Advanced Progesterone Purification: Scaling High-Purity Steroid Manufacturing
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and yield of critical hormonal APIs, particularly when transitioning from fermentation-derived precursors. A significant breakthrough in this domain is detailed in patent CN112250727A, which introduces a novel purification strategy for progesterone synthesized from phytosterol fermentation products. This technology addresses the persistent challenge of removing structurally similar intermediates that plague traditional downstream processing. By leveraging a specific oxidative washing protocol involving sodium hypochlorite within an ethyl acetate system, the method achieves a purity exceeding 99.5% while significantly mitigating the yield losses associated with repetitive recrystallization. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and economically viable steroid manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
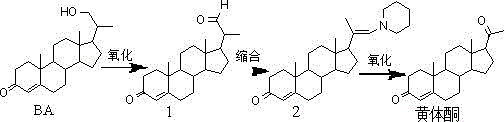
Historically, the production of progesterone from phytosterol fermentation product BA has been hindered by the presence of persistent intermediate impurities, specifically referred to as Compound (1) in the synthetic pathway. These impurities are notoriously difficult to separate because their solubility and polarity profiles in common organic solvents, such as ethanol, are nearly indistinguishable from the target progesterone molecule. Consequently, manufacturers relying on conventional ethanol recrystallization are forced to perform the purification cycle more than three times to meet quality standards. This repetitive processing not only drastically reduces the overall refining yield due to mechanical and solubility losses but also increases solvent consumption and energy usage, creating a bottleneck for cost-effective commercial scale-up of complex steroid intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112250727A circumvents these physical limitations through a chemically driven purification mechanism. The process initiates by dissolving the crude progesterone in ethyl acetate, followed by the strategic addition of methanol and sodium hypochlorite. This oxidative treatment effectively differentiates the impurity profile, likely modifying the polar characteristics of the stubborn intermediate Compound (1) without compromising the integrity of the progesterone skeleton. Following this reaction, a series of aqueous washes with potassium carbonate and water efficiently extract the modified impurities into the aqueous phase. This approach streamlines the workflow into a single, high-efficiency purification sequence rather than multiple recrystallization loops.
Mechanistic Insights into Oxidative Impurity Removal
The core innovation of this technology lies in the selective reactivity of sodium hypochlorite within the biphasic ethyl acetate-methanol system. While progesterone contains a conjugated enone system that is relatively stable under these specific mild oxidative conditions, the intermediate impurities often possess reactive functional groups, such as unprotected hydroxyls or reactive double bonds at different positions, which are susceptible to oxidation or chlorination. By introducing sodium hypochlorite, the process chemically alters these impurities, increasing their polarity or converting them into water-soluble salts. This chemical transformation is crucial because it breaks the azeotropic-like solubility behavior that makes physical separation so difficult, allowing for a clean partition between the organic product layer and the aqueous waste layer.
Furthermore, the subsequent washing steps with potassium carbonate solution serve a dual purpose of neutralizing any acidic byproducts generated during the oxidation and ensuring the complete removal of residual hypochlorite species. This rigorous washing protocol is essential for meeting the stringent purity specifications required for hormonal APIs, where trace oxidative residues could pose stability issues or safety concerns. The final crystallization from the concentrated ethyl acetate layer at controlled low temperatures (0-5°C) ensures that the crystal lattice forms exclusively around the pure progesterone molecules, excluding any remaining trace solvents or minor byproducts, thereby locking in the high purity profile of the final active pharmaceutical ingredient.
How to Synthesize Progesterone Efficiently
The implementation of this purification route requires precise control over stoichiometry and temperature to maximize the differential solubility effects. The process is designed to be scalable, moving seamlessly from laboratory validation to industrial production tanks. Operators must adhere to the specific weight ratios of ethyl acetate to crude product (8-10:1) and maintain the oxidative treatment temperature between 40-50°C to ensure optimal reaction kinetics without degradation. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are outlined in the guide below.
- Dissolve crude progesterone in ethyl acetate at 40-50°C, then treat with methanol and sodium hypochlorite to modify impurity profiles.
- Perform sequential aqueous washes using 10% potassium carbonate solution followed by water until the organic layer reaches neutrality.
- Concentrate the ethyl acetate layer under reduced pressure, cool to 0-5°C for crystallization, and dry to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates directly into enhanced operational efficiency and reduced total cost of ownership. By eliminating the need for multiple recrystallization cycles, the process drastically reduces the volume of solvents required and the energy consumed for heating and cooling loops. This simplification of the downstream processing unit operations allows for faster batch turnover times, enabling manufacturers to respond more agilely to market demand fluctuations without compromising on the quality of the high-purity progesterone supplied to downstream formulators.
- Cost Reduction in Manufacturing: The most significant economic driver here is the improvement in yield. By avoiding the cumulative losses associated with three or more recrystallization steps, the process recovers a substantially higher percentage of the valuable hormone from the crude feedstock. Additionally, the reduction in solvent usage lowers both the raw material costs for solvents and the waste disposal costs associated with spent mother liquors. This leaner manufacturing approach ensures that cost reduction in steroid manufacturing is achieved through process intensification rather than simply negotiating lower raw material prices.
- Enhanced Supply Chain Reliability: A simplified process with fewer unit operations inherently carries lower operational risk. There are fewer transfer points where product loss or contamination can occur, and the reduced processing time means that production slots are freed up more quickly. This efficiency contributes to reducing lead time for high-pularity steroids, ensuring a more consistent and reliable flow of materials to clients who depend on just-in-time delivery models for their own formulation schedules.
- Scalability and Environmental Compliance: The use of ethyl acetate, a solvent with a favorable environmental profile compared to chlorinated alternatives often used in older steroid chemistry, aligns with modern green chemistry initiatives. The process improves the utilization rate of the progesterone mother liquor, meaning less hazardous hormone waste is generated per kilogram of product. This facilitates easier compliance with increasingly strict environmental regulations regarding pharmaceutical effluent, making the commercial scale-up of complex steroid intermediates more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this purification technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in both purity and yield metrics.
Q: Why is conventional ethanol recrystallization inefficient for progesterone purified from phytosterols?
A: Conventional methods struggle because the intermediate impurity (Compound 1) possesses solubility and polarity characteristics nearly identical to progesterone in common solvents like ethanol. This similarity necessitates repetitive recrystallization cycles (often more than three times), leading to substantial product loss and low overall refining yields.
Q: What is the critical role of sodium hypochlorite in this purification protocol?
A: Sodium hypochlorite acts as a selective oxidative agent that likely modifies the chemical structure or polarity of the stubborn intermediate impurities without degrading the progesterone backbone. This chemical differentiation allows for efficient phase separation during the subsequent aqueous washing steps, effectively removing contaminants that physical recrystallization cannot.
Q: What purity levels and yield improvements does this patented method offer?
A: The method consistently achieves a final product purity of greater than or equal to 99.5%. Compared to prior art techniques, this process improves the overall yield of the refined product by approximately 5%, while simultaneously enhancing the utilization rate of the mother liquor and reducing environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Progesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the oxidative purification of progesterone are translated into reliable supply. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, including the >99.5% benchmark set by this advanced methodology.
We invite global partners to collaborate with us to leverage these process efficiencies for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can secure your supply of high-quality hormonal intermediates while optimizing your overall procurement budget.
