Advanced Synthesis of Bora Z-type Acene Derivatives for High-Performance OLED Commercialization
Advanced Synthesis of Bora Z-type Acene Derivatives for High-Performance OLED Commercialization
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit superior luminescent efficiency but also possess robust thermal stability and processability for large-scale manufacturing. Patent CN114249759A introduces a groundbreaking class of Bora Z-type acene derivatives that address the critical bottlenecks of stability and synthesis difficulty inherent in traditional organoboron compounds. By leveraging a uniqueSilicon-to-Boron exchange strategy, this technology enables the precise construction of electron-deficient pi-conjugated systems that serve as exceptional emitters or dopants in electroluminescent devices. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this patent represents a pivotal shift towards modular, high-yield synthetic pathways that can be scaled for commercial production without compromising purity or performance metrics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of high-performance organic boron luminescent materials has been hindered by the intrinsic instability of boron-carbon bonds and the complexity of introducing boron atoms into rigid conjugated skeletons. Conventional methods often struggle with low yields, harsh reaction conditions that degrade sensitive functional groups, and a lack of structural diversity, limiting the tunability of emission wavelengths from deep blue to near-infrared. Furthermore, direct boration strategies frequently result in materials with poor film-forming properties and low glass transition temperatures, leading to device degradation under operational heat. These technical barriers have restricted the variety of stable organic boron compounds available for practical application, forcing manufacturers to rely on expensive phosphorescent materials or less efficient fluorescent alternatives that cannot fully utilize triplet excitons for light emission.
The Novel Approach
The patented methodology overcomes these hurdles by employing a stable Sila Z-type acene derivative (precursor 1c) as a strategic template for boron incorporation. This innovative route utilizes the similar atomic radius and bonding characteristics of silicon and boron to first establish the rigid Z-type backbone, which is then converted into the target boron-doped structure via a controlled demethylation and substitution process using boron tribromide. This two-step strategy decouples the difficult backbone formation from the sensitive boron installation, allowing for high-purity synthesis of diverse derivatives (compounds 1-16) through simple variation of the final coupling partners. 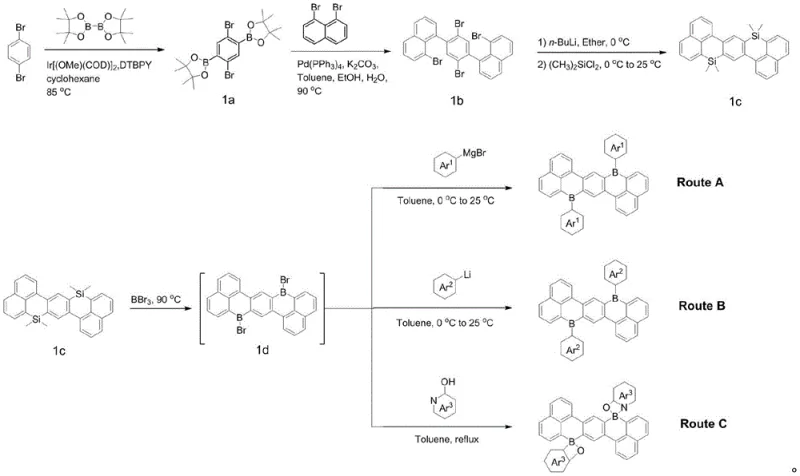
Mechanistic Insights into Boron Tribromide Mediated Si-B Exchange
The core chemical transformation relies on the potent Lewis acidity of boron tribromide (BBr3) to cleave the robust silicon-carbon bonds within the precursor 1c. Under inert atmosphere at elevated temperatures (90°C), BBr3 facilitates the replacement of dimethylsilyl groups with dibromo-boryl moieties, generating the highly reactive intermediate 1d. This intermediate serves as a versatile electrophilic hub that can be subsequently functionalized through three distinct pathways (Route A, B, and C) depending on the desired electronic properties. Route A employs Grignard reagents to install electron-donating aryl groups directly, while Route B utilizes lithiated heterocycles for more complex architectures, and Route C involves condensation with hydroxy-functionalized ligands to form rigid B-O or B-N chelated structures. This mechanistic flexibility ensures that the electron orbitals of the boron atoms are effectively conjugated with nitrogen or oxygen atoms in the final structure, realizing the thermal activation delayed fluorescence (TADF) characteristic essential for high internal quantum efficiency.
From an impurity control perspective, the use of the stable silicon precursor minimizes the formation of side products typically associated with direct boration, such as protodeboronation or oligomerization. The final purification steps, involving column chromatography or vacuum gradient sublimation, effectively remove residual boron species and unreacted precursors, ensuring the stringent purity specifications required for electronic grade materials. The resulting Bora Z-type acene derivatives exhibit a planar or slightly twisted conformation that prevents excessive pi-pi stacking, thereby reducing concentration quenching in solid-state films. 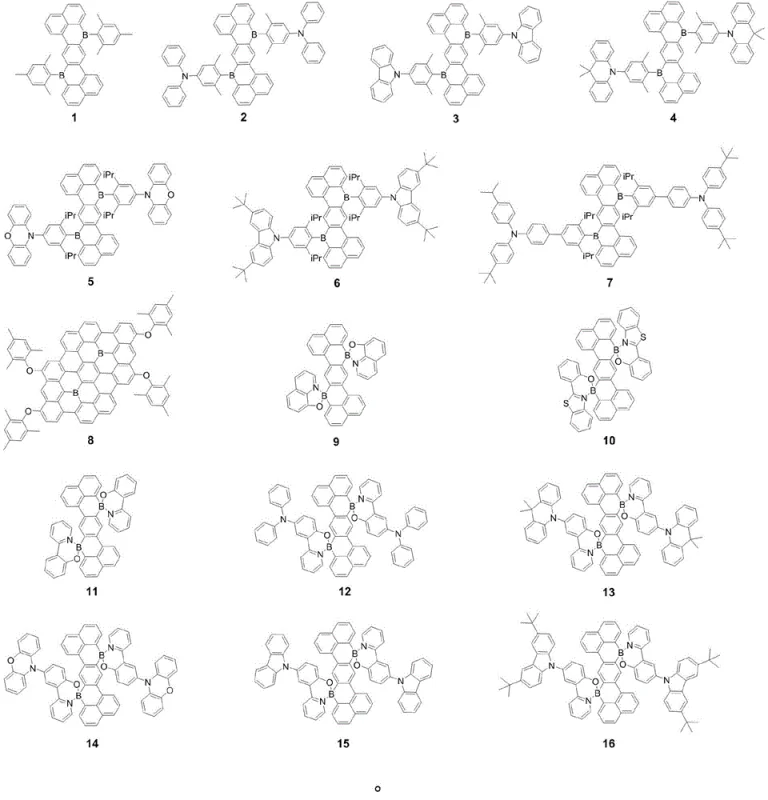
How to Synthesize Bora Z-type Acene Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced materials, starting from the commercially accessible p-dibromobenzene and progressing through the iridium-catalyzed borylation and palladium-coupling steps to form the silicon precursor. The detailed standardized synthesis steps below guide the operator through the critical conversion of precursor 1c to the final high-purity derivatives, emphasizing temperature control and inert atmosphere handling to maximize yield and safety.
- Preparation of Intermediate 1d: React precursor 1c with boron tribromide at 90°C for 10 hours under inert atmosphere to replace silicon atoms with boron.
- Route A (Grignard Addition): React intermediate 1d with specific Grignard reagents (Compound A) at 0°C to room temperature to yield derivatives 1-4.
- Route B & C (Lithiation/Condensation): Utilize n-butyllithium activated Compound B for derivatives 5-8, or direct condensation with Compound C at 90°C for derivatives 9-16.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the modular nature of this synthetic route offers profound logistical and economic benefits. By centralizing the production of the key intermediate 1c, manufacturers can maintain a lean inventory of a single stable precursor while rapidly responding to market demands for different emission colors or device architectures by simply varying the final coupling reagents. This strategy drastically simplifies the supply chain, reducing the need to stockpile numerous distinct finished goods and minimizing the risk of obsolescence. Furthermore, the elimination of complex multi-step boron installations in the early stages of synthesis reduces the consumption of expensive catalysts and reagents, leading to substantial cost savings in electronic chemical manufacturing.
- Cost Reduction in Manufacturing: The ability to synthesize a wide library of derivatives (1-16) from a common intermediate significantly lowers the cost of goods sold (COGS) by maximizing equipment utilization and minimizing batch changeover times. The avoidance of transition metal catalysts in the final functionalization steps (in certain routes) further reduces downstream purification costs associated with heavy metal removal, ensuring a more economical process flow that aligns with aggressive budget targets for mass production.
- Enhanced Supply Chain Reliability: The reliance on stable silicon-based precursors mitigates the supply risks often associated with sensitive organoboron intermediates, which can degrade during storage or transport. This stability ensures consistent quality and availability of raw materials, reducing lead time for high-purity OLED materials and enabling manufacturers to meet tight delivery schedules for consumer electronics production cycles without compromising on material integrity or performance consistency.
- Scalability and Environmental Compliance: The synthetic pathway utilizes standard organic solvents like toluene and ether, which are well-established in industrial settings, facilitating seamless scale-up from laboratory grams to metric ton quantities. The process avoids the generation of hazardous boron waste streams typical of direct boration methods, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations, thereby safeguarding the facility's operational license and reputation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and performance of these novel boron-doped materials, derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is critical for integrating these materials into next-generation display and lighting technologies.
Q: What is the primary advantage of using the SilaZ-type precursor 1c?
A: The SilaZ-type precursor 1c offers superior stability and precise structural control compared to direct boron doping methods. It allows for the modular synthesis of multiple derivatives (1-16) from a single intermediate, significantly streamlining the supply chain and reducing inventory complexity for OLED material manufacturers.
Q: How does the Bora Z-type structure improve device performance?
A: The unique Z-type acene skeleton combined with electron-deficient boron atoms enhances both electron and hole transport capabilities. This balanced charge transport, coupled with high thermal stability and a high glass transition temperature, results in organic electroluminescent devices with lower driving voltages, higher external quantum efficiency, and extended operational lifetimes.
Q: Can these materials be used in non-doped emissive layers?
A: Yes, the patent data indicates that these derivatives function effectively both as doping materials (typically at 8% concentration in hosts like mCP) and as non-doped emissive materials. However, doped configurations generally demonstrate superior external quantum efficiency and device longevity compared to non-doped films.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bora Z-type Acene Derivative Supplier
As the demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO is essential for translating patented laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Bora Z-type acene derivative meets the exacting standards required for high-performance optoelectronic applications.
We invite you to collaborate with our technical team to explore how this advanced synthetic technology can optimize your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your time-to-market for next-generation organic electroluminescent devices.
