Scalable Synthesis of 4-Tert-Butyl-2-Acylaminothiazole Derivatives for Oncology Applications
Introduction to Novel Thiazole-Based Anticancer Intermediates
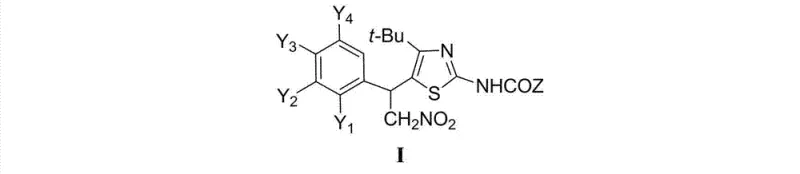
The pharmaceutical landscape is constantly evolving with the discovery of new heterocyclic scaffolds that offer potent biological activity. Patent CN102964312A introduces a significant advancement in this field by disclosing a series of 4-tert-butyl-5-(1-aryl-2-nitroethyl)-2-acylaminothiazole derivatives and their salts. These compounds represent a sophisticated class of pharmaceutical intermediates designed specifically for the development of next-generation anticancer agents. The core innovation lies in the strategic combination of a sterically bulky tert-butyl group at the 4-position of the thiazole ring and a functionalized nitroethyl side chain at the 5-position. This unique structural arrangement not only enhances the metabolic stability of the molecule but also provides versatile handles for further medicinal chemistry optimization. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this chemical space is crucial for driving innovation in oncology drug discovery.
The structural versatility of these compounds is defined by the variable substituents on the aryl rings and the acyl group, allowing for extensive structure-activity relationship (SAR) studies. The general formula encompasses a wide range of electronic and steric variations, including halogens, alkyl groups, and electron-withdrawing nitro or trifluoromethyl moieties. This flexibility enables researchers to fine-tune the physicochemical properties of the final drug candidate, such as solubility, lipophilicity, and receptor binding affinity. Furthermore, the ability to form stable salts, particularly hydrobromides, addresses common formulation challenges associated with free base forms. By leveraging this patented technology, pharmaceutical companies can accelerate their pipeline development with high-quality, well-characterized building blocks that have demonstrated promising in vitro cytotoxicity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to functionalized thiazoles often suffer from harsh reaction conditions that can compromise sensitive functional groups, particularly the nitroethyl moiety which is prone to decomposition under strong acidic or basic environments. Conventional methods frequently rely on non-selective cyclization strategies that result in complex mixtures of regioisomers, necessitating difficult and costly purification steps that reduce overall yield. Additionally, many existing protocols utilize expensive transition metal catalysts or hazardous reagents that pose significant environmental and safety risks during commercial scale-up. The lack of modularity in older synthesis pathways limits the ability to rapidly generate diverse libraries of analogs, slowing down the lead optimization process for potential anticancer drugs. These inefficiencies translate directly into higher production costs and longer lead times for critical pharmaceutical intermediates.
The Novel Approach
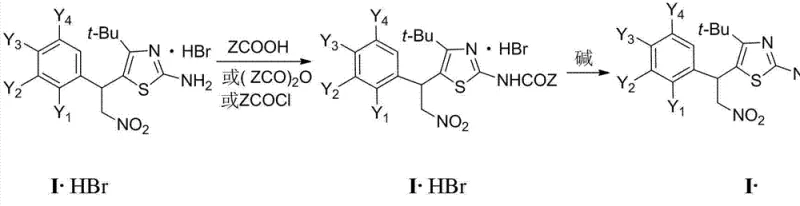
The methodology described in the patent offers a robust and modular alternative that overcomes these historical bottlenecks through a carefully sequenced multi-step synthesis. The process begins with a mild Michael addition to install the nitroethyl chain, followed by a controlled halogenation that activates the ketone for efficient ring closure with thiourea. This stepwise approach ensures high regioselectivity and preserves the integrity of the nitro group throughout the synthesis. The final acylation step is particularly advantageous as it can be performed under mild conditions using a variety of acylating agents, from simple acid anhydrides to complex aromatic acid chlorides. This flexibility allows for the rapid generation of diverse derivatives without the need for protecting group strategies that add unnecessary complexity. The result is a streamlined process that delivers high-purity intermediates suitable for direct use in downstream drug manufacturing.
Mechanistic Insights into Acylation and Cyclization Chemistry
The core of this synthetic strategy relies on the precise orchestration of nucleophilic substitution and condensation reactions to construct the thiazole nucleus. The cyclization step involves the nucleophilic attack of the sulfur atom in thiourea on the alpha-halo ketone intermediate, followed by intramolecular condensation to close the ring. This mechanism is highly favorable due to the electrophilic nature of the alpha-carbon activated by the adjacent carbonyl and halogen groups. The presence of the tert-butyl group plays a critical role in stabilizing the transition state and preventing unwanted side reactions at the 4-position of the ring. Following cyclization, the resulting 2-aminothiazole serves as a potent nucleophile for the subsequent acylation reaction. The nitrogen lone pair attacks the carbonyl carbon of the acylating agent, displacing the leaving group to form the stable amide bond. This transformation is essential for modulating the electronic properties of the thiazole ring and enhancing the biological activity of the final compound.
Understanding the impurity profile is vital for maintaining high quality standards in pharmaceutical manufacturing. The patent highlights the importance of controlling reaction parameters such as temperature and stoichiometry to minimize the formation of by-products like di-acylated species or hydrolysis products. The use of specific solvents and bases, such as pyridine or triethylamine, helps to scavenge acidic by-products and drive the equilibrium towards the desired product. Furthermore, the formation of salt forms, particularly the hydrobromide salt, serves as an effective purification strategy, as the salt often precipitates out of the reaction mixture with high purity. This crystallization-induced purification reduces the reliance on chromatographic separation, making the process more economically viable for large-scale production. The mechanistic clarity provided by this patent ensures that manufacturers can reproduce the results with high consistency and reliability.
How to Synthesize 4-Tert-Butyl-2-Acylaminothiazole Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized reaction conditions outlined in the patent to ensure maximum yield and purity. The process typically involves dissolving the amino-thiazole precursor in a suitable organic solvent such as dichloromethane or tetrahydrofuran, followed by the addition of a base to neutralize the generated acid. The acylating agent is then introduced slowly to control the exotherm and prevent local overheating which could degrade the product. Monitoring the reaction progress via TLC or HPLC is essential to determine the optimal endpoint and avoid over-reaction. Once the reaction is complete, standard workup procedures involving aqueous extraction and drying are employed to isolate the crude product, which can then be purified by recrystallization or column chromatography depending on the scale. For detailed operational parameters and specific molar ratios, please refer to the standardized synthesis guide below.
- Perform Michael addition of 4,4-dimethyl-1-aryl-1-enyl-3-pentanone with nitromethane to form the nitro-ketone precursor.
- Execute alpha-halogenation using bromine to activate the ketone for subsequent heterocyclic ring closure.
- Conduct cyclization with thiourea followed by acylation with acid chlorides or anhydrides to yield the final acylaminothiazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial opportunities for cost reduction in pharmaceutical intermediate manufacturing. The starting materials, such as 4,4-dimethyl-1-aryl-1-enyl-3-pentanone and nitromethane, are commodity chemicals that are readily available from multiple global suppliers, reducing supply chain risk and price volatility. The elimination of expensive transition metal catalysts and complex protecting group manipulations significantly lowers the raw material cost per kilogram of the final product. Furthermore, the high selectivity of the reaction minimizes waste generation and simplifies the purification process, leading to reduced solvent consumption and lower disposal costs. These efficiency gains translate directly into a more competitive pricing structure for buyers seeking high-quality oncology intermediates without compromising on purity specifications.
Enhanced supply chain reliability is another critical benefit derived from this robust synthetic methodology. The process operates under relatively mild conditions and utilizes standard reactor equipment, making it easily transferable between different manufacturing sites without the need for specialized infrastructure. The scalability of the reaction has been demonstrated through successful gram-to-kilogram scale-ups, ensuring that supply can be ramped up quickly to meet clinical trial demands or commercial launch requirements. The ability to produce stable salt forms also improves the shelf-life and handling characteristics of the intermediate, reducing losses during storage and transportation. For supply chain heads, this means a more predictable and resilient sourcing strategy that can withstand market fluctuations and logistical challenges.
Scalability and environmental compliance are increasingly important factors in vendor selection, and this process excels in both areas. The use of common organic solvents and the absence of heavy metals simplify the waste treatment process, aligning with modern green chemistry principles and regulatory requirements. The high atom economy of the cyclization and acylation steps ensures that a large proportion of the input materials end up in the final product, minimizing the environmental footprint of the manufacturing operation. This commitment to sustainable practices not only reduces regulatory burden but also enhances the corporate social responsibility profile of the supply chain. Partners who prioritize environmental stewardship will find this technology to be a strategic fit for their long-term sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these thiazole derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating these intermediates into their drug discovery programs. The information covers aspects ranging from reaction mechanisms to biological activity profiles, providing a comprehensive overview of the technology's capabilities.
Q: What is the primary therapeutic application of these thiazole derivatives?
A: These compounds exhibit significant antitumor activity against cervical cancer (Hela), lung adenocarcinoma (A549), and liver cancer (Bel7402) cell lines, serving as critical intermediates for COX-2 inhibitor development.
Q: How does the acylation step impact the stability of the nitroethyl group?
A: The acylation protects the amino group on the thiazole ring without disturbing the sensitive nitroethyl side chain, ensuring high purity and structural integrity during scale-up.
Q: Are salt forms available for improved solubility?
A: Yes, the patent describes various pharmaceutically acceptable salts including hydrobromide, hydrochloride, and methanesulfonate forms to enhance bioavailability and processing characteristics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Tert-Butyl-2-Acylaminothiazole Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex patent technologies into commercial reality. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the lab bench to the manufacturing plant. We understand the critical importance of stringent purity specifications in oncology drug development and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee batch-to-batch consistency. Our commitment to quality extends beyond mere compliance; we proactively identify and mitigate potential process risks to deliver intermediates that exceed industry standards. Partnering with us means gaining access to a wealth of technical expertise and a supply chain built on reliability and trust.
We invite you to explore the potential of these advanced thiazole intermediates for your next breakthrough therapy. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Contact us today to request specific COA data and route feasibility assessments that will help you make informed decisions about your supply strategy. Whether you are in the early stages of lead optimization or preparing for commercial launch, NINGBO INNO PHARMCHEM is your dedicated partner in bringing life-saving medicines to patients faster and more efficiently.
