Advanced Impurity Removal for Progesterone: Ensuring Pharmaceutical Grade Purity and Scalability
The pharmaceutical industry faces continuous challenges in maintaining the stringent purity profiles required for steroid hormone active pharmaceutical ingredients (APIs), particularly when dealing with complex synthesis byproducts. Patent CN113956317A introduces a groundbreaking impurity removal method specifically designed for progesterone, addressing the persistent issue of bis-aldehyde impurities that often plague crude batches. This innovation is critical for manufacturers aiming to comply with the European Pharmacopoeia 9.0, which mandates that bis-aldehydic impurities (2) and (3) must remain below 0.6% to avoid product rejection. The disclosed technology leverages a clever chemical differentiation strategy, transforming problematic aldehyde contaminants into acetal derivatives that can be easily separated from the target molecule through solubility manipulation. By shifting the purification paradigm from harsh oxidative treatments to mild, selective acetalization, this method offers a robust pathway for producing high-purity progesterone with an HPLC content exceeding 99.5% and total aldehyde impurities reduced to less than 0.2%. For global supply chains, this represents a significant advancement in reliable API intermediate supplier capabilities, ensuring consistent quality without the environmental burden of heavy metal waste.
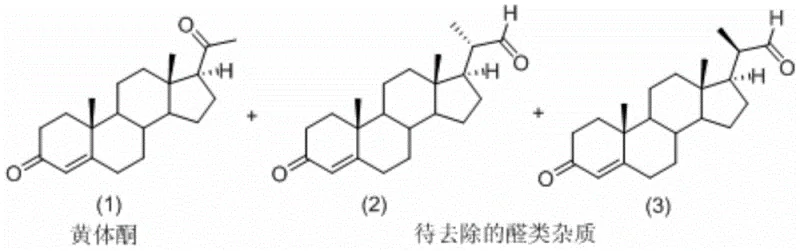
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of progesterone containing aldehyde impurities has relied on oxidative treatments, such as the chromic anhydride method detailed in comparative examples within the patent literature. These conventional approaches suffer from severe drawbacks, primarily due to the use of toxic heavy metal oxidants like chromium trioxide, which introduce significant environmental hazards and require complex waste disposal protocols. Furthermore, oxidative conditions are inherently harsh and non-selective, often leading to the degradation of the sensitive steroid backbone or the formation of new, unpredictable impurities alongside the intended purification. In the comparative data provided, the traditional chromic anhydride method resulted in a product with only 95.0% HPLC purity and an aldehyde impurity content of 0.82%, failing to meet the stringent pharmacopoeial limits. The presence of heavy metal residues also necessitates additional downstream processing steps for removal, increasing both the operational complexity and the overall cost of goods sold. Consequently, these legacy methods pose a substantial risk to cost reduction in pharmaceutical intermediates manufacturing, as they struggle to balance yield, purity, and regulatory compliance effectively.
The Novel Approach
In stark contrast, the novel acetalization-based purification method described in CN113956317A offers a温和 (mild) and highly selective alternative that circumvents the pitfalls of oxidative degradation. By utilizing common alcohol organic solvents such as methanol or ethanol in the presence of a catalytic amount of acid, the process specifically targets the aldehyde functional groups on the impurities while leaving the progesterone ketone functionality intact under controlled conditions. This chemical transformation converts the structurally similar bis-aldehyde impurities into acetals, which exhibit markedly different physical properties, particularly solubility, compared to the native progesterone. The subsequent crystallization step allows the pure progesterone to precipitate out of the solution while the acetalized impurities remain dissolved in the mother liquor, achieving a separation efficiency that recrystallization alone cannot accomplish. This approach not only eliminates the need for toxic oxidants but also simplifies the workflow to a series of dissolution, reaction, neutralization, and filtration steps, thereby enhancing the commercial scale-up of complex hormones. The result is a streamlined process that delivers superior purity metrics with a total mass yield consistently higher than 90%, demonstrating clear advantages for modern green chemistry initiatives.
Mechanistic Insights into Acid-Catalyzed Acetalization and Solubility Differentiation
The core mechanism driving this purification breakthrough relies on the differential reactivity of carbonyl groups under acidic conditions in an alcoholic medium. As illustrated in the reaction scheme, the bis-aldehyde impurities (2) and (3) possess aldehyde moieties that are highly susceptible to nucleophilic attack by alcohol molecules (R-OH) when activated by an acid catalyst such as p-toluenesulfonic acid or malonic acid. This reaction proceeds through a hemiacetal intermediate to form a stable diethyl or dimethyl acetal derivative, effectively masking the polar aldehyde group and altering the molecule's overall polarity and crystal lattice energy. Conversely, the C3 and C20 ketone groups on the progesterone molecule (1) are significantly less reactive towards acetal formation under these specific mild conditions, or any transient species formed are unstable and revert to the ketone, ensuring the API remains chemically unchanged. This selectivity is the linchpin of the process, allowing for the chemical modification of contaminants without affecting the product integrity. The transformation creates a distinct chemical species (acetals 4 and 5) that no longer co-crystallizes with progesterone, setting the stage for physical separation.
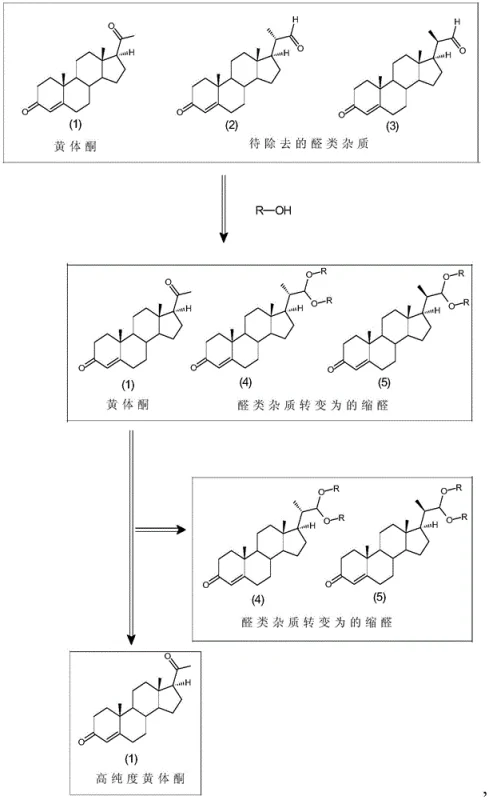
Following the chemical conversion, the purification leverages solubility differentiation to achieve final isolation. Once the aldehyde impurities have been fully converted to their acetal forms, the reaction mixture is concentrated to remove the primary alcohol solvent, and a secondary organic solvent B, such as methyl tert-butyl ether or ethyl acetate, is introduced. The choice of Solvent B is critical; it is selected based on its ability to keep the newly formed acetal impurities in solution while having poor solubility for progesterone at low temperatures. Upon cooling the mixture to a range of -20°C to 5°C, the solubility of progesterone drops precipitously, causing it to nucleate and grow as high-purity crystals. Meanwhile, the acetalized impurities, being more soluble in this specific solvent system, remain in the supernatant mother liquor. This thermodynamic driving force ensures that the impurities are physically excluded from the crystal lattice of the product. The final filtration step thus yields solid progesterone with drastically reduced impurity levels, validating the efficacy of combining chemical derivatization with physical crystallization for difficult separations.
How to Synthesize High-Purity Progesterone Efficiently
Implementing this purification protocol requires precise control over reaction parameters to maximize the conversion of impurities while minimizing product loss. The process begins by dissolving the crude progesterone feedstock, which may contain up to 3% aldehyde impurities, in a volume of alcohol solvent ranging from 3 to 30 times the weight of the crude material. An acid catalyst is then added at a loading of 0.005 to 0.05 equivalents, and the mixture is stirred at temperatures between 20°C and 60°C. Monitoring via thin-layer chromatography (TLC) is essential to confirm the complete disappearance of the aldehyde spots before proceeding. Once the reaction is complete, the acid is neutralized with an organic base like triethylamine or pyridine to prevent any potential acid-catalyzed degradation during the workup. The detailed standardized synthesis steps, including specific solvent swaps and crystallization cooling rates, are outlined in the guide below.
- Dissolve crude progesterone containing aldehyde impurities in an alcohol organic solvent (e.g., methanol, ethanol) and add an acid catalyst such as p-toluenesulfonic acid.
- Stir the mixture at 20-60°C until TLC analysis confirms the aldehyde impurities have completely reacted to form acetals.
- Neutralize with an organic base, concentrate to remove the alcohol solvent, add a second organic solvent (e.g., methyl tert-butyl ether), cool to -20 to 5°C, and filter to isolate high-purity progesterone crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acetalization-based purification method translates directly into enhanced operational efficiency and risk mitigation. The elimination of hazardous oxidants like chromic anhydride removes the regulatory and logistical burdens associated with handling and disposing of Class 1 carcinogens and heavy metals. This shift not only aligns with increasingly strict environmental regulations but also simplifies the facility's safety protocols, reducing insurance premiums and audit complexities. Furthermore, the use of commodity chemicals such as methanol, ethanol, and p-toluenesulfonic acid ensures that the supply chain is resilient against raw material shortages, as these reagents are globally available from multiple vendors. The simplicity of the unit operations—primarily stirring, concentration, and filtration—means that the process can be executed in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. This accessibility significantly lowers the barrier to entry for contract manufacturing organizations looking to expand their steroid portfolio.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the removal of expensive and hazardous reagents, as well as the reduction in waste treatment costs. By avoiding the use of chromium-based oxidants, manufacturers eliminate the need for costly heavy metal scavenging resins and specialized wastewater treatment facilities required to meet discharge limits. Additionally, the high recovery yield of over 90% ensures that valuable starting material is not lost to degradation or excessive mother liquor retention, directly improving the overall material throughput. The mild reaction conditions also result in lower energy consumption for heating and cooling compared to processes requiring extreme temperatures or vacuum distillation. These factors combine to create a leaner cost structure, allowing for competitive pricing in the global API market without sacrificing margin.
- Enhanced Supply Chain Reliability: Reliability in the supply of high-purity hormones is paramount for downstream drug product manufacturers, and this method enhances consistency by reducing batch-to-batch variability. The robustness of the acetalization reaction against minor fluctuations in temperature or stoichiometry means that off-spec batches are less likely to occur, ensuring a steady flow of compliant material. Moreover, the short cycle time associated with the simple workup procedure allows for faster turnover of production campaigns, enabling suppliers to respond more agilely to market demand spikes. The ability to consistently meet the European Pharmacopoeia 9.0 specification of <0.6% bis-aldehydes provides a strong quality assurance guarantee, reducing the risk of customer rejections and supply disruptions. This stability is crucial for long-term supply agreements where continuity is valued as highly as price.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the absence of exothermic hazards or gas evolution typically associated with oxidation reactions. The reaction is homogeneous and easy to control, making it ideal for large-scale batch reactors where heat transfer can be a limiting factor in other chemistries. From an environmental perspective, the process generates minimal hazardous waste, as the primary byproducts are acetals and salts that are easier to manage than heavy metal sludge. The solvents used are largely recoverable and recyclable, further reducing the environmental footprint and aligning with green chemistry principles. This sustainability profile is increasingly becoming a key differentiator in vendor selection processes for major pharmaceutical companies committed to reducing their Scope 3 emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this impurity removal technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing lines. The answers reflect the specific performance metrics achieved in the examples, such as yield and purity, offering a realistic expectation of the process outcomes.
Q: How does this method remove bis-aldehyde impurities from progesterone?
A: The method utilizes an acid-catalyzed reaction where aldehyde impurities react with an alcohol solvent to form acetals. These acetals have different solubility properties compared to progesterone, allowing them to remain in the mother liquor while pure progesterone crystallizes out.
Q: What purity levels can be achieved with this purification technique?
A: This process consistently yields progesterone with an HPLC content greater than 99.5% and reduces total aldehyde impurities to below 0.2%, meeting strict European Pharmacopoeia 9.0 standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses mild reaction conditions (20-60°C), commercially available reagents, and simple filtration steps, making it highly scalable and cost-effective for commercial manufacturing without requiring complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Progesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising purification technologies like the acetalization method described here can be seamlessly integrated into your supply chain. We are committed to delivering materials that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against pharmacopoeial standards before release. Our facility is equipped to handle the specific solvent systems and crystallization requirements of steroid hormones, guaranteeing that the high purity and low aldehyde profiles achieved in the patent examples are replicated at scale. By choosing us as your strategic partner, you gain access to a supply chain that prioritizes quality, compliance, and continuous improvement.
We invite you to engage with our technical procurement team to discuss how this advanced purification method can optimize your specific project requirements. Whether you are looking to upgrade an existing process or develop a new supply line for high-purity progesterone, we are prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data from our recent batches and comprehensive route feasibility assessments. Our goal is to support your R&D and commercial goals with transparent data and reliable execution, ensuring that your projects move forward without interruption.
