Scalable Photocatalytic Ortho-Fluorination of Phenols for Advanced Fine Chemical Manufacturing
Scalable Photocatalytic Ortho-Fluorination of Phenols for Advanced Fine Chemical Manufacturing
The landscape of fine chemical synthesis is undergoing a transformative shift towards greener, more efficient methodologies, particularly in the realm of C-H functionalization. A groundbreaking approach detailed in patent CN110950739A introduces a novel method for the direct ortho-position fluorination of phenolic compounds, bypassing the limitations of traditional multi-step syntheses. This technology leverages visible-light photocatalysis to achieve high regioselectivity under remarkably mild conditions, specifically at room temperature. By utilizing commercially available fluorinating agents such as Selectfluor and inexpensive organic photocatalysts like Eosin Y, this process offers a streamlined pathway to produce valuable fluorinated phenol derivatives. These compounds serve as critical building blocks in the development of pharmaceuticals, agrochemicals, and advanced materials, where the introduction of a fluorine atom can drastically alter metabolic stability and binding affinity. The significance of this patent lies not only in its chemical elegance but also in its potential for immediate industrial application, offering a robust alternative to legacy methods that often suffer from poor atom economy and harsh operational requirements.
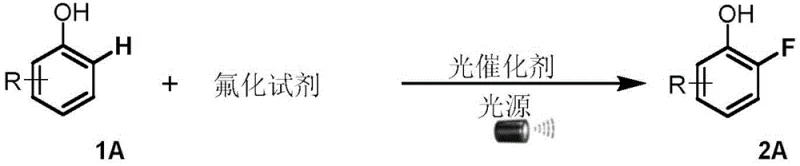
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of phenolic compounds to introduce halogen substituents, particularly fluorine, has been a challenging endeavor for process chemists. Traditional synthetic routes typically necessitate a tedious sequence of protection and deprotection steps to prevent unwanted side reactions at the hydroxyl group. This classical approach often involves the use of strong bases, hazardous reagents, and elevated temperatures, which not only increase the operational complexity but also pose significant safety risks in a manufacturing environment. Furthermore, the reliance on stoichiometric amounts of expensive directing groups or transition metal catalysts can lead to substantial cost inflation and generate large volumes of toxic waste. The low reaction yields and poor site selectivity associated with these older methods frequently result in difficult purification processes, requiring extensive chromatography or recrystallization to remove isomeric impurities. Consequently, the overall production timeline is extended, and the cost of goods sold (COGS) for the final fluorinated intermediate becomes prohibitively high for many commercial applications.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct C-H activation strategy driven by organic photocatalysis, effectively eliminating the need for pre-functionalization or protecting groups. By employing a simple system comprising a phenol substrate, a fluorinating reagent, and a photocatalyst in a benign solvent system like aqueous acetic acid, the reaction proceeds efficiently at room temperature under visible light irradiation. This one-step protocol dramatically simplifies the workflow, reducing the time from raw material to finished product. The use of Eosin Y, a cheap and environmentally friendly organic dye, replaces costly and potentially toxic noble metal catalysts, aligning the process with green chemistry principles. Moreover, the reaction demonstrates exceptional ortho-selectivity, driven by the intrinsic electronic properties of the phenol ring and the specific radical mechanism, ensuring that the fluorine atom is installed precisely where it is needed without generating significant amounts of meta- or para-isomers. This level of precision minimizes downstream purification burdens and maximizes the overall yield of the desired pharmaceutical or agrochemical intermediate.
Mechanistic Insights into Organic Photocatalytic C-H Fluorination
From a mechanistic perspective, this transformation represents a sophisticated interplay between photo-excited states and radical chemistry. Upon irradiation with blue light, the photocatalyst (such as Eosin Y) absorbs photons to reach an excited state, which then facilitates a single-electron transfer (SET) process with the fluorinating reagent, typically Selectfluor. This interaction generates a highly reactive fluorine radical species capable of abstracting a hydrogen atom or adding to the aromatic ring. The hydroxyl group on the phenol substrate plays a crucial dual role: it activates the aromatic ring towards electrophilic attack and acts as a directing group to ensure the fluorination occurs exclusively at the ortho-position. The resulting cyclohexadienyl radical intermediate subsequently undergoes oxidation and deprotonation to restore aromaticity, yielding the stable ortho-fluorophenol product. Understanding this catalytic cycle is vital for R&D teams, as it highlights the sensitivity of the reaction to light intensity and oxygen levels, parameters that must be tightly controlled during scale-up to maintain consistent quality and reproducibility across different batch sizes.
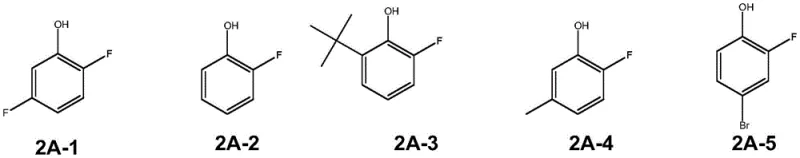
Furthermore, the robustness of this catalytic system is evidenced by its broad substrate scope, accommodating a wide variety of functional groups without compromising efficiency. As illustrated in the patent examples, substrates containing electron-withdrawing groups like halogens (chlorine, bromine) and electron-donating groups like alkyl or alkoxy chains all undergo smooth fluorination. Even sensitive functionalities such as esters and ketones remain intact under these mild, neutral-to-acidic conditions, which is a significant advantage over traditional harsh fluorination methods that might degrade such moieties. The ability to tolerate diverse substituents implies that this method can be applied late-stage in the synthesis of complex drug candidates, allowing medicinal chemists to rapidly generate fluorinated analogs for structure-activity relationship (SAR) studies. The high purity of the resulting compounds, confirmed by NMR and mass spectrometry data in the patent, suggests that the reaction generates minimal byproducts, thereby simplifying the impurity profile and facilitating regulatory approval for pharmaceutical applications.
How to Synthesize Ortho-Fluorinated Phenols Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters outlined in the patent to ensure optimal performance. The standard procedure involves dissolving the phenolic starting material and the Eosin Y catalyst in a 10% aqueous acetic acid solution, followed by the addition of Selectfluor as the fluorine source. The mixture is then subjected to irradiation from a 12W blue LED light source at room temperature for a duration of approximately 6 hours. Following the reaction, the workup is straightforward: the mixture is extracted with an organic solvent like dichloromethane, dried over anhydrous sodium sulfate, and concentrated. The crude product is typically purified via silica gel column chromatography using a gradient of ethyl acetate and petroleum ether. For detailed operational specifics and safety guidelines regarding the handling of fluorinating agents and light sources, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by dissolving the phenolic substrate and Eosin Y photocatalyst in a 10% aqueous acetic acid solution.
- Add the fluorinating agent (preferably Selectfluor) to the solution and irradiate the mixture with a 12W blue light source at room temperature for approximately 6 hours.
- Upon completion, extract the product with dichloromethane, dry the organic layer, and purify the crude residue via silica gel column chromatography to obtain the high-purity fluorinated phenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic fluorination technology presents a compelling value proposition centered on cost efficiency and supply reliability. By shifting from a multi-step, protection-heavy synthesis to a direct, one-step process, manufacturers can significantly reduce the consumption of raw materials and solvents, leading to a leaner and more cost-effective production model. The elimination of expensive transition metal catalysts in favor of ubiquitous organic dyes like Eosin Y removes a major cost driver and mitigates the supply risk associated with scarce precious metals. Additionally, the mild reaction conditions—operating at room temperature and atmospheric pressure—lower the energy requirements for heating and cooling, contributing to a smaller carbon footprint and reduced utility costs. These factors collectively enhance the economic viability of producing fluorinated phenol intermediates, making them more accessible for large-scale applications in the pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the drastic simplification of the synthetic route. By removing the need for hydroxyl protection and deprotection steps, the process saves on reagents, labor, and time, effectively cutting the number of unit operations required. The use of Selectfluor, while a specialized reagent, is balanced by the high atom efficiency and the avoidance of stoichiometric metal waste. Furthermore, the ability to use aqueous acetic acid as a solvent reduces the reliance on expensive, anhydrous organic solvents, lowering both material costs and waste disposal fees associated with hazardous solvent treatment.
- Enhanced Supply Chain Reliability: The reliance on commercially available, commodity-grade chemicals ensures a stable supply chain. Eosin Y and acetic acid are produced in massive quantities globally, insulating the manufacturing process from the volatility often seen in the market for specialized organometallic catalysts. The robustness of the reaction against moisture and oxygen (to a reasonable extent) also means that stringent inert atmosphere conditions are less critical, allowing for more flexible manufacturing schedules and reducing the risk of batch failures due to minor environmental fluctuations. This reliability translates to shorter lead times and more predictable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but advancements in flow chemistry and LED technology have made this increasingly feasible. The mild nature of this reaction makes it inherently safer to scale up compared to exothermic traditional fluorinations that require cryogenic conditions. From an environmental standpoint, the generation of less hazardous waste and the use of a metal-free catalyst align with increasingly strict global environmental regulations. This compliance reduces the administrative burden of waste reporting and permits, facilitating smoother operations in regions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ortho-fluorination technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows or R&D pipelines.
Q: What are the advantages of using Eosin Y over transition metal catalysts for phenol fluorination?
A: Eosin Y is an inexpensive, organic, metal-free photocatalyst that eliminates the risk of heavy metal contamination in the final API or intermediate, significantly simplifying downstream purification and regulatory compliance compared to Ruthenium or Iridium complexes.
Q: Does this photocatalytic method require harsh reaction conditions or protecting groups?
A: No, the method operates at room temperature using visible light and does not require the traditional protection and deprotection of the hydroxyl group, thereby reducing the number of synthetic steps and improving overall atom economy.
Q: What is the regioselectivity of this fluorination reaction?
A: The reaction exhibits high ortho-site selectivity due to the directing effect of the hydroxyl group and the specific radical mechanism involved, ensuring the fluorine atom is installed precisely at the position adjacent to the hydroxyl group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Fluorophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality fluorinated intermediates in modern drug discovery and agricultural science. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex fluorinated phenols or reliable supply of standard intermediates, our infrastructure is designed to support your long-term growth and innovation goals.
We invite you to collaborate with us to explore how this advanced photocatalytic technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Let us be your partner in driving efficiency and excellence in fine chemical manufacturing.
