Revolutionizing m-Terphenyl Production: A Metal-Free Route for Industrial Scale-Up
Revolutionizing m-Terphenyl Production: A Metal-Free Route for Industrial Scale-Up
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable manufacturing processes. A significant breakthrough in this domain is documented in Chinese Patent CN111205152B, which details a novel preparation method for m-terphenyl compounds. This technology represents a paradigm shift from traditional metal-catalyzed cross-coupling reactions to a streamlined, acid-catalyzed condensation strategy. For R&D directors and procurement managers in the pharmaceutical and electronic materials sectors, this innovation offers a compelling value proposition: the ability to produce high-purity m-terphenyl intermediates without the burden of heavy metal contamination or the high costs associated with precious metal catalysts. The versatility of m-terphenyl structures, ranging from high-energy materials like nonanitroterphenyl (NONA) to bioactive natural products, underscores the strategic importance of mastering their synthesis.
Furthermore, the structural diversity achievable through this method allows for the efficient generation of complex scaffolds required in advanced material science. As depicted in the patent literature, m-terphenyl derivatives serve as critical building blocks for conductive polymers, liquid crystal materials, and heat transfer fluids. The ability to access these motifs through a robust, one-pot synthetic route significantly de-risks the supply chain for downstream manufacturers. By leveraging aryl methyl ketones and triethyl orthoformate as primary feedstocks, the process bypasses the need for pre-functionalized halides or organometallic reagents, thereby simplifying the raw material sourcing strategy. This technological advancement positions suppliers who adopt this methodology as reliable partners capable of delivering consistent quality while adhering to increasingly stringent environmental regulations regarding heavy metal discharge.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the m-terphenyl skeleton has relied heavily on transition metal-catalyzed cross-coupling reactions, such as the Suzuki-Miyaura or Ullmann couplings. While these methods are chemically robust, they present significant logistical and economic challenges for large-scale industrial applications. The reliance on palladium, copper, or nickel catalysts introduces substantial raw material costs, which are subject to volatile global market fluctuations. Moreover, the presence of residual metals in the final product is a critical quality attribute, especially for pharmaceutical intermediates and electronic chemicals, necessitating expensive and time-consuming purification steps to meet strict ppm-level specifications. Additionally, alternative routes involving Grignard reagents, as shown in earlier literature, require stringent anhydrous conditions and cryogenic temperatures, posing safety risks and increasing energy consumption.
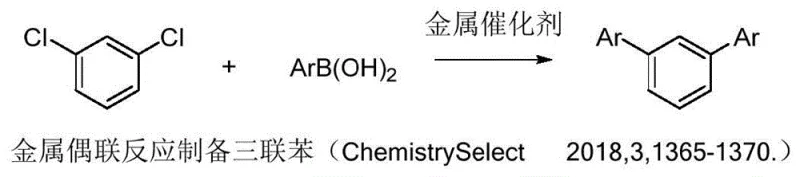
Beyond the economic factors, the environmental footprint of conventional metal-coupling methodologies is considerable. The generation of stoichiometric amounts of metal salts as waste byproducts complicates waste management and increases the overall E-factor of the process. Furthermore, the substrate scope in many metal-catalyzed reactions can be limited by functional group tolerance, often requiring protective group strategies that add extra synthetic steps and reduce overall yield. The need for specialized ligands to stabilize the metal catalysts further adds to the complexity and cost of the supply chain. For procurement managers, these factors translate into higher unit costs and longer lead times, as the availability of high-grade catalysts and ligands can sometimes be a bottleneck. Consequently, there is a pressing industry demand for alternative synthetic strategies that can circumvent these inherent limitations while maintaining high efficiency and selectivity.
The Novel Approach
The methodology disclosed in patent CN111205152B offers a transformative solution by utilizing a Brønsted acid-catalyzed condensation between aryl methyl ketones and triethyl orthoformate. This approach fundamentally alters the economic and operational dynamics of m-terphenyl production. By replacing expensive transition metals with catalytic amounts of organic acids like trifluoromethanesulfonic acid (TfOH), the process drastically reduces the cost of goods sold (COGS). The reaction proceeds under remarkably mild conditions, typically at 50°C, which minimizes energy consumption compared to the high temperatures or cryogenic conditions required by older methods. Perhaps most significantly, the reaction can be performed under solvent-free conditions or in common solvents like ethanol, offering flexibility in process design and reducing the volume of organic waste generated. This green chemistry aspect is a major advantage for facilities aiming to reduce their carbon footprint and comply with modern environmental standards.
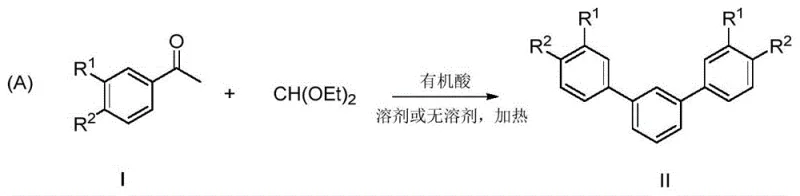
From an operational perspective, the simplicity of this one-pot protocol cannot be overstated. The elimination of multiple synthetic steps, such as the preparation of organometallic reagents or the installation of leaving groups, streamlines the manufacturing workflow. The broad substrate scope demonstrated in the patent, accommodating various alkyl and halogen substituents on the aromatic ring, ensures that this method is applicable to a wide range of m-terphenyl derivatives needed for different applications. For supply chain heads, this translates to enhanced reliability and agility; the raw materials are commodity chemicals that are readily available in bulk quantities, reducing the risk of supply disruptions. The combination of low operating temperatures, short reaction times (approximately 2 hours), and high yields makes this novel approach highly attractive for commercial scale-up, offering a clear pathway to cost reduction in fine chemical manufacturing without compromising on product purity or quality.
Mechanistic Insights into Acid-Catalyzed Condensation
The core of this innovative synthesis lies in the activation of the carbonyl group of the aryl methyl ketone by the strong Brønsted acid catalyst. In the presence of trifluoromethanesulfonic acid, the ketone undergoes protonation, increasing its electrophilicity and facilitating nucleophilic attack by the orthoformate species. This initial condensation step generates a reactive intermediate that subsequently participates in a series of electrophilic aromatic substitution events. The mechanism likely involves the formation of a stabilized carbocation or enol ether intermediate, which then attacks the electron-rich aromatic rings of additional ketone molecules. This cascade reaction effectively stitches three aromatic units together in a single operational sequence, constructing the central benzene ring of the m-terphenyl system while simultaneously eliminating small molecule byproducts like ethanol. The regioselectivity observed, favoring the meta-substitution pattern, is driven by the electronic properties of the intermediates and the steric constraints imposed during the cyclization process.
Understanding the impurity profile is crucial for R&D teams aiming to implement this process. The primary byproducts typically stem from incomplete condensation or over-alkylation, but these are generally easier to separate than the metal complexes found in coupling reactions. The use of a strong acid catalyst ensures rapid conversion, minimizing the residence time of reactive intermediates that could lead to polymerization or tar formation. Furthermore, the quenching step with saturated sodium carbonate solution effectively neutralizes the acid, allowing for straightforward phase separation. The absence of metal residues means that the final purification, often achieved via silica gel chromatography or recrystallization, yields products of exceptional purity suitable for sensitive applications like OLED materials or API synthesis. This mechanistic clarity provides a solid foundation for process optimization, allowing engineers to fine-tune parameters such as acid loading and temperature to maximize yield and minimize waste.
How to Synthesize m-Terphenyl Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to ensure reproducibility and safety. The general protocol involves mixing the aryl methyl ketone and triethyl orthoformate under an inert atmosphere to prevent moisture interference, although the reaction shows some tolerance. Heating the mixture to 50°C prior to catalyst addition ensures a homogeneous solution, which is critical for consistent reaction kinetics. The addition of the acid catalyst should be controlled to manage the exotherm, although the reaction is generally mild. Following the reaction period, standard aqueous workup procedures are sufficient to isolate the crude product. For detailed operational specifics, including exact molar ratios and purification techniques for specific derivatives, please refer to the standardized synthesis guide below.
- Mix aryl methyl ketone and triethyl orthoformate in a reactor under inert gas protection, optionally with a solvent like ethanol or THF.
- Heat the mixture to 50°C and stir for 0.5 hours to ensure homogeneity before catalyst addition.
- Add trifluoromethanesulfonic acid (TfOH) as the catalyst, maintain reaction at 50°C for approximately 2 hours, then quench and purify.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route offers tangible strategic benefits that extend beyond simple unit price reductions. The elimination of precious metal catalysts removes a significant variable cost component that is often subject to geopolitical instability and market speculation. This stability allows for more accurate long-term budgeting and pricing contracts with customers. Additionally, the simplified downstream processing—specifically the lack of need for specialized metal scavenging resins or complex filtration systems—reduces capital expenditure on equipment and lowers operational overhead. The ability to run the reaction under solvent-free conditions further amplifies these savings by reducing the volume of solvents that need to be purchased, stored, recovered, or disposed of, leading to substantial cost savings in waste management and regulatory compliance.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the substitution of expensive palladium or copper catalysts with catalytic amounts of organic acids. This change not only lowers the direct material cost but also eliminates the associated costs of catalyst recovery and metal testing. The high atom economy of the condensation reaction means that a larger proportion of the raw material mass ends up in the final product, reducing the effective cost per kilogram of output. Furthermore, the short reaction time of approximately 2 hours increases the throughput of existing reactor assets, allowing for higher production volumes without the need for new infrastructure investment. These factors combine to create a highly competitive cost structure for m-terphenyl manufacturing.
- Enhanced Supply Chain Reliability: Sourcing aryl methyl ketones and triethyl orthoformate is significantly less risky than sourcing specialized organoboron reagents or halogenated aromatics required for cross-coupling. These starting materials are commodity chemicals produced by multiple vendors globally, ensuring a robust and resilient supply chain. The reduced dependency on single-source catalyst suppliers mitigates the risk of production stoppages due to raw material shortages. Moreover, the mild reaction conditions reduce the wear and tear on reactor vessels and ancillary equipment, leading to higher asset availability and reduced maintenance downtime. This reliability is crucial for meeting tight delivery schedules and maintaining trust with downstream pharmaceutical and electronic material clients.
- Scalability and Environmental Compliance: The green chemistry credentials of this process facilitate easier regulatory approval and permit acquisition for new production lines. The absence of heavy metals simplifies the environmental impact assessment and reduces the burden of hazardous waste disposal. Scaling up a one-pot condensation reaction is inherently safer and more straightforward than scaling multi-step sequences involving sensitive organometallics. The potential for solvent-free operation aligns perfectly with corporate sustainability goals, reducing the facility's volatile organic compound (VOC) emissions. This environmental advantage can be leveraged in marketing to eco-conscious customers and helps in complying with increasingly strict international environmental regulations regarding chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this m-terphenyl synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. Whether the concern is about catalyst handling, substrate compatibility, or purification efficiency, the following insights clarify the operational realities of this novel method.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method utilizes organic acids like trifluoromethanesulfonic acid (TfOH) as catalysts, completely eliminating the need for costly palladium, copper, or iron metal catalysts typically found in Suzuki or Ullmann couplings.
Q: What are the typical reaction conditions for this m-terphenyl synthesis?
A: The reaction operates under mild conditions, specifically at a temperature of 50°C for about 2 hours. It can be performed either in solvents like ethanol or THF, or even under solvent-free conditions, enhancing its environmental profile.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up due to its simple one-pot operation, readily available raw materials (aryl methyl ketones), and the absence of complex metal removal steps, which significantly simplifies downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Terphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the global supply of m-terphenyl compounds. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to rapidly translate this patented laboratory method into a robust commercial manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless and efficient. We are committed to delivering high-purity m-terphenyl intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify the absence of metal contaminants and ensure batch-to-batch consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of acid-catalyzed reactions, including corrosion-resistant reactors and advanced solvent recovery systems.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can optimize your supply chain and reduce your overall manufacturing costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our m-terphenyl products against your internal standards. Let us help you secure a sustainable and cost-effective supply of these critical chemical building blocks for your next-generation pharmaceutical or electronic materials.
