Advanced Asymmetric Synthesis of 3-(3,4-Dihydroxyphenyl)-2-Hydroxypropionate for Cardiovascular APIs
The pharmaceutical industry's relentless pursuit of effective treatments for cardiovascular and cerebrovascular diseases has placed significant emphasis on the quality and purity of key active ingredients. Patent CN102030648B introduces a groundbreaking asymmetric synthesis method for 3-(3,4-dihydroxyphenyl)-2-hydroxypropionate, a critical metabolite and intermediate associated with Salvia miltiorrhiza (Danshen) derivatives. This technology addresses the longstanding challenges in producing chiral drugs by shifting away from racemic synthesis towards highly stereoselective catalytic processes. For R&D directors and procurement specialists, this patent represents a pivotal shift in manufacturing strategy, offering a pathway to high-purity intermediates that are essential for next-generation cardiovascular therapeutics. The method leverages the robust chemistry of Sharpless asymmetric dihydroxylation, ensuring that the final product meets the stringent optical purity requirements demanded by modern regulatory bodies without the inefficiencies of traditional resolution techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-(3,4-dihydroxyphenyl)-alpha-hydroxypropionic acid esters relied on pathways that were inherently inefficient and chemically cumbersome. Traditional methods often involved the synthesis of azlactones from acetylglycine, followed by hydrolysis and decarboxylation steps that inevitably produced racemic mixtures. The fundamental flaw in these legacy approaches is the generation of a 50:50 mixture of enantiomers, necessitating a subsequent resolution step to isolate the biologically active isomer. This resolution process not only caps the theoretical maximum yield at 50% but also introduces additional unit operations, solvent consumption, and waste generation, thereby inflating the cost of goods sold (COGS). Furthermore, the use of acetyl protecting groups and the harsh conditions required for their removal often led to side reactions and impurity profiles that were difficult to control, posing significant risks for clinical translation and regulatory approval.
The Novel Approach
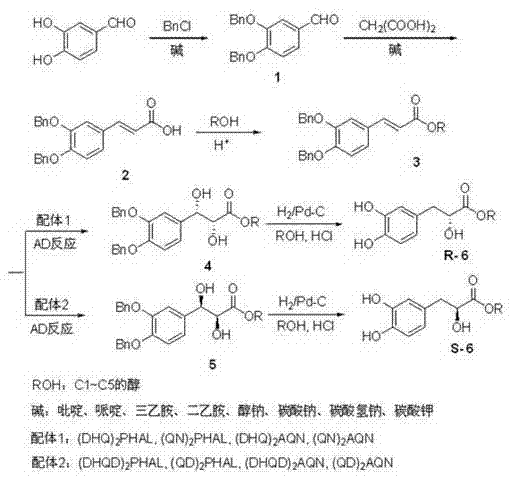
In stark contrast, the methodology disclosed in CN102030648B utilizes a convergent strategy centered on the asymmetric functionalization of an olefinic precursor. The process begins with the protection of 3,4-dihydroxybenzaldehyde, followed by a Knoevenagel condensation to establish the carbon backbone. The true innovation lies in the subsequent asymmetric dihydroxylation step, which installs two hydroxyl groups with precise stereochemical control in a single operation. By employing chiral cinchona alkaloid ligands in conjunction with osmium catalysis, the synthesis bypasses the formation of racemates entirely, delivering the desired enantiomer directly. This approach not only simplifies the synthetic sequence by eliminating the need for resolution but also enhances the overall atom economy. The final catalytic hydrogenation step efficiently removes the benzyl protecting groups under mild conditions, yielding the target 3-(3,4-dihydroxyphenyl)-2-hydroxypropionate with exceptional optical purity and structural integrity.
Mechanistic Insights into Sharpless Asymmetric Dihydroxylation
The core of this technological advancement is the application of the Sharpless asymmetric dihydroxylation (AD) reaction, a transformation renowned for its ability to convert achiral alkenes into chiral vicinal diols with high enantioselectivity. In this specific application, the Z-3-(3,4-dibenzyloxyphenyl)acrylate serves as the substrate. The mechanism involves the coordination of the alkene to an osmium(VIII) species modified by a chiral ligand, such as (DHQ)2PHAL or (DHQD)2PHAL. These ligands create a chiral environment around the metal center, directing the approach of the oxidant to one specific face of the double bond. This facial selectivity is crucial, as it dictates whether the resulting product will possess the (2R, 3S) or (2S, 3R) configuration. The reaction proceeds through a cyclic osmate ester intermediate, which is subsequently hydrolyzed to release the chiral diol and regenerate the osmium catalyst. The use of co-oxidants like potassium ferricyanide ensures the continuous turnover of the osmium species, allowing for catalytic rather than stoichiometric usage of the expensive metal, which is a key factor in process economics.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over non-catalytic methods. Because the chirality is induced during the bond-forming event rather than through separation, the potential for generating the wrong enantiomer is minimized at the source. The patent data indicates that by selecting the appropriate ligand family—either the DHQ or DHQD derivatives—manufacturers can access either enantiomer with optical purity values exceeding 99% ee. This high level of stereocontrol drastically reduces the burden on downstream purification processes. Furthermore, the mild reaction conditions typically employed in AD reactions (often near room temperature in biphasic solvent systems) prevent thermal degradation of the sensitive catechol moiety, ensuring that the final impurity profile remains clean and manageable. This mechanistic robustness is essential for maintaining batch-to-batch consistency in a commercial manufacturing setting.
How to Synthesize 3-(3,4-Dihydroxyphenyl)-2-Hydroxypropionate Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly during the asymmetric induction phase. The process initiates with the protection of the phenolic hydroxyl groups of 3,4-dihydroxybenzaldehyde using benzyl chloride in the presence of a base, followed by condensation with malonic acid to form the acrylic acid derivative. Subsequent esterification yields the requisite alkene substrate. The critical step involves the asymmetric dihydroxylation, where the choice of ligand determines the absolute configuration of the product. Following the establishment of chirality, the benzyl protecting groups are removed via catalytic hydrogenation using palladium on carbon, a standard and scalable industrial procedure. For a detailed breakdown of the specific reagents, molar ratios, and workup procedures required to execute this synthesis according to the patent specifications, please refer to the standardized guide below.
- Protect 3,4-dihydroxybenzaldehyde using benzyl chloride and base to form 3,4-dibenzyloxybenzaldehyde.
- Perform Knoevenagel condensation with malonic acid followed by esterification to obtain Z-3-(3,4-dibenzyloxyphenyl)acrylate.
- Execute asymmetric dihydroxylation using osmium tetroxide and cinchona alkaloid ligands (e.g., (DHQ)2PHAL) to establish chirality.
- Conduct catalytic hydrogenation using Pd/C to remove benzyl protecting groups and yield the final chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric synthesis route offers compelling economic and operational benefits that extend beyond simple yield improvements. The transition from a resolution-based process to a direct asymmetric synthesis fundamentally alters the cost structure of the intermediate. By eliminating the need to synthesize and then discard half of the material (the unwanted enantiomer), the effective utilization of raw materials is drastically improved. This enhancement in atom economy translates directly into reduced raw material costs per kilogram of active product. Additionally, the removal of resolution steps—such as diastereomeric salt formation or chiral chromatography—simplifies the manufacturing workflow, reducing both the capital expenditure required for equipment and the operational expenditure associated with labor and utilities. The streamlined process flow also shortens the overall cycle time, enabling faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of racemic resolution steps removes a significant cost center from the production budget. Traditional resolution methods are inherently wasteful, often requiring multiple recrystallizations and resulting in substantial material loss. By achieving high enantiomeric excess directly through catalysis, this method avoids these losses entirely. Furthermore, the use of catalytic amounts of osmium, regenerated in situ by inexpensive co-oxidants, minimizes the consumption of precious metals. The reliance on commodity chemicals like 3,4-dihydroxybenzaldehyde and malonic acid ensures that the raw material supply chain remains stable and cost-effective, shielding the project from the volatility associated with specialized chiral pool starting materials.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route contributes significantly to supply chain security. The starting materials are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The reaction conditions are mild and do not require extreme temperatures or pressures, which enhances operational safety and reduces the likelihood of unplanned shutdowns due to equipment failure. Moreover, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, ensuring that the final product consistently meets quality specifications. This reliability is crucial for maintaining uninterrupted supply to downstream API manufacturers, particularly in the highly regulated cardiovascular therapeutic sector where quality deviations can lead to costly recalls.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry, such as filtration, extraction, and catalytic hydrogenation. The avoidance of stoichiometric chiral auxiliaries reduces the generation of chemical waste, aligning the process with green chemistry principles. The catalytic nature of the key step means less heavy metal waste is generated compared to stoichiometric oxidation methods. Additionally, the final deprotection step uses hydrogen and a heterogeneous catalyst, producing water as the primary byproduct, which simplifies wastewater treatment. These factors collectively lower the environmental footprint of the manufacturing process, facilitating easier regulatory compliance and reducing the costs associated with waste disposal and environmental monitoring.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is critical for making informed decisions about technology transfer and process validation.
Q: What is the primary advantage of this asymmetric synthesis method over traditional routes?
A: Unlike traditional methods that produce racemic mixtures requiring difficult resolution, this patent utilizes Sharpless asymmetric dihydroxylation to directly synthesize single enantiomers with high optical purity (>99% ee), significantly reducing material waste and processing steps.
Q: Which chiral ligands are suitable for controlling the stereochemistry in this process?
A: The process employs specific cinchona alkaloid-derived ligands such as (DHQ)2PHAL or (QN)2PHAL for the levorotatory isomer, and (DHQD)2PHAL or (QD)2PHAL for the dextrorotatory isomer, allowing precise control over the R or S configuration.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method utilizes readily available raw materials like 3,4-dihydroxybenzaldehyde and standard industrial catalysts like palladium on carbon, with mild reaction conditions that facilitate safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(3,4-Dihydroxyphenyl)-2-Hydroxypropionate Supplier
The technical sophistication of patent CN102030648B underscores the complexity involved in producing high-value chiral intermediates for the pharmaceutical industry. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust manufacturing realities. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of enantiomeric excess using chiral HPLC. We understand that consistency is paramount in the supply of cardiovascular intermediates, and our quality management systems are designed to guarantee batch-to-batch reproducibility that meets the highest international standards.
We invite potential partners to engage with our technical team to explore how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments, allowing you to validate the quality and viability of our manufacturing capabilities against your internal benchmarks. Let us collaborate to bring safer, more effective cardiovascular therapies to the market through superior chemical manufacturing.
