Revolutionizing 4-Hydroxy-2-butynoic Acid Production with Safe Iron-Catalyzed Aerobic Oxidation Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and safe methodologies for synthesizing critical intermediates, particularly those serving as scaffolds for major therapeutic classes. A pivotal advancement in this domain is detailed in Chinese Patent CN113651684B, which discloses a novel preparation method for 4-hydroxy-2-butynoic acid. This compound serves as a fundamental building block for 1,4-dihydropyridine derivatives, the core structure of widely prescribed calcium channel blockers used in treating hypertension. The patent introduces a transformative catalytic system that replaces hazardous stoichiometric reagents with a synergistic combination of N-O free radical catalysts and iron salts, utilizing molecular oxygen or air as the terminal oxidant. This technological leap addresses long-standing safety and cost inefficiencies associated with legacy synthetic routes, offering a pathway that is not only chemically elegant but also commercially superior for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-hydroxy-2-butynoic acid has relied heavily on organometallic strategies that, while effective on a small laboratory scale, present formidable challenges for industrial implementation. The most prominent conventional route involves the use of propargyl alcohol or its protected derivatives treated with strong bases such as n-butyllithium to generate alkynyllithium species, which are subsequently carboxylated with carbon dioxide. As illustrated in the reaction scheme below, this approach demands rigorous exclusion of moisture and oxygen, requiring specialized equipment and inert atmosphere handling that significantly inflates operational expenditures.
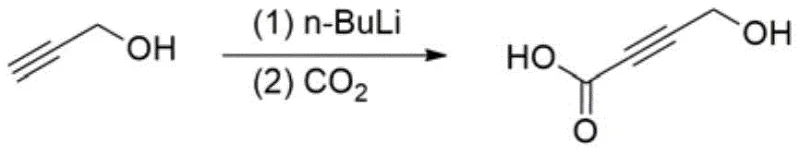
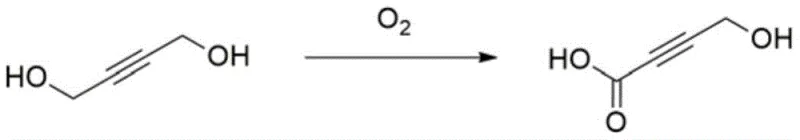
Furthermore, the inherent hazards of pyrophoric reagents like n-butyllithium cannot be overstated; these materials ignite spontaneously upon contact with air, creating severe safety liabilities in large-scale manufacturing environments. Alternative biological or electrochemical methods have been explored, such as the enzymatic oxidation or electrolytic oxidation of 1,4-butynediol shown below, but these suffer from critical scalability bottlenecks. These green alternatives often require extremely dilute reaction conditions to maintain enzyme activity or prevent electrode passivation, resulting in poor space-time yields and making them economically unviable for producing the tonnage quantities required by the global API market.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN113651684B utilizes a direct oxidative transformation of 1,4-butynediol. By employing a composite catalytic system comprising an N-O free radical mediator and an iron salt co-catalyst, the process enables the selective oxidation of one hydroxyl group to a carboxylic acid using inexpensive air or oxygen. The reaction equation depicted below highlights the simplicity and atom economy of this approach, where the only byproduct is water, eliminating the generation of stoichiometric metal waste streams typical of chromium or manganese-based oxidations.

This novel route operates effectively in common organic solvents such as dichloromethane, dichloroethane, or toluene, and tolerates ambient moisture levels that would quench organolithium reagents. The ability to run the reaction at high substrate concentrations dramatically improves reactor throughput, while the use of non-pyrophoric catalysts removes the need for cryogenic conditions or specialized fire-suppression infrastructure. This paradigm shift from stoichiometric, hazard-intensive chemistry to catalytic, aerobic oxidation represents a significant milestone in cost reduction in pharmaceutical intermediate manufacturing, aligning perfectly with modern principles of green chemistry and process safety.
Mechanistic Insights into FeCl3-Catalyzed TEMPO Oxidation
The efficacy of this transformation relies on the synergistic interplay between the nitroxyl radical catalyst and the transition metal co-catalyst. The N-O free radical, typically TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) or its 4-substituted derivatives as shown in the structural diagram below, acts as the primary hydrogen abstractor. It selectively oxidizes the primary alcohol moiety of 1,4-butynediol to the corresponding aldehyde and subsequently to the carboxylic acid. The iron salt, which can be a ferrous or ferric species like FeCl2 or Fe(NO3)3, plays a crucial role in the regeneration of the active oxoammonium species from the reduced hydroxylamine form of the catalyst, utilizing molecular oxygen as the terminal electron acceptor.
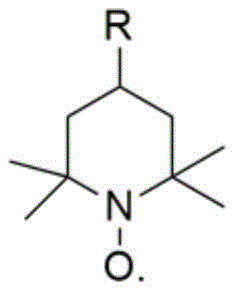
This catalytic cycle ensures that the expensive nitroxyl radical is used in minute catalytic quantities (as low as 0.2 mol%) rather than stoichiometric amounts. Crucially, the system exhibits remarkable chemoselectivity, preventing the over-oxidation of the alkyne triple bond or the formation of the dicarboxylic acid byproduct, 1,4-butynedioic acid. Experimental data from the patent indicates that under optimized conditions, the selectivity for the mono-acid product approaches 100%, with the diacid impurity remaining negligible. This high level of control simplifies downstream purification, as the product can often be isolated via simple filtration and recrystallization, avoiding complex chromatographic separations that would otherwise erode profit margins in commercial production.
How to Synthesize 4-Hydroxy-2-butynoic Acid Efficiently
The practical execution of this synthesis is straightforward and adaptable to standard chemical processing equipment. The general procedure involves charging a reactor with 1,4-butynediol, the chosen organic solvent, and the catalytic components, followed by the continuous sparging of air or oxygen. Reaction progress is conveniently monitored via HPLC, typically reaching completion within 12 to 24 hours depending on the specific catalyst loading and temperature profile. For detailed operational parameters, including specific solvent choices, catalyst loadings, and workup procedures that ensure maximum yield and purity, please refer to the standardized synthesis guide below.
- Charge a reactor with 1,4-butynediol, an organic solvent (such as DCM or DCE), and catalytic amounts of an N-O radical catalyst (e.g., TEMPO) and an iron salt.
- Introduce air or oxygen into the reaction mixture while maintaining the temperature between room temperature and reflux, monitoring progress via HPLC.
- Upon completion, filter the reaction mixture, recover the solvent via distillation, and purify the crude residue through acid washing and recrystallization to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed aerobic oxidation technology offers compelling strategic advantages that extend far beyond simple yield metrics. The transition away from hazardous organolithium reagents fundamentally alters the risk profile of the manufacturing process, reducing insurance premiums and safety compliance costs while enhancing the overall resilience of the supply chain against regulatory scrutiny. Furthermore, the reliance on commodity chemicals like iron salts and air, rather than specialized, imported reagents, insulates the production cost from volatile raw material markets, ensuring stable pricing for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of expensive, pyrophoric n-butyllithium and the associated cryogenic cooling requirements results in substantial cost savings. By replacing stoichiometric reagents with catalytic amounts of inexpensive iron salts and utilizing free air as the oxidant, the variable cost of goods sold is drastically reduced. Additionally, the high reaction concentration permitted by this method maximizes the utilization of reactor volume, lowering the fixed cost per kilogram of product produced.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against moisture and oxygen ingress simplifies logistics and storage requirements for raw materials. Unlike sensitive organometallics that require cold chain shipping and strict inventory rotation, the catalysts and substrates for this process are stable, shelf-stable commodities. This stability reduces the risk of production delays caused by degraded reagents and allows for more flexible just-in-time manufacturing strategies.
- Scalability and Environmental Compliance: The process generates minimal waste, primarily water, and avoids the heavy metal contamination issues associated with traditional stoichiometric oxidants like Jones reagent. This clean profile simplifies wastewater treatment and environmental permitting, facilitating faster scale-up from pilot plant to multi-ton commercial production. The use of standard gas-liquid reactors means that existing manufacturing infrastructure can often be utilized without significant capital expenditure on specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners evaluating this route for their supply chain.
Q: Why is the iron-catalyzed oxidation method superior to the traditional alkyllithium route?
A: The traditional alkyllithium method requires hazardous, pyrophoric reagents like n-butyllithium and strictly anhydrous conditions, posing significant safety risks and high operational costs. In contrast, the iron-catalyzed aerobic oxidation utilizes benign air or oxygen as the oxidant, operates under mild conditions without strict moisture exclusion, and achieves near-quantitative selectivity, drastically improving process safety and economic viability.
Q: What represents the primary impurity in this oxidation process and how is it controlled?
A: The primary potential over-oxidation impurity is 1,4-butynedioic acid (acetylenedicarboxylic acid derivative). The patented composite catalytic system, utilizing specific N-O radicals like TEMPO alongside iron salts, demonstrates exceptional chemoselectivity, keeping the content of this diacid impurity negligible and achieving selectivity close to 100% under optimized conditions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly amenable to scale-up. Unlike enzymatic or electrolytic methods which suffer from low substrate concentrations and electrode fouling, this chemical oxidation supports high reaction concentrations in standard organic solvents. It utilizes conventional gas-liquid reactors and avoids expensive, sensitive reagents, making it ideal for multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-2-butynoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a viable reaction scheme; it demands a partner with the technical acumen to navigate complex scale-up challenges. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity 4-hydroxy-2-butynoic acid that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this innovative iron-catalyzed technology for their projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to discuss route feasibility assessments and to obtain specific COA data that demonstrates our commitment to quality and reliability as your trusted partner in fine chemical synthesis.
