Scalable Synthesis of (2R,4R)-4-Fluoropyrrolidine-2-Carboxylic Acid via Safe DeoxoFluor Chemistry
The pharmaceutical industry continuously demands robust and safe methodologies for synthesizing complex fluorinated building blocks, particularly for the development of next-generation peptidomimetics and protease inhibitors. Patent CN116396201A introduces a groundbreaking preparation method for (2R,4R)-4-fluoropyrrolidine-2-carboxylic acid, a critical chiral intermediate with the CAS number 913820-71-0. This novel protocol addresses significant safety and scalability bottlenecks associated with legacy synthetic routes by replacing hazardous cryogenic fluorination with a milder, more controllable DeoxoFluor-mediated process. By utilizing (2R,4S)-N-Boc-4-hydroxy-proline as a readily available starting material, the invention establishes a streamlined four-step sequence that ensures high stereochemical integrity while drastically improving operational safety profiles. For global supply chain leaders and R&D directors, this technology represents a pivotal shift towards greener, more reliable manufacturing of high-value fluorinated amino acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
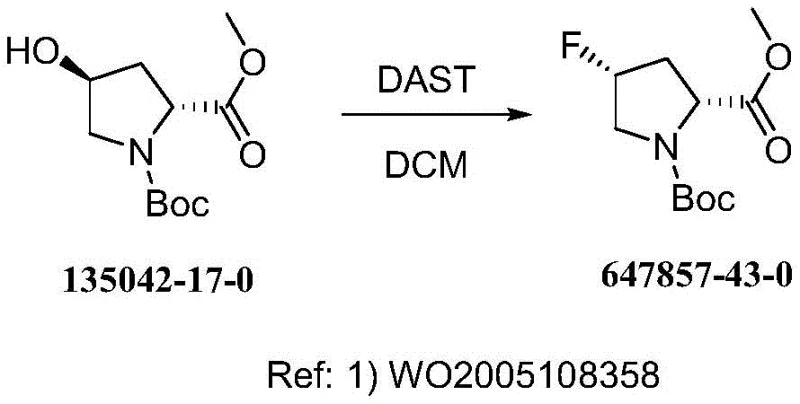
Historically, the synthesis of 4-fluoropyrrolidine derivatives has been plagued by the reliance on diethylaminosulfur trifluoride (DAST) as the primary fluorinating agent. As illustrated in the prior art reaction scheme, this conventional approach necessitates extremely harsh cryogenic conditions, typically requiring reaction temperatures as low as -78°C to maintain reagent stability and control exothermicity. Such ultra-low temperature requirements impose severe constraints on industrial scalability, demanding expensive specialized cooling infrastructure and significantly increasing energy consumption. Furthermore, DAST is notoriously thermally unstable; during post-reaction workup or accidental heating, it can undergo rapid and violent decomposition, posing substantial safety risks to personnel and facilities. These inherent dangers, coupled with the difficulty of maintaining consistent quality control under such extreme conditions, have long hindered the efficient mass production of this valuable pharmaceutical intermediate.
The Novel Approach
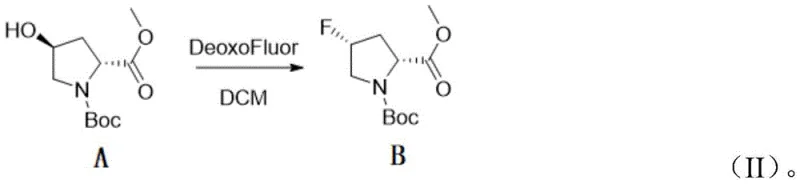
In stark contrast to the perilous legacy methods, the patented process leverages the superior stability and reactivity profile of DeoxoFluor (bis(2-methoxyethyl)aminosulfur trifluoride). This modern reagent allows the critical fluorination step to proceed efficiently at much milder temperatures, ranging from 0°C to ambient room temperature, thereby eliminating the need for energy-intensive cryogenic cooling. The transition to DeoxoFluor not only mitigates the risk of thermal runaway and explosive decomposition but also simplifies the reactor setup, making the process inherently safer and more amenable to large-scale batch operations. By operating under these benign conditions, manufacturers can achieve consistent conversion rates and minimize the formation of thermal degradation byproducts, ensuring a cleaner reaction profile. This strategic substitution of reagents fundamentally transforms the manufacturing landscape, turning a hazardous, niche laboratory procedure into a robust, commercially viable industrial process.
Mechanistic Insights into DeoxoFluor-Mediated Stereoinversion
The core chemical transformation in this synthesis is the nucleophilic substitution of the hydroxyl group with a fluorine atom, which proceeds with precise stereochemical control. The mechanism involves the activation of the 4-hydroxyl group by the sulfur center of the DeoxoFluor reagent, forming a reactive intermediate that facilitates an SN2-type displacement. This backside attack by the fluoride ion results in a complete inversion of configuration at the C4 position, successfully converting the (4S)-hydroxy precursor into the desired (4R)-fluoro product. The mild reaction conditions provided by DeoxoFluor are crucial here, as they prevent competing elimination reactions or racemization that often occur under the harsher thermal stress associated with DAST. Consequently, the process delivers the target intermediate with exceptional optical purity, preserving the chiral integrity essential for downstream biological activity in final drug substances.
Beyond stereocontrol, the refined reaction conditions play a pivotal role in impurity management and overall product quality. The avoidance of extreme cold and the use of a more stable fluorinating agent significantly reduce the generation of sulfur-containing byproducts and elimination impurities that are difficult to remove. The subsequent steps, including Boc deprotection with trifluoroacetic acid and ester hydrolysis with lithium hydroxide, are performed under standard conditions that further safeguard the molecule against degradation. This holistic approach to process design ensures that the final (2R,4R)-4-fluoropyrrolidine-2-carboxylic acid meets stringent purity specifications required for GMP manufacturing. For quality assurance teams, this means a more predictable impurity profile and reduced burden on purification processes, ultimately leading to higher yields of pharmaceutical-grade material.
How to Synthesize (2R,4R)-4-Fluoropyrrolidine-2-Carboxylic Acid Efficiently
The synthesis begins with the esterification of the starting material to protect the carboxylic acid, followed by the key fluorination step that installs the fluorine atom with inverted stereochemistry. Subsequent removal of the amine protecting group and hydrolysis of the ester yields the final zwitterionic product. This sequence is designed for operational simplicity, utilizing common organic solvents and avoiding exotic catalysts.
- Perform methyl esterification of the starting hydroxy-proline derivative using methanol and p-toluenesulfonic acid under reflux conditions.
- Execute stereoselective fluorination using DeoxoFluor reagent in dichloromethane at 0°C to room temperature to invert the stereochemistry.
- Remove the Boc protecting group using trifluoroacetic acid (TFA) followed by basic hydrolysis with LiOH to yield the final free acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented methodology offers profound economic and logistical benefits that extend far beyond simple yield improvements. By eliminating the dependency on cryogenic infrastructure, facilities can repurpose existing standard reactors for this synthesis, drastically reducing capital expenditure requirements for new production lines. The enhanced safety profile of DeoxoFluor compared to DAST also translates into lower insurance premiums and reduced regulatory hurdles related to hazardous material handling. Furthermore, the robustness of the process ensures consistent batch-to-batch reliability, minimizing the risk of costly production delays or failed batches that can disrupt the supply of critical API intermediates to downstream customers.
- Cost Reduction in Manufacturing: The shift from cryogenic (-78°C) to near-ambient temperature processing results in a massive reduction in energy consumption, as there is no longer a need for continuous operation of low-temperature chillers or liquid nitrogen systems. Additionally, the higher stability of the reagents reduces waste disposal costs associated with quenching hazardous decomposition products, leading to substantial overall cost savings in the production budget. The simplified workup procedures also decrease solvent usage and labor hours, further driving down the cost of goods sold for this high-value intermediate.
- Enhanced Supply Chain Reliability: DeoxoFluor is a commercially stable reagent with a longer shelf life and safer transport classification compared to the highly reactive and unstable DAST. This stability ensures a more secure raw material supply chain, reducing the risk of shortages due to storage degradation or transport restrictions. The ability to run the reaction at room temperature also increases plant flexibility, allowing for faster turnaround times between batches and enabling manufacturers to respond more agilely to fluctuating market demands for fluorinated pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard solvents like dichloromethane and tetrahydrofuran that are easily recovered and recycled in modern solvent recovery units. The absence of extreme thermal hazards simplifies the safety validation process for larger reactor vessels, facilitating a smoother transition from pilot plant to commercial tonnage production. Moreover, the improved atom economy and reduced formation of toxic byproducts align with green chemistry principles, helping companies meet increasingly stringent environmental regulations and sustainability goals without compromising production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel fluorination technology. These insights are derived directly from the experimental data and safety assessments detailed in the patent documentation, providing a clear understanding of the process capabilities.
Q: Why is DeoxoFluor preferred over DAST for this fluorination?
A: DeoxoFluor offers superior thermal stability compared to DAST, eliminating the need for hazardous cryogenic conditions (-78°C) and reducing the risk of explosive decomposition during scale-up.
Q: What is the stereochemical outcome of the fluorination step?
A: The reaction proceeds via an SN2 mechanism, resulting in the inversion of configuration from the (4S)-hydroxy precursor to the desired (4R)-fluoro product with high stereoselectivity.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process operates at mild temperatures (0°C to room temperature) and uses standard solvents like DCM and THF, making it highly adaptable for multi-kilogram commercial production without specialized cryogenic infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2R,4R)-4-Fluoropyrrolidine-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of innovative therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific safety and processing requirements of fluorination chemistry, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee product consistency. We are committed to delivering (2R,4R)-4-fluoropyrrolidine-2-carboxylic acid that meets the highest international standards, supporting your pipeline with reliable supply and technical excellence.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can enhance your project's economics and timeline. Contact our Customized Cost-Saving Analysis department today to discuss your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments tailored to your unique development needs, ensuring a partnership that drives value and innovation in your drug discovery programs.
