Advanced Nickel-Catalyzed Synthesis of 1,1-Diyne Compounds for Scalable Pharmaceutical Manufacturing
Introduction to Breakthrough 1,1-Diyne Synthesis Technology
The landscape of organic synthesis for complex pharmaceutical intermediates is constantly evolving, driven by the need for more efficient and selective methodologies. A significant advancement in this field is detailed in Chinese Patent CN110981900A, which discloses a robust metal-catalyzed method for preparing 1,1-diacetylene compounds directly from terminal olefins. This technology represents a paradigm shift from traditional approaches that were often limited to activated substrates. By leveraging a specialized nickel catalytic system, this invention enables the direct functionalization of non-activated terminal olefins with excellent regioselectivity. For R&D directors and process chemists, this opens new avenues for constructing carbon skeletons found in bioactive molecules and drug lead compounds without the need for pre-functionalized starting materials. The ability to introduce two alkyne moieties simultaneously onto a terminal alkene backbone provides a versatile platform for generating highly functionalized architectures.
The strategic importance of this patent lies in its operational simplicity and the high value of the resulting 1,1-diyne scaffolds. These compounds serve as critical building blocks in the synthesis of natural products and advanced materials. The methodology described ensures that the transition from bench-scale discovery to pilot production is streamlined, addressing common pain points such as harsh reaction conditions and poor yield consistency. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge synthetic routes is essential for maintaining a competitive edge in the global market. The following analysis delves into the technical specifics, comparing this novel approach against conventional limitations and highlighting its commercial viability for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of olefins with alkyne groups has been predominantly restricted to activated systems, such as electron-deficient alkenes or those bearing specific directing groups. Conventional 1,2-alkyne functionalization strategies often suffer from limited substrate scope, requiring expensive and sensitive reagents that are not conducive to large-scale operations. Furthermore, achieving 1,1-diethynylation on non-activated terminal olefins has remained a significant challenge in organic chemistry, with few reported methods capable of delivering high selectivity and yield. Traditional processes frequently involve multi-step sequences, harsh thermal conditions, or stoichiometric amounts of toxic heavy metals, which complicate purification and increase environmental burdens. These limitations create bottlenecks in the supply chain for complex drug intermediates, leading to increased costs and extended lead times for procurement teams seeking reliable sources of high-purity materials.
The Novel Approach
In stark contrast, the method disclosed in CN110981900A utilizes a nickel-catalyzed system that effectively overcomes these barriers by enabling the direct transformation of inexpensive, non-activated terminal olefins. The core innovation involves the use of a nickel catalyst (such as NiI2), a specialized ligand, bis(pinacolato)diboron, and a base in an organic solvent. This combination facilitates a highly selective coupling reaction with triisopropylsilylacetylene bromide (TIPS-Br).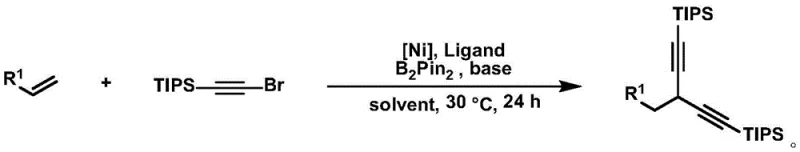 The reaction proceeds under remarkably mild conditions, typically at 30°C, which significantly reduces energy consumption compared to high-temperature alternatives. This novel approach not only simplifies the synthetic route by avoiding pre-activation steps but also ensures excellent regioselectivity, placing the alkyne groups precisely at the terminal position. The use of TIPS protecting groups adds another layer of versatility, allowing for subsequent orthogonal deprotection and functionalization, thereby expanding the utility of the final products in medicinal chemistry campaigns.
The reaction proceeds under remarkably mild conditions, typically at 30°C, which significantly reduces energy consumption compared to high-temperature alternatives. This novel approach not only simplifies the synthetic route by avoiding pre-activation steps but also ensures excellent regioselectivity, placing the alkyne groups precisely at the terminal position. The use of TIPS protecting groups adds another layer of versatility, allowing for subsequent orthogonal deprotection and functionalization, thereby expanding the utility of the final products in medicinal chemistry campaigns.
Mechanistic Insights into Nickel-Catalyzed 1,1-Diethynylation
The success of this transformation hinges on the intricate interplay between the nickel center and the tailored ligand environment. The catalytic cycle likely initiates with the oxidative addition of the alkynyl bromide to the nickel species, followed by transmetallation with the boron reagent to generate a nucleophilic nickel-alkynyl complex. The presence of the specific oxazoline-pyridine ligands, as illustrated in the patent data, is crucial for stabilizing the active catalytic species and controlling the stereochemical outcome of the reaction.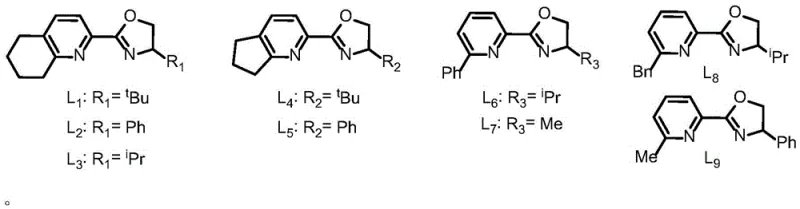 These ligands modulate the electronic and steric properties of the nickel center, facilitating the insertion of the terminal olefin and ensuring that the reaction proceeds with high 1,1-selectivity rather than forming linear or 1,2-isomers. The mechanistic pathway avoids the formation of unstable radical intermediates that could lead to polymerization or side reactions, thus ensuring a clean impurity profile which is vital for pharmaceutical applications.
These ligands modulate the electronic and steric properties of the nickel center, facilitating the insertion of the terminal olefin and ensuring that the reaction proceeds with high 1,1-selectivity rather than forming linear or 1,2-isomers. The mechanistic pathway avoids the formation of unstable radical intermediates that could lead to polymerization or side reactions, thus ensuring a clean impurity profile which is vital for pharmaceutical applications.
Furthermore, the utility of the resulting 1,1-diyne compounds extends beyond their immediate isolation. The patent highlights a secondary transformation where the silyl groups can be removed to generate highly reactive allene-alkyne motifs. This downstream processing capability is demonstrated by treating the protected diyne with TBAF in dichloromethane.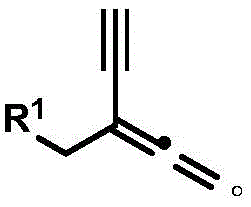 This step converts the stable, protected intermediate into a highly functionalized scaffold ready for cyclization or further coupling. For R&D teams, this means a single synthetic platform can access a diverse library of complex heterocycles and conjugated systems, accelerating the structure-activity relationship (SAR) studies required for drug discovery. The ability to control impurity formation through precise catalyst tuning ensures that the final API intermediates meet stringent purity specifications without extensive recrystallization.
This step converts the stable, protected intermediate into a highly functionalized scaffold ready for cyclization or further coupling. For R&D teams, this means a single synthetic platform can access a diverse library of complex heterocycles and conjugated systems, accelerating the structure-activity relationship (SAR) studies required for drug discovery. The ability to control impurity formation through precise catalyst tuning ensures that the final API intermediates meet stringent purity specifications without extensive recrystallization.
How to Synthesize 1,1-Diyne Compounds Efficiently
The practical implementation of this technology involves a straightforward one-pot procedure that is well-suited for both laboratory optimization and industrial scale-up. The process begins with the preparation of the catalytic mixture under an inert atmosphere to prevent oxidation of the sensitive nickel species. Key reagents including the nickel source, ligand, diboron ester, and base are dissolved in a polar aprotic solvent like N-methylpyrrolidone (NMP). The substrate olefin and the alkynyl bromide are then introduced, and the mixture is stirred at a controlled temperature. Detailed standardized synthesis steps follow below.
- Prepare the catalytic system by dissolving Nickel Iodide (NiI2), a specific oxazoline-pyridine ligand, bis(pinacolato)diboron (B2Pin2), and Lithium Methoxide (LiOMe) in anhydrous N-methylpyrrolidone (NMP) under an inert atmosphere.
- Add the non-activated terminal olefin substrate and triisopropylsilylacetylene bromide (TIPS-Br) to the reaction mixture, maintaining a molar ratio optimized for high regioselectivity.
- Stir the reaction at 30°C for 24 hours, followed by filtration, solvent removal, and purification via column chromatography to isolate the target 1,1-diyne product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this nickel-catalyzed methodology offers substantial benefits that directly address the priorities of procurement managers and supply chain heads. The primary advantage lies in the utilization of non-activated terminal olefins, which are commodity chemicals available in vast quantities at low cost. By eliminating the need for expensive, pre-functionalized starting materials, the overall cost of goods sold (COGS) for the final intermediate is significantly reduced. Additionally, the mild reaction conditions (30°C) translate to lower energy requirements and reduced stress on reactor equipment, enhancing the safety profile and operational longevity of manufacturing facilities. This efficiency allows for cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or yield.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts like palladium in favor of abundant nickel drastically lowers raw material costs. Furthermore, the high atom economy of the reaction minimizes waste generation, reducing disposal costs associated with hazardous byproducts. The simplified workup procedure, involving basic filtration and chromatography, streamlines the production timeline, allowing for faster turnover of batches and improved capital efficiency.
- Enhanced Supply Chain Reliability: Since the starting materials are bulk chemicals with established global supply chains, the risk of raw material shortages is minimized. The robustness of the catalytic system ensures consistent batch-to-batch reproducibility, which is critical for maintaining continuous supply to downstream customers. This reliability reduces lead time for high-purity pharmaceutical intermediates, enabling partners to meet tight project deadlines and regulatory submission schedules with confidence.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are manageable on a multi-ton scale. The reduced toxicity profile compared to traditional heavy-metal catalyzed processes aligns with increasingly strict environmental regulations. This facilitates easier permitting and compliance auditing, ensuring long-term operational continuity and supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the process parameters and scope. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development teams.
Q: What are the key advantages of this nickel-catalyzed method over traditional alkyne functionalization?
A: Unlike traditional methods that often require activated olefins or harsh conditions, this protocol utilizes non-activated terminal olefins under mild conditions (30°C), offering superior regioselectivity and broader substrate compatibility for complex molecule synthesis.
Q: Can the silyl protecting groups be removed for further downstream processing?
A: Yes, the triisopropylsilyl (TIPS) groups can be efficiently removed using TBAF in dichloromethane at room temperature, yielding highly functionalized 1,1-alkyne allene compounds suitable for further derivatization.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: The process utilizes commercially available reagents like NiI2 and standard solvents like NMP, and operates at low temperatures, making it highly amenable to scale-up for the commercial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Diyne Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed synthesis for the next generation of pharmaceutical intermediates. Our team of expert process chemists is equipped to adapt and optimize this technology for your specific molecular targets, ensuring seamless translation from gram-scale discovery to commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and reliability. Our state-of-the-art facilities feature rigorous QC labs and stringent purity specifications, ensuring that every batch of 1,1-diyne compound delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, efficiency, and trust.
