high purity materials while generating waste streams that are easier to treat and dispose of in compliance with environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this blue organic semiconductor technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and process feasibility.
Q: How does the 2,6-di-tert-butylanthracene core improve OLED performance?
A: The introduction of bulky tert-butyl groups at the 2 and 6 positions effectively inhibits close molecular packing and strong pi-pi interactions. This structural modification mitigates the Aggregation-Caused Quenching (ACQ) effect common in planar anthracene derivatives, thereby significantly enhancing solid-state fluorescence quantum yield and device stability.
Q: What is the scalability of the Suzuki coupling process described in the patent?
A: The synthesis utilizes standard palladium-catalyzed Suzuki cross-coupling conditions with readily available reagents like potassium carbonate and tetrakis(triphenylphosphine)palladium. The reaction proceeds in a mixed solvent system of toluene, ethanol, and water at moderate temperatures (110°C), making it highly amenable to kilogram-scale manufacturing without requiring exotic high-pressure equipment.
Q: Can these materials be used in non-doped OLED devices?
A: Yes, the patent explicitly demonstrates that these materials possess excellent film-forming properties and high solid-state luminous efficiency. Specific examples, such as compound DPAC-TABI, achieved a maximum external quantum efficiency of 5.81% in non-doped devices, indicating they are suitable for simplified device architectures that eliminate the need for complex host-guest doping systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Blue OLED Material Supplier
As the demand for high-resolution and energy-efficient displays continues to surge, the need for reliable sources of advanced organic electronic materials has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in organic synthesis to deliver high-purity OLED materials that meet the rigorous standards of global display manufacturers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from the R&D phase through to mass manufacturing. With our stringent purity specifications and rigorous QC labs, we guarantee that every batch of 2,6-di-tert-butylanthracene derivatives delivered meets the exacting requirements for defect-free OLED panel production.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development cycles. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and device architecture. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and innovation in your next-generation display projects.
