Advanced SmI2-Catalyzed Tetrahydrofuran Ring-Opening for High-Purity Silane Intermediates
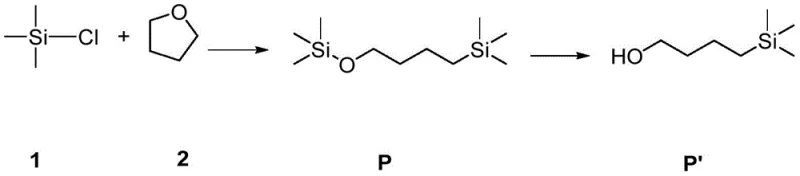
The landscape of organosilicon synthesis is undergoing a significant transformation driven by the need for safer, more efficient methodologies to access functionalized linear alkanes. A pivotal advancement in this domain is detailed in Chinese Patent CN113292589B, which discloses a novel method for the ring-opening double-silylation of tetrahydrofuran (THF). This technology addresses long-standing challenges in generating terminal bifunctional n-butane derivatives, which are critical precursors for high-purity silane intermediates used in pharmaceutical and material science applications. Traditionally, accessing these structures required harsh conditions or dangerous reagents, but this new approach utilizes a samarium iodide (SmI2) catalytic system to achieve high yields under remarkably mild conditions. For global procurement teams and R&D directors, this represents a strategic opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering complex building blocks with superior purity profiles and reduced operational risk.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of tetrahydrofuran via ring-opening has been plagued by severe inefficiencies and safety hazards. Conventional protocols often rely on the use of active alkali metals, which are pyrophoric and require stringent handling procedures, thereby increasing operational complexity and cost. Furthermore, earlier attempts using trimethylchlorosilane at elevated temperatures around 130°C resulted in poor selectivity and low yields, typically hovering around 58% after prolonged reaction times of 12 hours. Alternative methods involving metal oxides like alumina or titania suffered from similar issues, producing complex mixtures of trimethylsiloxybutane and chlorinated byproducts that were difficult to separate. These legacy processes not only compromise the economic viability of cost reduction in fine chemical manufacturing but also introduce significant impurity profiles that complicate downstream purification, making them unsuitable for sensitive applications such as API synthesis.
The Novel Approach
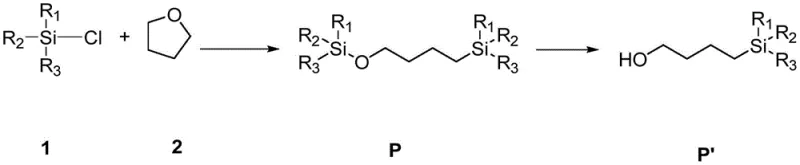
The methodology disclosed in CN113292589B offers a transformative solution by employing a reductive ring-opening strategy catalyzed by SmI2 in the presence of magnesium powder. This innovative route allows for the direct conversion of THF and chlorosilanes into bis-silylated targets in a single step, bypassing the need for hazardous alkali metals entirely. The process operates at温和 temperatures ranging from 40°C to 80°C, drastically reducing energy consumption and thermal stress on the equipment. Most notably, the introduction of a synergistic additive, tris(pentafluorophenyl)borane, enhances the catalytic activity to achieve GC yields of up to 89% for the intermediate and 82% for the final hydrolyzed alcohol. This leap in efficiency demonstrates the potential for commercial scale-up of complex organosilicon compounds, providing a robust platform for producing high-value intermediates with consistent quality.
Mechanistic Insights into SmI2-Catalyzed Reductive Ring-Opening
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the samarium catalyst system. Unlike simple nucleophilic substitutions, this reaction likely proceeds through a single-electron transfer (SET) mechanism initiated by the low-valent samarium species generated in situ from SmI2 and magnesium. This reactive species activates the C-O bond of the tetrahydrofuran ring, prompting a cleavage that generates a radical or anionic intermediate capable of trapping the silyl electrophile. The presence of magnesium serves as a stoichiometric reductant to regenerate the active catalytic species, ensuring the cycle continues efficiently without the accumulation of inactive byproducts. This mechanism avoids the formation of unstable alkoxides that typically revert to THF, a common pitfall in less optimized systems, thereby locking in the linear chain structure essential for downstream utility.
Furthermore, the impurity control mechanism is inherently superior due to the high chemoselectivity of the SmI2 catalyst. In traditional acid-catalyzed ring openings, competing polymerization or elimination reactions often degrade the product quality. However, the mild Lewis acidic nature of the borane additive combined with the specific redox potential of the samarium center directs the reaction exclusively toward the desired bis-silylation. This precision minimizes the formation of oligomeric siloxanes or chlorinated side products, simplifying the workup procedure to a basic aqueous quench and extraction. For R&D teams focused on reducing lead time for high-purity building blocks, this means fewer chromatography steps and higher overall throughput, directly translating to faster project timelines and lower solvent waste.
How to Synthesize 4-(Trimethylsilyl)butan-1-ol Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to strict inert atmosphere protocols to maintain catalyst activity. The process begins with the precise combination of magnesium powder, the chlorosilane substrate, and the THF feedstock within a sealed reactor. The addition of the SmI2 catalyst and the borane additive must be carefully metered to achieve the optimal molar ratios identified in the patent examples, specifically targeting a catalyst loading of roughly 0.004 to 0.009 equivalents relative to the substrate. Following the reaction period at controlled temperatures, the mixture undergoes a hydrolytic workup where water and dilute acid are introduced to cleave the silyl ether bond, releasing the final alcohol product. Detailed standardized operating procedures for this specific transformation are outlined below to ensure reproducibility and safety.
- In an inert atmosphere glovebox, combine magnesium powder, trimethylchlorosilane (TMSCl), tetrahydrofuran (THF), and the catalyst system comprising SmI2 and tris(pentafluorophenyl)borane.
- Seal the reaction vessel and heat the mixture to a controlled temperature between 40°C and 80°C, stirring continuously for approximately 24 hours to ensure complete conversion.
- Quench the reaction mixture with water and acid (e.g., 3M HCl), separate the organic phase, dry, and concentrate to isolate the high-purity 4-(trimethylsilyl)butan-1-ol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this SmI2-catalyzed technology offers profound benefits for supply chain resilience and cost management. By eliminating the reliance on active alkali metals, manufacturers can significantly reduce the costs associated with specialized storage, handling, and disposal of hazardous materials. The use of commercially available and inexpensive reagents such as magnesium powder and common chlorosilanes further drives down the raw material bill of materials, creating substantial margin opportunities for large-scale production. Additionally, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to extreme corrosion or high-pressure environments, facilitating easier technology transfer across different manufacturing sites.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous alkali metals removes a major cost driver from the production budget. Furthermore, the high selectivity of the reaction reduces the volume of solvents and adsorbents required for purification, leading to significant operational expenditure savings. The ability to run the reaction at lower temperatures also decreases energy consumption for heating and cooling cycles, contributing to a leaner and more sustainable manufacturing footprint that aligns with modern green chemistry initiatives.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including THF, trimethylchlorosilane, and magnesium, are commodity chemicals with robust global supply chains, minimizing the risk of procurement bottlenecks. The catalyst system, while specialized, uses components that are stable and easy to source compared to sensitive organometallic reagents. This stability ensures consistent production schedules and reduces the likelihood of batch failures due to reagent degradation, providing partners with a dependable source of critical intermediates for their own synthesis pipelines.
- Scalability and Environmental Compliance: The one-pot nature of the reaction simplifies the process flow, reducing the number of unit operations and the associated physical footprint of the plant. The avoidance of heavy metal catalysts or toxic byproducts simplifies wastewater treatment and waste disposal compliance, lowering the environmental regulatory burden. This streamlined process is inherently safer and more scalable, allowing for seamless transition from kilogram-scale development to multi-ton commercial production without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on reaction parameters and substrate scope. Understanding these nuances is essential for process chemists evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using SmI2 over traditional alkali metals for THF ring-opening?
A: Unlike traditional methods requiring hazardous active alkali metals or extreme temperatures (130°C), the SmI2-catalyzed process operates under mild conditions (40-80°C) with significantly higher selectivity and yield (up to 89%), eliminating safety risks associated with pyrophoric reagents.
Q: Can other catalysts besides Samarium Iodide be used in this protocol?
A: Yes, while SmI2 is preferred for optimal performance, the patent discloses that NaI, KI, and ZnI2 can also function as catalysts, offering flexibility in sourcing depending on specific supply chain requirements.
Q: What is the role of tris(pentafluorophenyl)borane in the reaction?
A: Tris(pentafluorophenyl)borane acts as a synergistic additive that works in concert with SmI2 to promote the ring-opening double-silylation, resulting in a '1+1>2' promotional effect that drastically improves reaction efficiency compared to using the catalyst alone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(Trimethylsilyl)butan-1-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies like the SmI2-mediated ring-opening described in CN113292589B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of silane intermediate meets the exacting standards required for pharmaceutical and electronic grade applications. We are committed to leveraging our technical expertise to optimize this specific chemistry for your unique volume and quality requirements.
We invite you to engage with our technical team to explore how this innovative synthesis route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your specific production volumes. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply security.
