Advanced Photocatalytic Strategy for Scalable 1,2-Allenyl Ketone Production
Advanced Photocatalytic Strategy for Scalable 1,2-Allenyl Ketone Production
The construction of allene skeletons represents a critical challenge and opportunity in modern organic synthesis, particularly for the development of high-value pharmaceutical intermediates and advanced functional materials. Patent CN113773183A introduces a groundbreaking methodology for the photocatalytic synthesis of 1,2-allenyl ketones, addressing long-standing limitations in substrate universality and reaction conditions. This technology leverages a visible-light-promoted catalytic system to facilitate the addition, reduction, and isomerization of alkyl radicals with 1,3-enyne compounds. By shifting away from harsh thermal or cryogenic requirements, this innovation offers a robust pathway for generating polysubstituted allenes with exceptional functional group tolerance. For global supply chain leaders, this represents a significant advancement in process chemistry, enabling the production of complex molecular architectures under mild, operationally simple conditions that are conducive to large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trisubstituted 1,2-dienyl ketones has relied heavily on ionic pathways involving organometallic reagents. A classic approach utilizes alkyl zinc reagents reacting with 1,3-enyne compounds, but this necessitates extremely low temperatures, typically around -78 °C, and the use of pyrophoric materials that pose significant safety hazards in large-scale operations. 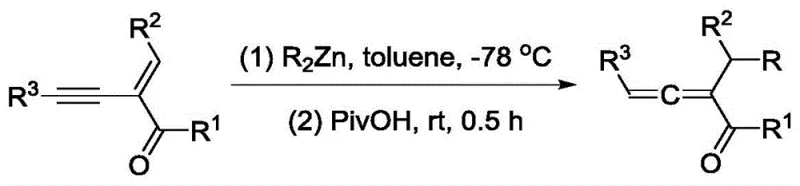 Furthermore, alternative radical pathways utilizing copper catalysis have been explored; however, these methods often suffer from limited substrate scope and are frequently restricted to specific types of radical intermediates, such as nitrogen-centered radicals, rather than the more versatile carbon-centered radicals required for diverse chemical libraries. These conventional techniques not only incur high energy costs due to cryogenic cooling but also generate substantial metal waste, complicating purification processes and increasing the environmental footprint of the manufacturing workflow.
Furthermore, alternative radical pathways utilizing copper catalysis have been explored; however, these methods often suffer from limited substrate scope and are frequently restricted to specific types of radical intermediates, such as nitrogen-centered radicals, rather than the more versatile carbon-centered radicals required for diverse chemical libraries. These conventional techniques not only incur high energy costs due to cryogenic cooling but also generate substantial metal waste, complicating purification processes and increasing the environmental footprint of the manufacturing workflow.
The Novel Approach
In stark contrast to these legacy methods, the disclosed invention employs a visible-light photocatalytic strategy that operates efficiently at room temperature. This novel approach utilizes an Iridium-based photosensitizer to generate alkyl radicals from stable silicate precursors, which then undergo addition to 1,3-enynes followed by isomerization to yield the desired 1,2-allenyl ketones. 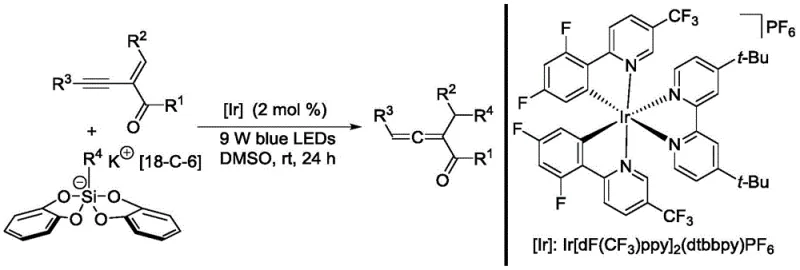 The elimination of cryogenic conditions and stoichiometric metal reagents marks a paradigm shift in process efficiency. By utilizing 9W blue LEDs as the energy source, the reaction maintains high atom economy and operational simplicity. This method effectively overcomes the defects of existing ionic and radical pathways, providing a new strategy for the synthesis of polysubstituted allenes that is both economically viable and environmentally superior, aligning perfectly with the goals of green chemistry in fine chemical manufacturing.
The elimination of cryogenic conditions and stoichiometric metal reagents marks a paradigm shift in process efficiency. By utilizing 9W blue LEDs as the energy source, the reaction maintains high atom economy and operational simplicity. This method effectively overcomes the defects of existing ionic and radical pathways, providing a new strategy for the synthesis of polysubstituted allenes that is both economically viable and environmentally superior, aligning perfectly with the goals of green chemistry in fine chemical manufacturing.
Mechanistic Insights into Visible-Light Photocatalytic Isomerization
The core of this technological breakthrough lies in the sophisticated interplay between the photocatalyst and the radical precursor. The reaction initiates when the Iridium complex, specifically Ir[dF(CF3)ppy]2(dtbbpy)PF6, absorbs photons from the blue LED source to reach an excited state. This excited species facilitates a single-electron transfer (SET) process with the alkyl bis(catechol)silicate-18-crown-6-potassium salt, triggering the fragmentation of the silicate to release a reactive carbon-centered alkyl radical. This radical then selectively adds to the triple bond of the 2-acyl-1,3-enyne substrate, forming a vinyl radical intermediate. Subsequent reduction and protonation steps, driven by the catalytic cycle, lead to the formation of the allene skeleton through a precise isomerization sequence. This mechanistic pathway ensures high regioselectivity and minimizes the formation of unwanted byproducts, which is crucial for maintaining high purity in pharmaceutical applications.
From an impurity control perspective, the mild reaction conditions play a pivotal role in ensuring product quality. Traditional methods operating at -78 °C or with aggressive Lewis acids often promote side reactions such as polymerization or non-selective additions, leading to complex impurity profiles that are difficult to separate. In this photocatalytic system, the gentle generation of radicals at room temperature allows for better kinetic control over the reaction trajectory. The use of DMSO as a solvent further stabilizes the ionic and radical species involved, preventing premature decomposition. Consequently, the resulting 1,2-allenyl ketones exhibit cleaner crude profiles, reducing the burden on downstream purification steps like column chromatography and distillation, thereby enhancing the overall yield and consistency of the final active pharmaceutical ingredient (API) intermediate.
How to Synthesize 1,2-Allenyl Ketone Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard laboratory and pilot plant settings. The process begins by combining the photosensitizer, the 1,3-enyne substrate, and the radical precursor in dry dimethyl sulfoxide (DMSO) under an inert nitrogen atmosphere. The reaction mixture is then subjected to irradiation using 9W blue LEDs at room temperature for approximately 24 hours, allowing sufficient time for the photocatalytic cycle to reach completion. Following the reaction, a straightforward workup procedure involving extraction with ethyl acetate, washing with brine, and drying over magnesium sulfate yields the crude product.
- Combine photosensitizer Ir[dF(CF3)ppy]2(dtbbpy)PF6, 1,3-enyne compound, and alkyl bis(catechol)silicate radical precursor in dry DMSO under nitrogen.
- Irradiate the reaction mixture with 9W blue LEDs at room temperature for 24 hours while stirring.
- Quench with saturated Na2CO3, extract with ethyl acetate, dry over MgSO4, and purify via column chromatography using petroleum ether/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers transformative benefits regarding cost structure and operational reliability. The shift from cryogenic conditions to ambient temperature processing eliminates the need for specialized low-temperature equipment and the associated high energy consumption, leading to significantly reduced utility costs per kilogram of product. Furthermore, the replacement of hazardous alkyl zinc reagents with stable, shelf-stable silicate salts enhances workplace safety and reduces the regulatory burden associated with handling pyrophoric materials. This simplification of the input material profile ensures a more resilient supply chain, less susceptible to disruptions caused by the scarcity of specialized reagents.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the drastic simplification of the reaction infrastructure and purification workflows. By eliminating the requirement for stoichiometric transition metals like copper or zinc, the process avoids the costly and time-consuming steps of heavy metal scavenging and removal, which are mandatory for pharmaceutical grade intermediates. Additionally, the use of low-loading photocatalysts (2 mol%) compared to stoichiometric reagents minimizes raw material expenses. The mild conditions also reduce equipment wear and tear, extending the lifecycle of reactor vessels and lowering capital expenditure requirements for new production lines dedicated to allene synthesis.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 1,3-enynes and organosilicates, mitigates the risk of supply bottlenecks often associated with sensitive organometallic reagents. The robustness of the reaction across a wide range of substrates means that a single production line can be adapted to manufacture various derivatives without extensive requalification, providing flexibility to meet fluctuating market demands. This versatility ensures consistent delivery schedules for downstream customers, as the process is less prone to batch failures caused by reagent instability or stringent environmental controls.
- Scalability and Environmental Compliance: Scaling this photochemical process is facilitated by the modularity of LED lighting systems, which can be easily arranged in flow reactors or large batch vessels to ensure uniform light penetration. The absence of volatile organic solvents like toluene in favor of DMSO, combined with the lack of toxic metal waste, aligns the process with increasingly stringent environmental regulations. This eco-friendly profile not only reduces waste disposal costs but also enhances the sustainability credentials of the final product, a key factor for multinational corporations aiming to reduce their carbon footprint in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this technology for industrial applications.
Q: What are the advantages of this photocatalytic method over traditional ionic pathways?
A: Unlike traditional ionic pathways that require cryogenic conditions (-78 °C) and pyrophoric alkyl zinc reagents, this photocatalytic method operates at room temperature using stable radical precursors, significantly improving operational safety and energy efficiency.
Q: Does this method avoid heavy metal contamination issues?
A: Yes, while it uses an Iridium photocatalyst, the loading is very low (2 mol%), and the process avoids stoichiometric amounts of transition metals like Copper or Zinc found in other radical or ionic methods, simplifying downstream purification and metal removal.
Q: Is the substrate scope suitable for complex pharmaceutical intermediates?
A: The method demonstrates excellent functional group compatibility, tolerating various substituents on the 1,3-enyne and radical precursor, making it highly suitable for synthesizing diverse and complex pharmaceutical intermediates without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Allenyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this photocatalytic innovation for the next generation of pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to leveraging this advanced chemistry to deliver high-quality 1,2-allenyl ketones that empower your drug development pipelines.
We invite you to collaborate with our technical team to explore how this cost-effective and sustainable synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your target molecules. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing.
