Advanced Synthetic Strategy for Gamma-Lycorane: Enhancing Purity and Scalability for Pharmaceutical Applications
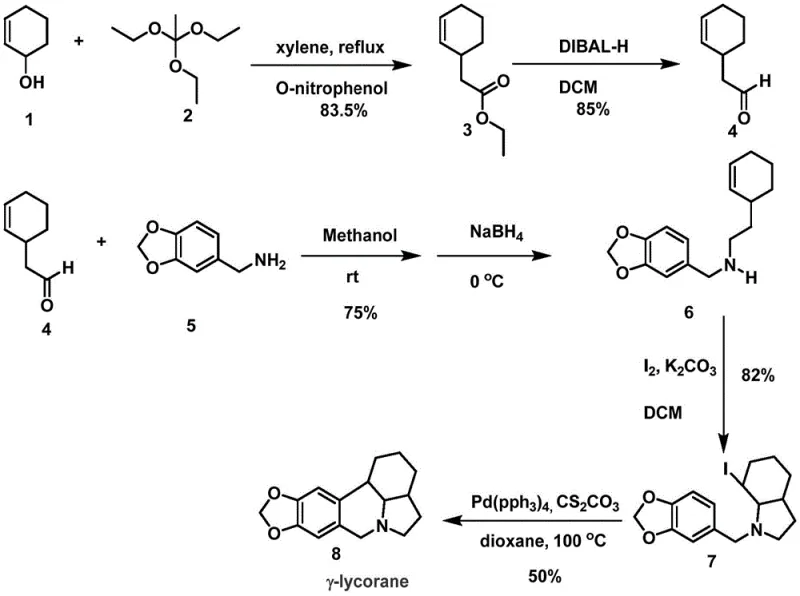
The pharmaceutical industry continuously seeks robust and scalable pathways for complex alkaloid scaffolds, particularly those exhibiting potential anticancer and antiviral properties. Patent CN109336899B, published in September 2022, introduces a groundbreaking methodology for the formal synthesis of gamma-lycorane, a core structure within the lycorine alkaloid family. This intellectual property outlines a strategic departure from traditional, labor-intensive syntheses by leveraging a concise five-step sequence that prioritizes operational simplicity and reagent availability. For R&D directors and process chemists, this patent represents a significant optimization in the construction of the octahydroindole core, utilizing a Johnson-Claisen rearrangement as the foundational step to establish critical carbon-carbon bonds early in the sequence. The technical disclosure emphasizes mild reaction conditions and high selectivity, addressing the perennial challenges of impurity management and yield erosion that plague multi-step alkaloid synthesis.
From a commercial perspective, the implications of this technology extend far beyond the laboratory bench. By replacing hazardous or exotic reagents with commodity chemicals, the described route offers a compelling value proposition for procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing. The reliance on standard solvents like xylene and dichloromethane, coupled with widely accessible catalysts, mitigates supply chain risks associated with specialized reagents. Furthermore, the streamlined nature of the synthesis suggests a reduced environmental footprint and lower waste disposal costs, aligning with modern green chemistry initiatives. As a leading reliable pharmaceutical intermediates supplier, understanding these nuanced process improvements is essential for evaluating the long-term viability and economic feasibility of integrating gamma-lycorane derivatives into broader drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of lycorane alkaloids has been fraught with significant technical hurdles that hinder commercial scalability. Prior art, including methodologies reported by Hideo Iido and Hiroki Yoshizaki, often relies on the generation of highly reactive benzyne intermediates or complex photochemical transformations. These traditional approaches frequently necessitate harsh reaction conditions, such as extreme temperatures or high-energy irradiation, which can lead to unpredictable decomposition pathways and difficult-to-remove byproducts. Moreover, many existing routes suffer from excessive linearity, requiring upwards of ten to fifteen steps to reach the target molecule, which inevitably compounds yield losses at each stage. The use of expensive transition metal catalysts in non-optimized cycles further exacerbates production costs, making these methods economically unviable for large-scale commercial scale-up of complex pharmaceutical intermediates. Additionally, the handling of unstable intermediates poses safety risks in an industrial setting, creating bottlenecks that delay project timelines and increase operational overhead.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN109336899B presents a refined and efficient strategy that circumvents these historical limitations through intelligent retrosynthetic design. The novel approach anchors the synthesis on a highly efficient Johnson-Claisen rearrangement, transforming simple 2-cyclohexen-1-ol into a functionalized ester with exceptional stereocontrol and an impressive yield of 83.5%. This early establishment of the carbon skeleton reduces the need for late-stage functionalization, thereby minimizing the risk of degrading sensitive motifs later in the sequence. The subsequent steps utilize robust and predictable chemistry, such as DIBAL-H reduction and sodium borohydride-mediated reductive amination, which are well-understood processes in process chemistry. By focusing on convergent bond formations and avoiding high-energy intermediates, this route ensures a smoother progression from raw materials to the final bicyclic core. The integration of an iodine-catalyzed cyclization followed by a palladium-mediated ring closure demonstrates a sophisticated yet practical application of modern catalysis to solve structural complexity without compromising safety or cost.
Mechanistic Insights into Johnson-Claisen Rearrangement and Pd-Catalyzed Cyclization
The cornerstone of this synthetic achievement lies in the meticulous execution of the Johnson-Claisen rearrangement, a pericyclic reaction that serves as the primary engine for molecular complexity in this route. In this specific transformation, 2-cyclohexen-1-ol reacts with triethyl orthoacetate in the presence of a catalytic amount of o-nitrophenol within a xylene solvent system. The mechanism proceeds through a [3,3]-sigmatropic shift, effectively migrating the allylic alcohol functionality to form a gamma,delta-unsaturated ester with high regioselectivity. The choice of xylene as a high-boiling solvent is critical, as it allows the reaction to proceed at reflux temperatures around 150°C, providing the necessary thermal energy to overcome the activation barrier of the rearrangement without requiring pressurized equipment. This step is particularly advantageous for R&D teams because it establishes the quaternary center and the side chain required for subsequent cyclization in a single operation, drastically reducing the step count compared to stepwise alkylation strategies. The high yield of 83.5% reported in the patent underscores the efficiency of this transformation, indicating minimal formation of constitutional isomers or polymerization byproducts.
Following the construction of the acyclic precursor, the synthesis employs a dual-catalytic strategy to forge the nitrogen-containing heterocyclic rings, which are defining features of the lycorane scaffold. The fourth step involves an iodine-catalyzed oxidative cyclization, where molecular iodine acts as a mild electrophile to activate the aromatic ring towards nucleophilic attack by the pendant amine. This electrophilic aromatic substitution is conducted under basic conditions using potassium carbonate, facilitating the formation of the tetrahydroisoquinoline core found in compound 7. The final closure of the pentacyclic system is achieved through an intramolecular palladium-catalyzed coupling reaction. Utilizing tetrakis(triphenylphosphine)palladium(0) and cesium carbonate in dioxane at 100°C, this step effects a C-N or C-C bond formation (depending on the specific substrate activation) to lock the conformation into the rigid gamma-lycorane structure. The use of cesium carbonate as a base is notable for its ability to solubilize organic substrates while maintaining mild basicity, preventing the epimerization of sensitive chiral centers. This mechanistic elegance ensures that the final product, gamma-lycorane, is obtained with high structural fidelity, meeting the stringent purity requirements demanded by medicinal chemistry applications.
How to Synthesize Gamma-Lycorane Efficiently
The synthesis of gamma-lycorane described in this patent offers a clear roadmap for process chemists aiming to replicate these results or adapt them for larger scales. The protocol is designed to be modular, allowing for the isolation and characterization of key intermediates such as the aldehyde (Compound 4) and the iodinated precursor (Compound 7), which serves as a crucial quality control checkpoint before the final ring closure. The detailed experimental procedures provided in the patent specify precise stoichiometric ratios, such as the use of 1.5 equivalents of DIBAL-H for the reduction step and 5 equivalents of potassium carbonate for the iodine cyclization, ensuring reproducibility. Operators are advised to maintain strict temperature controls, particularly during the cryogenic reduction at -78°C and the high-temperature reflux at 150°C, to maximize yield and minimize side reactions. For a comprehensive guide on executing these transformations with optimal safety and efficiency, please refer to the standardized operating procedures outlined below.
- Perform Johnson-Claisen rearrangement of 2-cyclohexen-1-ol with triethyl orthoacetate in xylene at 150°C to yield the ester intermediate.
- Reduce the ester to an aldehyde using DIBAL-H in dichloromethane at -78°C, followed by reductive amination with a benzyl amine derivative.
- Execute iodine-catalyzed N-heterocycle construction followed by palladium-catalyzed intramolecular coupling to close the final ring system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits that extend well beyond simple chemical yield. The primary advantage lies in the substantial simplification of the raw material portfolio; by utilizing 2-cyclohexen-1-ol and triethyl orthoacetate as starting points, the process leverages commodities that are produced in massive volumes globally, ensuring price stability and consistent availability. This stands in sharp contrast to legacy routes that might require custom-synthesized building blocks with long lead times and volatile pricing structures. Furthermore, the elimination of hazardous reagents like benzyne precursors reduces the regulatory burden and insurance costs associated with handling high-risk chemicals, directly contributing to cost reduction in pharmaceutical intermediates manufacturing. The operational simplicity of the workflow, characterized by standard workup procedures like aqueous quenching and column chromatography, means that existing manufacturing infrastructure can be utilized without the need for costly retrofits or specialized equipment investments.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the key rearrangement step and the use of inexpensive, recoverable solvents. Unlike photochemical methods that require specialized reactor hardware and high energy consumption, this thermal rearrangement and subsequent solution-phase chemistry can be performed in standard glass-lined or stainless steel reactors. The avoidance of precious metal catalysts in the early stages, reserving palladium only for the final step, significantly lowers the direct material cost per kilogram of product. Additionally, the high yields observed in the initial steps (over 80%) mean that less raw material is wasted, reducing the overall cost of goods sold (COGS) and improving the margin profile for commercial production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the short linear sequence of the synthesis, which reduces the number of potential failure points and inventory holding times. With only five distinct chemical transformations required to reach the target, the risk of batch failure due to cumulative errors is minimized compared to longer, more convoluted routes. The reliance on off-the-shelf reagents such as sodium borohydride, iodine, and cesium carbonate ensures that production schedules are not held hostage by the delivery of niche chemicals. This reliability is critical for maintaining continuous supply to downstream clients who depend on timely delivery of high-purity pharmaceutical intermediates for their own drug development programs, thereby strengthening long-term partnerships and contractual obligations.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the process is designed to be inherently safer and cleaner. The solvents used, primarily xylene, dichloromethane, and dioxane, are well-understood in terms of waste treatment and recycling protocols, facilitating compliance with increasingly strict environmental regulations. The absence of explosive intermediates or highly toxic gases simplifies the safety assessment for plant scale-up, allowing for faster technology transfer from the lab to the pilot plant. The robustness of the reaction conditions implies that the process can be scaled from 100 kgs to multi-ton quantities with predictable outcomes, supporting the growing demand for complex alkaloid scaffolds in the oncology and antiviral sectors without compromising on safety or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of gamma-lycorane, derived directly from the specific embodiments and claims of the referenced patent. These insights are intended to clarify the operational parameters and strategic benefits of this novel methodology for stakeholders evaluating its potential integration into their supply chains. Understanding these details is crucial for making informed decisions about process adoption and resource allocation.
Q: What are the key advantages of this gamma-lycorane synthesis route compared to traditional methods?
A: This route utilizes conventional chemical reagents and avoids hazardous benzyne intermediates or complex photochemical setups. It features a shorter linear sequence with high-yielding steps like the Johnson-Claisen rearrangement (83.5% yield), significantly simplifying the operational complexity for industrial scale-up.
Q: Is the starting material for this synthesis readily available for large-scale production?
A: Yes, the synthesis begins with 2-cyclohexen-1-ol, which is a commodity chemical widely available in the global market. This ensures a stable supply chain and prevents bottlenecks often associated with exotic or custom-synthesized starting materials in alkaloid production.
Q: How does the patent address impurity control during the cyclization steps?
A: The process employs specific catalytic systems, such as iodine for N-heterocycle construction and palladium for the final ring closure, under controlled conditions. These methods are designed to minimize side reactions, resulting in a cleaner crude product profile that facilitates easier purification and higher final purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Lycorane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex academic patents into reliable commercial reality. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of the Johnson-Claisen route are fully realized in a GMP-compliant environment. We understand that the successful production of complex alkaloids like gamma-lycorane requires not just chemical expertise, but also rigorous adherence to stringent purity specifications and robust quality control protocols. Our state-of-the-art facilities are equipped with advanced analytical instrumentation and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets the exacting standards required for preclinical and clinical research applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this innovative synthetic route for their specific needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized pathway can reduce your overall project costs. We encourage you to contact us today to request specific COA data for our reference standards and to discuss detailed route feasibility assessments for your upcoming projects. Let us be your trusted partner in navigating the complexities of alkaloid synthesis, delivering high-quality intermediates that accelerate your path to market.
